Team:Paris/Production overview
From 2009.igem.org
iGEM > Paris > Vesicle production system > Overview
Contents |
Vesicle production system : Main
Several gram-negative bacteria - including E. coli - have been shown to produce vesicles for various hypothetical reasons[1][2] (stress response, host-pathogen interaction...). In our project, we want to optimize vesicle production to develop a long distance communication system between gram-negative bacteria. Outer membrane vesicles (OMVs) production can be enhanced through the destabilization of the outer-membrane. We found in the literature that the Tol/Pal system could be a good target for this purpose[3]. The Tol/Pal system of Escherichia Coli is involved in anchoring the outer- to the inner-membrane and to the peptidoglycan layer. It is thus essential in maintaining membrane integrity. The system is composed of five membrane proteins (TolA, TolB, TolQ, TolR and Pal) associating in two complexes.[4][5]
See Tol/Pal part for more information and why we choose this system. Strategy and Construction part explains our aim in this OMV production system.
Vesicle production system : Tol/Pal
E. Coli is gram-. Its membrane is a complex that included: the inner membrane and the outer membrane. Between both, there is a layer of peptidoglycan. The outer membrane presents lipo-polysaccharides (LPS). We find also porins and other proteins. There are receivers for the entrance of nourishing elements, receivers in pili (bacterial conjugation), receivers where settles bacteriophage. It has properties of selective permeability, protection and support. This complex of proteins is in place to maintain the stability between the inner membrane and the outer membrane.
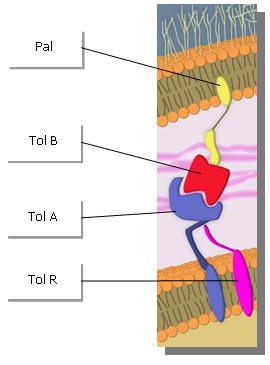
In particular the system called Tol-Pal consisted of five proteins, TolA, TolB, TolQ, TolR and Pal[3][4]. This complex allows to maintain the integrity of the membrane. Studies showed that if we destabilize this system, we also destabilize the membrane, leading then a production of vesicles. But when the membrane of the bacteria stays for such a long time to this state of destabilization, the bacteria produces too many vesicles and go in state of lysis[4].
What is Tol-Pal complex ?
Five proteins organised this complex[5]. The TolA/Q/R proteins form a protein complex in the inner membrane. TolB is a periplasmic protein associated with Pal, a lipoprotein. Pal is anchored to the outer membrane and interacts with the peptidoglycan layer. There are interactions with TolA–Pal and TolA–TolB.
Vesicle production system : Our strategy
We want to destabilize the outer membrane to create outer membrane vesicles (OMVs).
OMVs were formed upon periplasmic overproduction of soluble TolA and TolR domains, since these two short periplasmic domains (both less than 100 residues) have been previously found to disturb the Tol–Pal system[6][7]. (TolA and TolR have three domains: Nterminal, central and Cterminal domains)
An other study[5] focused on the development of a gene expression system able to induce production of large amounts of OMVs when they used different domains of these two proteins of Tol-Pal system. This team [5] send us their plasmid and we were able to begin our work quickly.
To achieve our goal, we decided to do like the Lloubès Team<[5] and take advantage of the soluble central domain of TolR (TolRII). We don’t use the third domain of TolA because, it doesn’t work so well and TolAIII have lot of PstI domain in his sequence. In order to achieve our project, we will over express specifically designed biobricks containing TolRII fused with OmpA signal which allows it to migrate in the periplasm[4]. So Tol-Pal system will become bad and the membrane integrity will be destabilised. Lot of vesicle could be creating.
If bacteria stay in this conformation, there is lysis. We try to create an ON/OFF system to stop the vesicles creation.
In the same framework, we could also over express various Tol ligand (like colicin) to destabilize the membrane. But it doesn’t work very well.[5]
BioBrick TolR domain II N-term fusion : Bba K257005
BioBrick ompA sequence signal : Bba_K257006
You can see now our Construction.
References
- ^Release of outer membrane vesicles by Gram-negative bacteria is a novel envelope stress response. McBroom AJ & Kuehn MJ 2007 - 17163978
- ^Biogenesis of bacterial membrane vesicles. Deatherage BL & Cookson BT 2009 - 19432795
- ^Transmembrane a-helix interactions are required for the functional assembly of the escherichia coli Tol complex. Lazzaroni & Geli 1995 - 179564
- ^The Tol-Pal proteins of the Escherichia coli cell envelope an energized system required for outer membrane integrity. Lloubès & Journet 2001 - 11501670
- ^Improved methods for producing outer membrane vesicles in gram-negative bacteria. Henry & Lloubès 2004 - 15249060
- ^Role of TolR N-terminal, central, and C-terminal domains in dimerization and interaction with TolA and tolQ. Journet L & Bénédetti H. 1999 - 10419942
- ^Role of the carboxyl-terminal domain of TolA in protein import and integrity of the outer membrane. Levengood-Freyermuth SK & Webster RE. 1993 - 8416897
 "
"