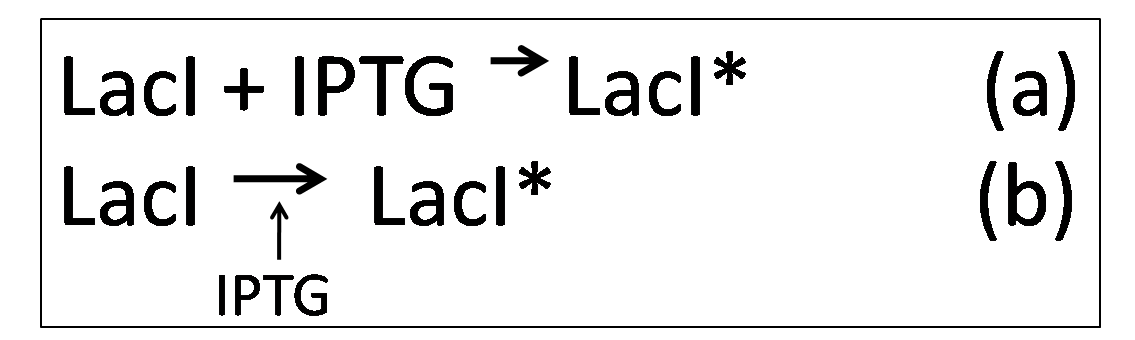Team:Newcastle/Modelling/KinAExpression
From 2009.igem.org
KinA Expression Model
Under normal conditions, LacI represses KinA.
However, in the presence of IPTG, KinA can be expressed, as IPTG binds to LacI, deactivating it. Equations (a) and (b) describes how IPTG binds to LacI, forming LacI*, which is the deactivated form of LacI
Over time, the deactivated form of LacI, LacI* may degrade. This is also taken into consideration while building the model.
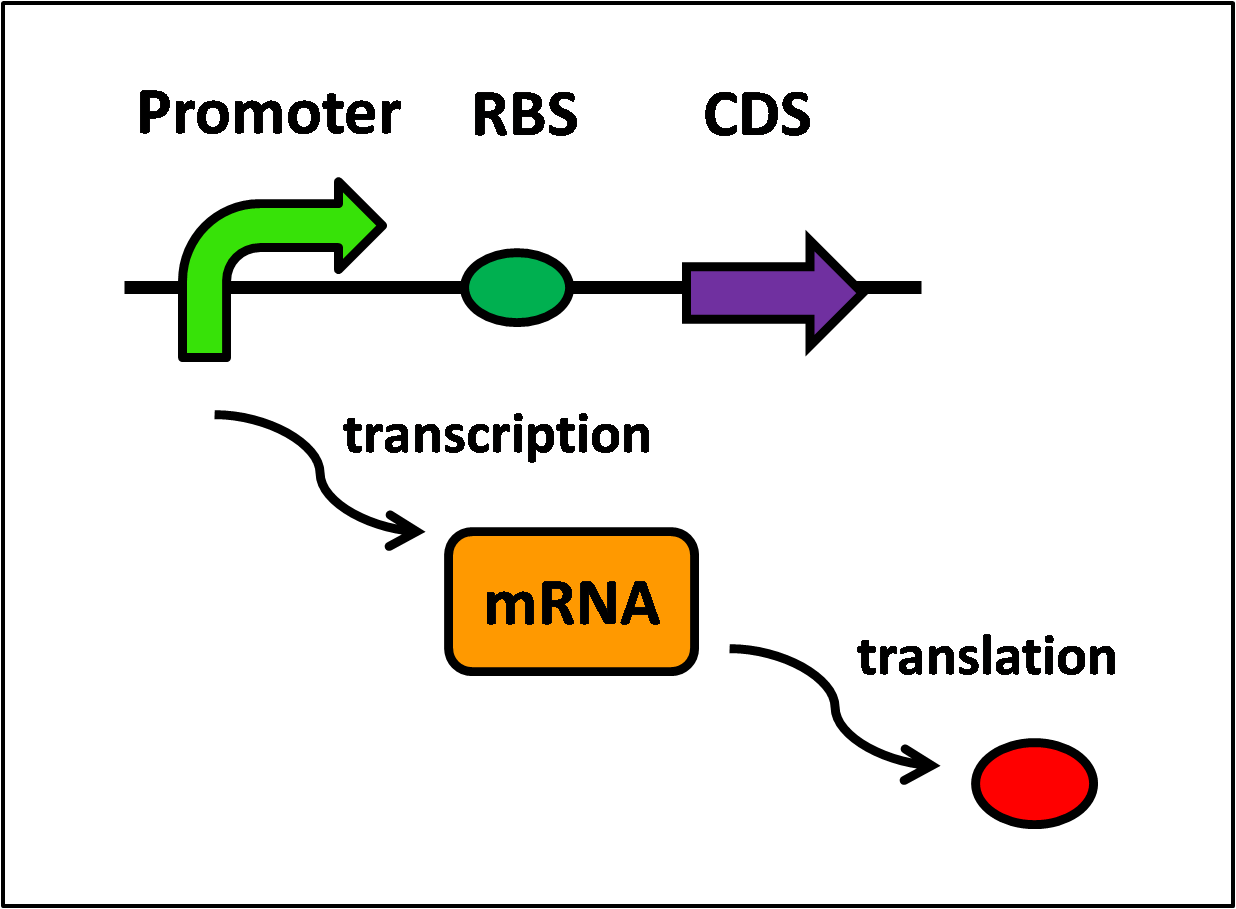
Protein synthesis requires two steps, transcription and translation, and is further illustrated in Figure 1, below.
Referring to Figure 1, the following equations can be written:
- Transcription of LacI,
- Translation of LacI,
where mRNA_LacI is inducing the formation of LacI.
- Transcription of KinA,
where LacI is repressing mRNA_KinA, therefore a lower concentration of LacI would result in a higher concentration of mRNA_KinA.
- Translation of KinA,
where mRNA_KinA is inducing the formation of KinA.
As parameters such as transcription and translation rates could not be found in literature, the following equations were used to calculate the transcription and translation rates whenever possible.
Using the transcription and translation rate equations as seen above, the rates were calculated for the components Spo0A, Spo0B, Spo0F and KinA, and the results are reported in the table below.
For components where information on the CDS length could not be found, we used the value 0.1 for both the translation and transcription rate.
Also, the values for the various rate constants could not be found in literature either. Therefore, the following values were used:
kforward = 0.001
kreverse = 0.05
Other parameters and formulas which were used in this model are as follows:
Protein Degradation Rate = 0.0012
mRNA Degradation Rate = 0.0058
The CDS lengths was found on the [http://www.ncbi.nlm.nih.gov/Genbank/ GenBank] using the accession number, AL009126.
These parameters and functions were also used in the Sporulation Tuning Model.
The KinA Expression Model is available for download as follows:
Results
The above equations were modelled in COPASI, and the following graphs show the behaviour of the system over time at different IPTG concentrations.
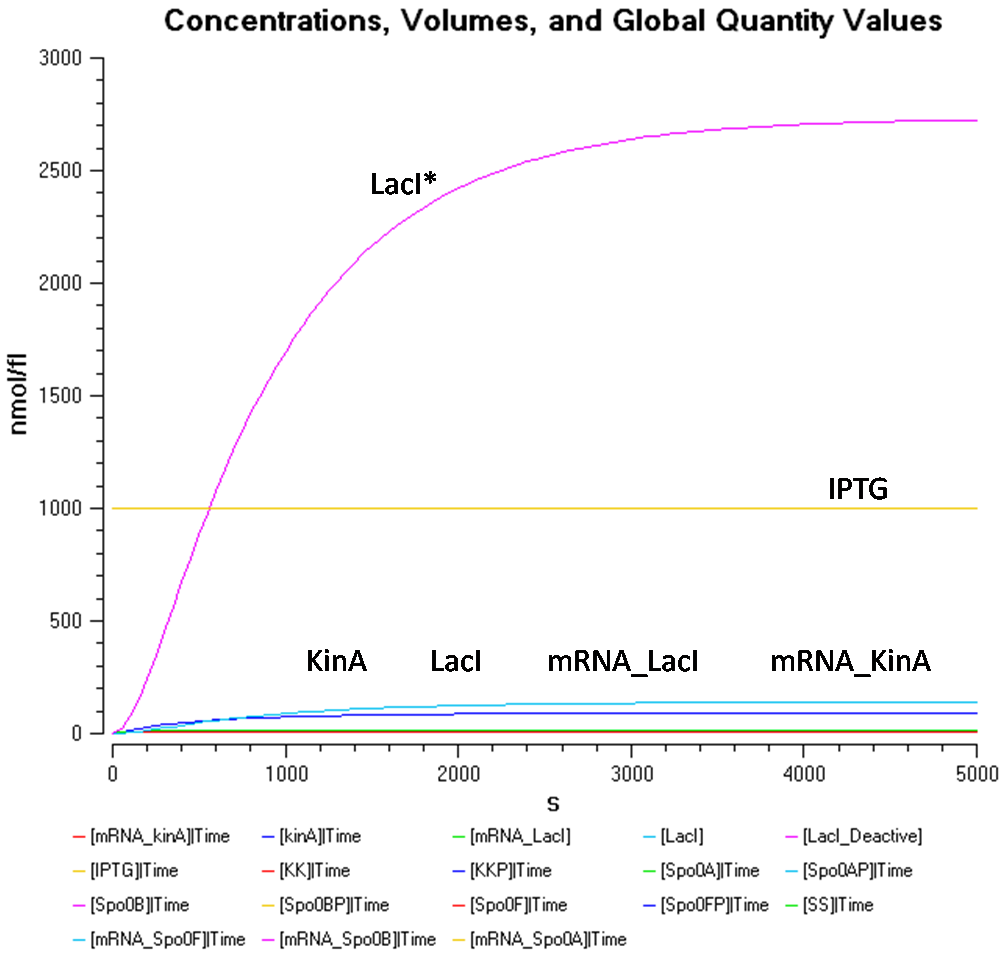
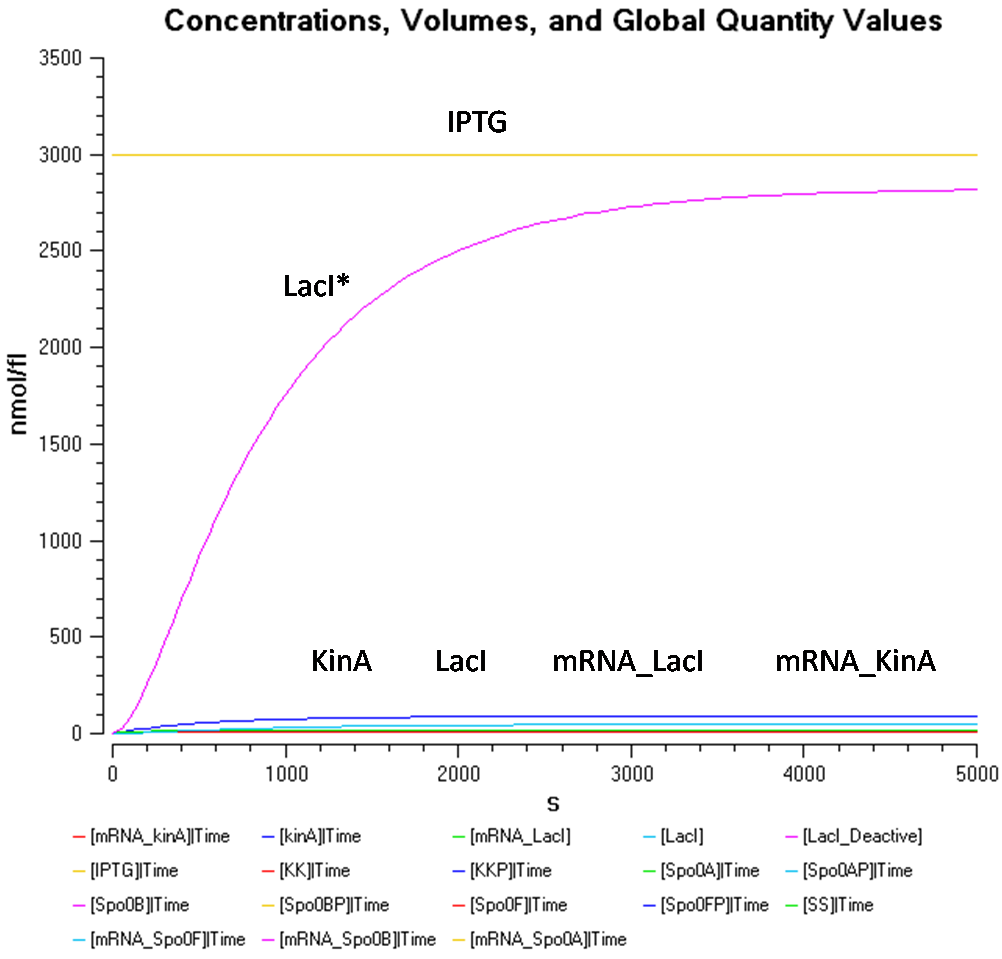
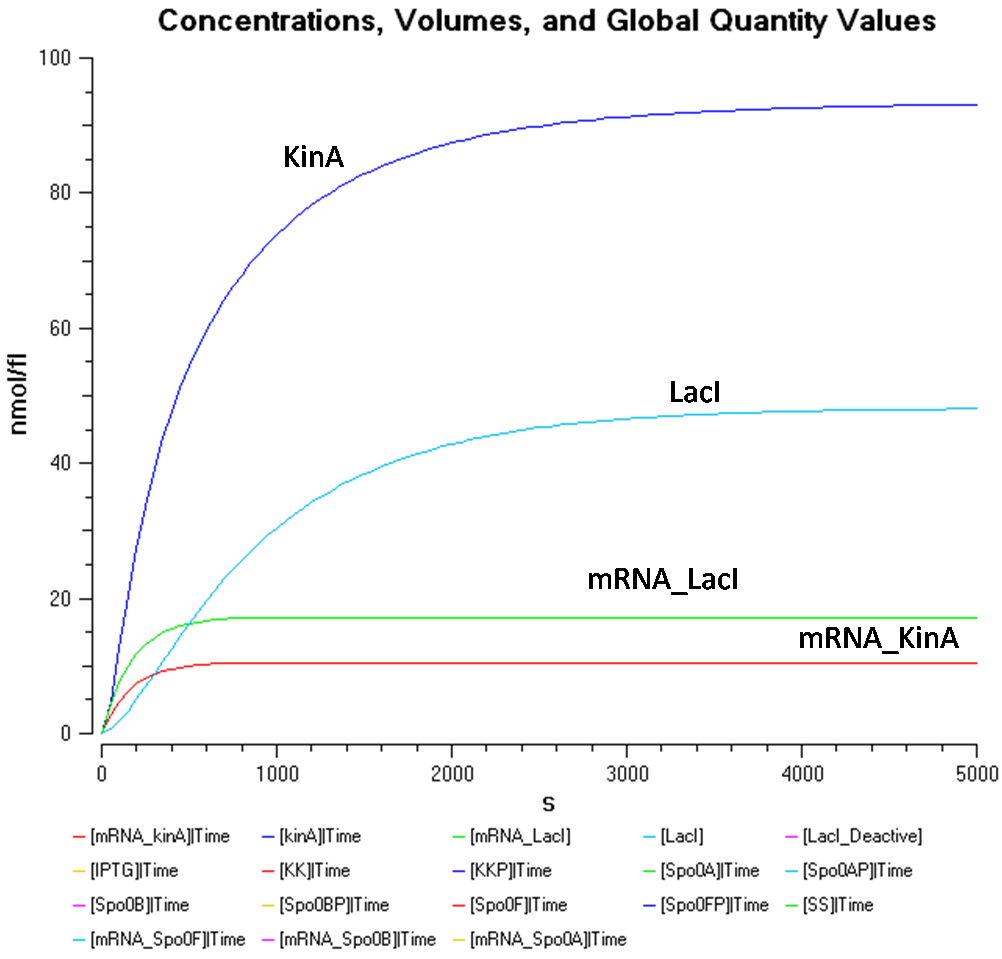
- IPTG concentration = 1000 nmol/fl
In Figure 1.1, at a IPTG concentration of 1000nmol/fl, due to the high concentration of LacI*, it is difficult to view the results for the other species, such as, KinA, LacI, mRNA_KinA, and mRNA_LacI as they are all concentrated at the bottom.
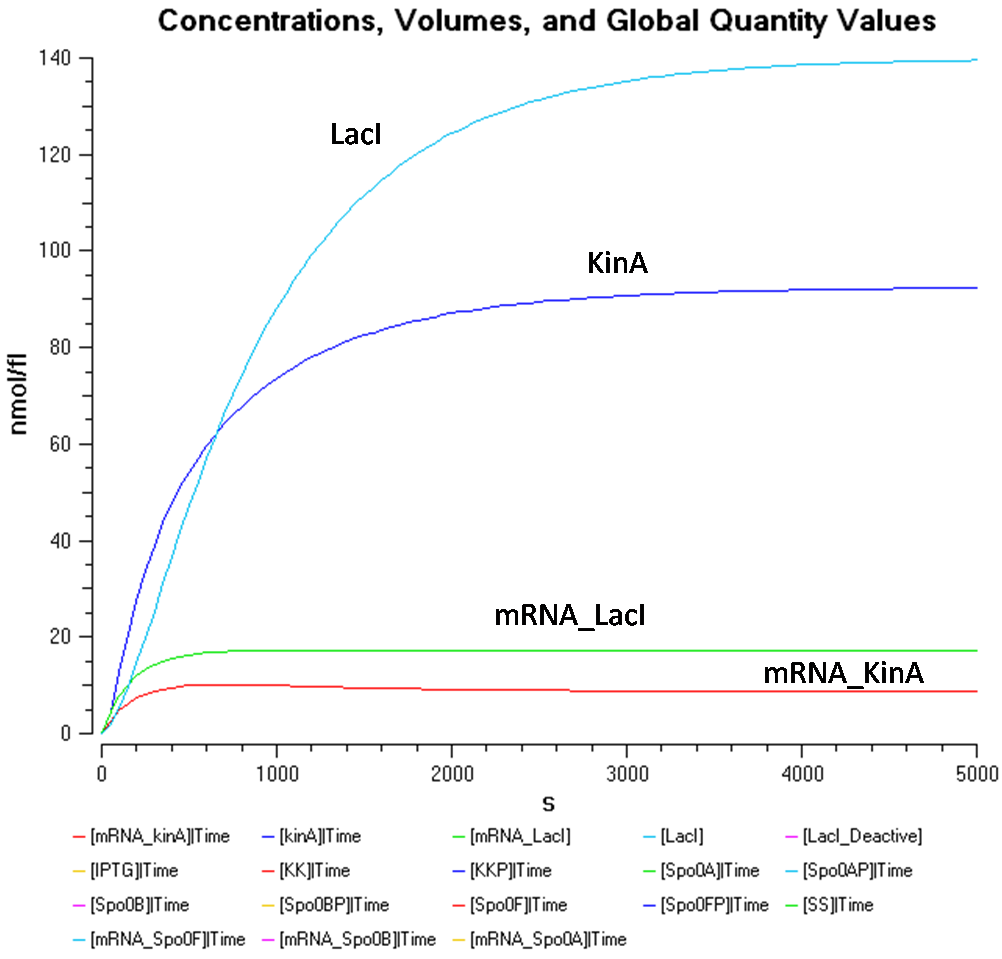
Figure 1.2 shows the behaviour of KinA, LacI, mRNA_KinA and mRNA_LacI more clearly, at the IPTG concentration of 1000nmol/flu.
From Figure 1.1 and 1.2, it is observed that the concentration of LacI* is significantly higher than the other species when the IPTG concentration is set at 1000nmol/fl. Looking at Figure 1.2, while the concentration of KinA is increasing, it is still lower than that of LacI.
- IPTG concentration = 3000 nmol/fl
Comparing Figure 1.1 and 2.1, though the IPTG concentration in Figure 2.1 is 3000nmol/fl, the LacI* concentration is not particularly different.
As the IPTG concentration increases from 1000 to 3000nmol/fl, the KinA concentration is greater than the LacI concentration. However, upon a closer look, the KinA concentration did not increase due to the higher IPTG concentration. The KinA concentration is now higher than the LacI concentration because the LacI concentration decreased.
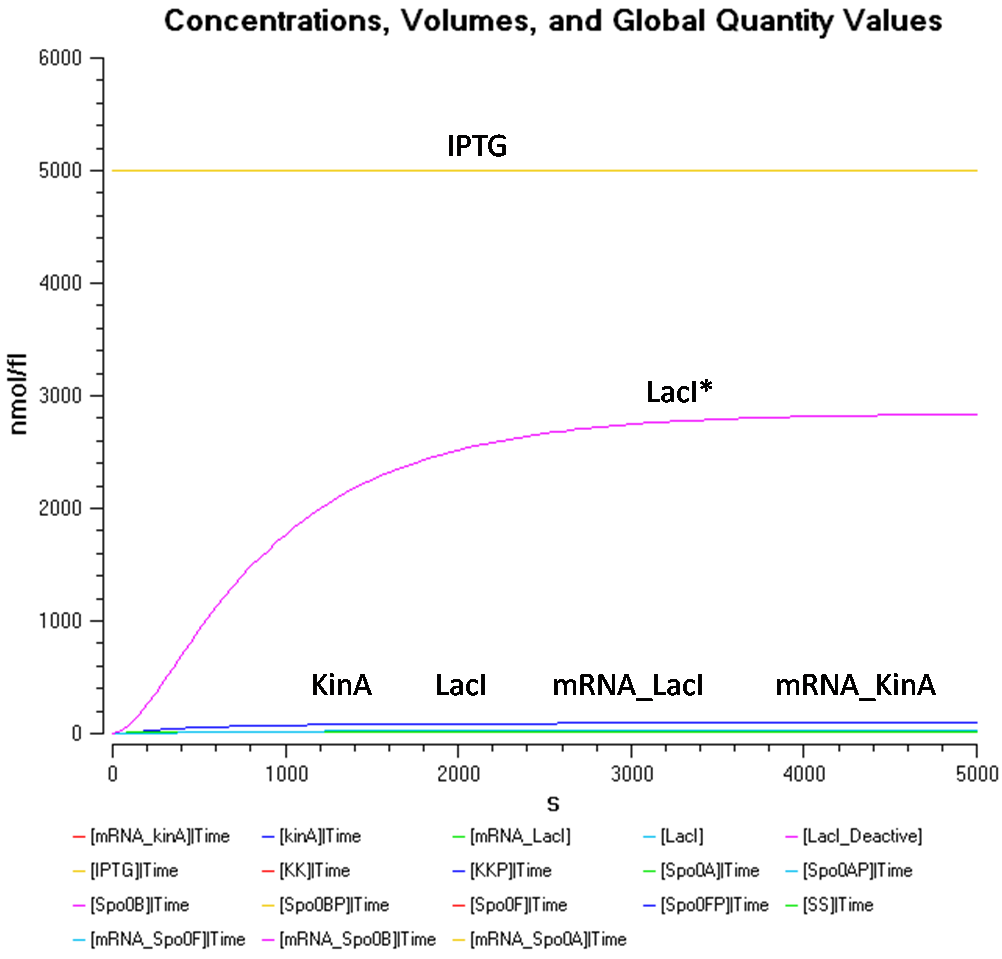
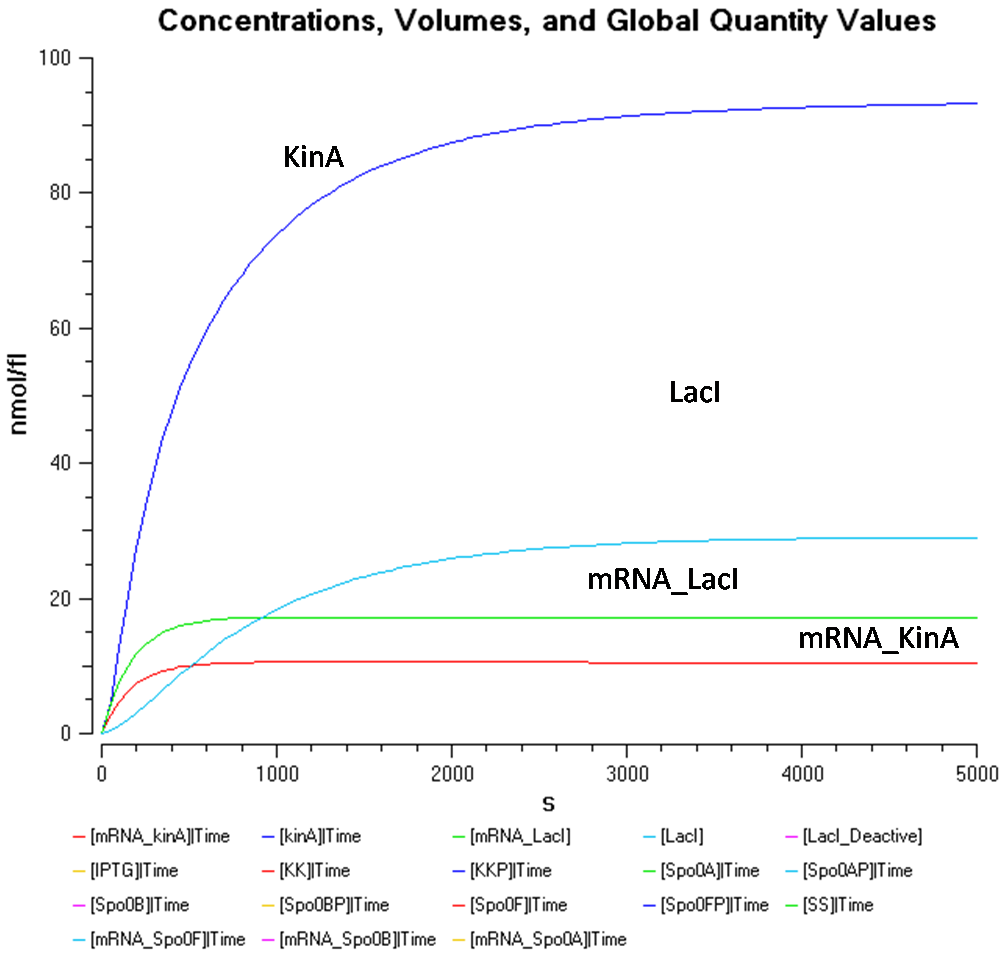
- IPTG concentration = 5000 nmol/fl
Once again, as obsered in Figure 1.1 and 2.1, the concentration of LacI* in Figure 3.1 seems to reach the similar concentrations although the concentration of IPTG has been increased in each case. This seems to say that the concentration of LacI* has reached a threshold concentration.
The KinA concentration in Figure 3.2 is greater than that of LacI once again. However, KinA concentration seems to be stagnant at approximately 90nmol/fl, while the LacI concentration constantly decreases with increased IPTG concentration. Therefore, the IPTG concentration only seems to affect the LacI concentration.
Other Models
- See Sin Operon Model
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"