Team:Newcastle/Labwork/21 September 2009
From 2009.igem.org
(→Formal Lab Session - 21st September 2009) |
(→Formal Lab Session - 21st September 2009) |
||
| Line 53: | Line 53: | ||
===Introduction=== | ===Introduction=== | ||
| + | * We have already got the double clone,the next thing need to do is to ligate this sleB:cwlJ fragment with pMutin4 plasmid to get the pspac sequence from pMutin4. pMutin4 contains HindIII and BamHI restriction sites, but our double clone sequence didn't contain those sites. If we want to ligate double clone fragment with pMutin4 backbone, we need PCR our double clone fragment and add HindIII and BamHI restriction sites.We designed cwlJsleB primers here. | ||
| + | * We digest pMutin4 with HindIII and BamHI to get our plasmid backbone. | ||
| + | * Midi Prep L1 and L2 transformation strains to get large amount of plasmid. | ||
| + | * Our kinA synthesized part was arrived, we need to construct this gene into our integrate plasmid and transform it into B.subtilis.Before the characterization of kinA, we need to | ||
| + | <br> | ||
| + | ===Experiment procedure=== | ||
| + | ==== PCR doulbe clone fragment ==== | ||
| + | * Prepare buffer mix and Taq | ||
| + | |||
| + | Reaction buffer 10ul x 4 = 40ul | ||
| + | dNTP 2ul x 4 = 8ul | ||
| + | -------------------------------- | ||
| + | 48ul | ||
| + | 12ul for each reaction | ||
<br> | <br> | ||
| + | GoTaq(5U/ul) 0.25ul x 2 = 0.5ul | ||
| + | ddH2O 4.5ul | ||
| + | ---------------------------------- | ||
| + | 5ul | ||
| + | 2.5ul for each reaction | ||
| - | |||
| - | = | + | {| border="1" |
| - | + | |+ Exp. PCR experiment for gene cwlJ:sleB | |
| + | ! No. !! Water (ul) !! PCR Mix (ul) !! Forward Primer (ul) !! Reverse Primer (ul) !! Template (ul) !! Polymerase used (ul) !! Total Volume (ul) | ||
| + | |- | ||
| + | | 1 || 28.5 || 12 || cwlJsleBForward 2.5ul || cwlJsleBReverse 2.5ul || pSB1AT3:cwlJ:sleB 2ul || GoTaq 2.5ul || 50 | ||
| + | |- | ||
| + | | 2 || 30.5 || 12 || cwlJsleBForward 2.5ul || cwlJsleBReverse 2.5ul || 0ul || GoTaq 2.5ul|| 50 | ||
| + | |- | ||
| + | |} | ||
| + | <br> | ||
| + | * PCR procedure: STD50 | ||
| + | <br> | ||
| + | 95C 2min -->[95C 30S --> 50C 30S --> 75C 1min] 30 runs --> 75C 5min --> store at 4C | ||
| - | ==== | + | ==== Midi Prep ==== |
| + | * Before we carried out Midi Prep, we take out 1ml of cell mix and put them into 1.5ml eppendorf tube to keep in fridge. | ||
| + | * 50ml of cells were used for Midi Prep. | ||
| + | * Midi prep processed by following the standard protocal of Plasmid Midi Prep Kit(Sigma). | ||
| + | ==== Digest pMutin4 plasmid for ligation ==== | ||
| + | * Prepare the digest reaction. | ||
| + | dd H2O 7ul | ||
| + | 10X fast digest buffer 7ul | ||
| + | Fast HindIII 3ul | ||
| + | Fast BamHI 3ul | ||
| + | pMutin4 plasmid 50ul | ||
| + | ------------------------------ | ||
| + | 70ul | ||
| + | * Incubate 1 hour at 37 degree. | ||
| - | === | + | ==== Digest kinA fragment ==== |
| + | * kinA gene is one of the sporelation related genes | ||
| + | |||
| + | |||
| + | ==== Prepare cwlJ and sleB fragment for Plan B==== | ||
<br> | <br> | ||
| - | + | ||
| + | |||
| + | |||
| + | |||
| + | ===Conclusion=== | ||
| + | |||
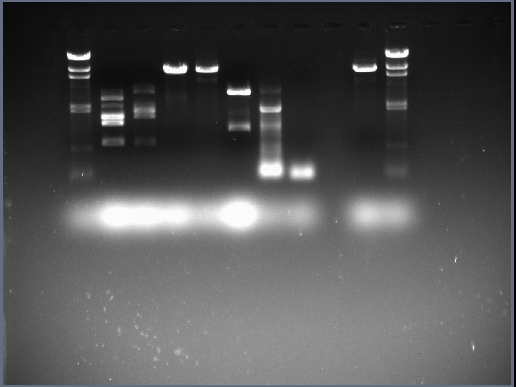
| + | * The concentration of our Midi Prep plasmid pSB1AT3:sleB is 109.3ng/ul. | ||
===Futher plan=== | ===Futher plan=== | ||
| + | * Ligate cwlJ with pSB1AT3:sleB and transform the ligated plasmid into E.coli. | ||
{{:Team:Newcastle/Project/Labwork/CalTemplate}} | {{:Team:Newcastle/Project/Labwork/CalTemplate}} | ||
Revision as of 21:46, 20 October 2009
Formal Lab Session - 21st September 2009
Stochastic switch team
Today we digested the sac minipreps with EcoRI and XbaI. It seems clear that there was something wrong with the minipreps (such as poor procedure) as there seemed to be little or no DNA in the sample.
Did a gel fragment clean up on the sspb fragment cut from the gel, this was put in the 'fragments' box in the freezer ready for later.
50ul of ara+pSB1AT3 plasmid was cut with SpeI and PstI in order to insert the sspb fragment after the operator. Rather than doing a gel fragment prep a PCR clean up (kit) was done on the digest to save time.
We ligated the ara + sspb fragments using Chris' method:
- 2ul Ligase buffer
- 2.5ul Vector DNA
- 14.5ul insert DNA
- 1ul ligase
= 20ul
This was left in the fridge overnight to improve the ligation conditions.
Metal Sensing Team
Introduction
Practical Outline
Here are the list of tasks which need to be accomplished by the end of the day:
- Run analysis digests of the 5 midi-preps of cotC, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3 (midi-prepped in the 11/09/09 lab session and digested in the 18/09/09 lab session)
- If successful, double digest 40ul of pMUTIN4 with BamHI and HindIII and place under DNA gel electrophoresis; excise band produced and extract DNA
- If unsuccessful, inoculate 5ml of LB with E. coli containing pMUTIN4 (for mini-preps and midi-preps)
- Check PCR reaction (amplification of cotC-GFP-smtA) on gel and clean up if successful
- If successful, digest with BamHI and HindIII
- Should the pMUTIN4 DNA extraction be a success and the cotC-GFP-smtA BioBrick be successfully amplified (by PCR) and extracted, ligate the two fragments together.
- Reactions include:
- vector + insert + ligase
- vector + ligase
- vector and NO ligase
- Reactions include:
- Inoculate 3ml of LB with TPA2 cells (for production of competent DH5-alpha E. coli cells)
Procedure
Analysing midi-prep digests
Double digesting pMUTIN4 midi-prep DNA
Cleaning up cotC PCR product
Excising digested pMUTIN4 from gel
Quantification of cotC and pMUTIN4
Chassis team
Introduction
- We have already got the double clone,the next thing need to do is to ligate this sleB:cwlJ fragment with pMutin4 plasmid to get the pspac sequence from pMutin4. pMutin4 contains HindIII and BamHI restriction sites, but our double clone sequence didn't contain those sites. If we want to ligate double clone fragment with pMutin4 backbone, we need PCR our double clone fragment and add HindIII and BamHI restriction sites.We designed cwlJsleB primers here.
- We digest pMutin4 with HindIII and BamHI to get our plasmid backbone.
- Midi Prep L1 and L2 transformation strains to get large amount of plasmid.
- Our kinA synthesized part was arrived, we need to construct this gene into our integrate plasmid and transform it into B.subtilis.Before the characterization of kinA, we need to
Experiment procedure
PCR doulbe clone fragment
- Prepare buffer mix and Taq
Reaction buffer 10ul x 4 = 40ul
dNTP 2ul x 4 = 8ul
--------------------------------
48ul
12ul for each reaction
GoTaq(5U/ul) 0.25ul x 2 = 0.5ul
ddH2O 4.5ul
----------------------------------
5ul
2.5ul for each reaction
| No. | Water (ul) | PCR Mix (ul) | Forward Primer (ul) | Reverse Primer (ul) | Template (ul) | Polymerase used (ul) | Total Volume (ul) |
|---|---|---|---|---|---|---|---|
| 1 | 28.5 | 12 | cwlJsleBForward 2.5ul | cwlJsleBReverse 2.5ul | pSB1AT3:cwlJ:sleB 2ul | GoTaq 2.5ul | 50 |
| 2 | 30.5 | 12 | cwlJsleBForward 2.5ul | cwlJsleBReverse 2.5ul | 0ul | GoTaq 2.5ul | 50 |
- PCR procedure: STD50
95C 2min -->[95C 30S --> 50C 30S --> 75C 1min] 30 runs --> 75C 5min --> store at 4C
Midi Prep
- Before we carried out Midi Prep, we take out 1ml of cell mix and put them into 1.5ml eppendorf tube to keep in fridge.
- 50ml of cells were used for Midi Prep.
- Midi prep processed by following the standard protocal of Plasmid Midi Prep Kit(Sigma).
Digest pMutin4 plasmid for ligation
- Prepare the digest reaction.
dd H2O 7ul
10X fast digest buffer 7ul
Fast HindIII 3ul
Fast BamHI 3ul
pMutin4 plasmid 50ul
------------------------------
70ul
- Incubate 1 hour at 37 degree.
Digest kinA fragment
- kinA gene is one of the sporelation related genes
Prepare cwlJ and sleB fragment for Plan B
Conclusion
- The concentration of our Midi Prep plasmid pSB1AT3:sleB is 109.3ng/ul.
Futher plan
- Ligate cwlJ with pSB1AT3:sleB and transform the ligated plasmid into E.coli.
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"