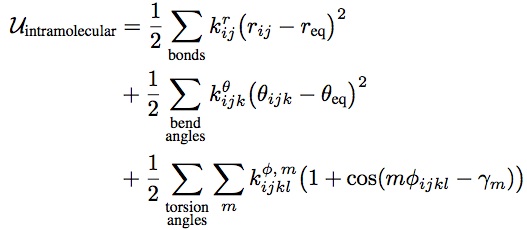Team:EPF-Lausanne/Theory
From 2009.igem.org
Molecular dynamics simulation consists of the numerical, step-by-step, solution of the classical equations of motion. For this purpose we need to be able to calculate the forces acting on the atoms, and these are usually derived from a potential energy. This potential energy can be divided into:
the non-bonded interactions:
- The Lennard-Jones potential is the most commonly used form, with two parameters: σ, the diameter, and ε, the well depth. It takes into account the Van der Waals forces. It represents the non-bonded forces and the total potential energy can be calculated from the sum of energy contributions between pairs of atoms.
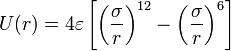
- when electrostatic charges are present, we add the Coulomb force, where Q1, Q2 are the charges and ϵ0 is the permittivity of free space
the bonded interactions:
Angles, bonds and dihedral angles have to be taken into account
To understand a bit more, you can see the following article:
Introduction to Molecular Dynamics Simulation - Michael P. Allen
 "
"