Team:UNIPV-Pavia/Notebook/Week5May
From 2009.igem.org
(Difference between revisions)
(→May, 27th) |
|||
| Line 148: | Line 148: | ||
*We diluted 1:10 the 5 ml overnight cultures of J23100, J23101, J23118, and E0240 in 5 ml of LB + Amp. | *We diluted 1:10 the 5 ml overnight cultures of J23100, J23101, J23118, and E0240 in 5 ml of LB + Amp. | ||
*We incubated the diluted cultures at 37°C, 220 rpm for 4 and 1/2 hours. | *We incubated the diluted cultures at 37°C, 220 rpm for 4 and 1/2 hours. | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<table width="100%"> | <table width="100%"> | ||
Latest revision as of 10:17, 21 October 2009

|
|
|
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
Week from May 25th, to May 31st, 2009
 Previous Week
Previous Week
|
Next Week

|
May, 25th
- iGEM starts now!!!
- We planned to dedicate the first two weeks to prepare a glycerol stock for every BioBrick of interest for our project. The following weeks will be dedicated to the BioBrick assembly.
- DNA resuspension from iGEM 2009 plates. We resuspended all the high quality constitutive promoter family members from UCBerkeley collection and GFP protein generator:
|
- Transformation of resuspended DNA (2 ul) in TOP10 E. coli, plated transformed bacteria and incubated the plates overnight at 37°C.
May, 26th
- All the 15 incubated plates showed colonies!
Moreover, many UCBerkeley promoter family members showed red fluorescent colonies, according to the ranking of promoters' estimated strength (see http://partsregistry.org/Promoters/Catalog/Anderson).
|
|
|
Preparation of the first experiment with Tecan F200
|
|
May, 27th
- We wanted to resuspend Q04400 and Q04121 in order to build up respectively an aTc and an IPTG/lactose sensor, but we realized that MIT QC classified their sequences as "inconsistent"...
- DNA resuspension from iGEM 2009 plates. We resuspended three lysis-involved parts, a double transcriptional terminator and lacZ transcriptional unit in order to build up a beta-galactosidase protein generator (last year, a beta-galactosidase protein generator was available in 2008 Distributon - I732950 - but its sequence was almost completely wrong):
|
- Only B0015 and I732017 QC sequence analysis was available...we hoped that the three lysis parts' sequences were correct!
- Transformation of resuspended DNA (2 ul) in TOP10 E. coli, plated transformed bacteria and incubated the plates overnight at 37°C.
- We planned how to substitute Q04400 and Q04121. We searched for smaller BioBricks to re-build them with a minimum number of assemblies:
- Q04400 = P0440 (available in iGEM 2009 plates) + R0040 (available in iGEM 2009 plates)
- Q04121 = P0412 (not available in iGEM 2009 plates) + R0011 (available in iGEM 2009 plates)
Preparation of the first experiment with Tecan F200
- We diluted 1:10 the 5 ml overnight cultures of J23100, J23101, J23118, and E0240 in 5 ml of LB + Amp.
- We incubated the diluted cultures at 37°C, 220 rpm for 4 and 1/2 hours.
|
|
|
May, 28th
- All the five overnight plates showed colonies!
- We picked a colony for each plate to infect 1 ml of LB + Amp. We incubated the inocula at 37°C, 220 rpm for 5 and 1/2 hours.
- Glycerol stocks for the grown cultures.
- TBE 5X preparation.
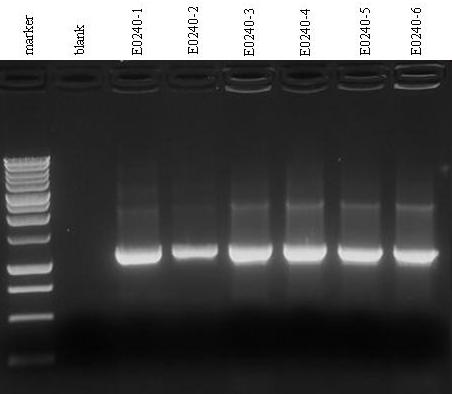
- Colony PCR to check VF2 and VR primers quality. The primers had been ordered the previous year for iGEM 2008. We decided to perform this check on six colonies of E0240 plate.
- Electrophoresis for PCR reactions.
- Gel results (picture not available): no band could be seen on the gel (only the ladder)...Strange result, we will repeat the experiment tomorrow.
May, 29th
|
|
 Previous Week
Previous Week
|
Next Week

|
 "
"