Notebook > AND Gate 1 > Core > Transfer backbone: lacP+SupD/GFP to low copy pSB4K5
Transfer backbone: lacP+SupD/GFP to low copy pSB4K5
Motivation:
In order to make use of lacIQ of the F plasmid of JM109 (more detail refer to my notes 0817), the AND gates should be build on low copy plasmid. And transfer at this time can save a lot of workload.
Resource:
lacP-SupD-term (K228822): plasmid, from myself; renamed as LS;
lacP-GFP (K228821): plasmid, from myself; LG;
low copy backbone, pSB4K5: vector, has already digested by EcoR1 and Pst1, from ShenShan.
2009.8.21
Plasmid mini prep:
LG, LS1, LS2;
Double digest:
LG, LS1, LS2:
| Pst1 | 1uL
|
| EcoR1 | 1uL
|
| plasmid | 10uL
|
| Buffer | 2uL
|
| water | 6uL
|
37 ℃ 4 hour
Gel electrophoresis:
Products of double digest of LG, LS1 and LS2;
marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
voltage and time: 60V 5min; 120V 15min
lane1: LG;
lane3,4: LS1, LS2;
lane2: marker;
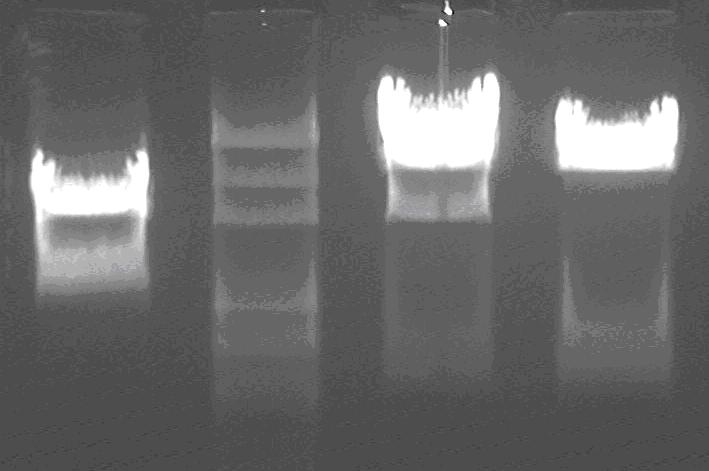
The insert of LG is 1.1k, correct;
The insert of LS is 600bp, correct;
DNA Gel purification:
Insert of LG, LS1 and LS2.
DNA ligation:
| System | 10uL
|
| Insert | 6uL
|
| vector | 2uL
|
| buffer | 1uL
|
| T4 DNA ligase | 1uL
|
16℃ 4 hour
Insert: LG, LS1 and LS2;
Vertor: pSB4K5
Transformation:
Products of ligation, competent cells 50uL each,
Smear to LB plate with Kan.
2009.8.22
PCR: (colony PCR)
| Master mix | 5ul
|
| primer (standard primer) | 0.5uL each
|
| water | 4uL
|
| template |
|
Gel electrophoresis:
Products of PCR
Marker: 100bp 250bp 500bp 750bp 1kb 2kb 3kb 5kb
loading buffer and DNA dye: 6×
Voltage and time: 60V 5min; 120V 15min
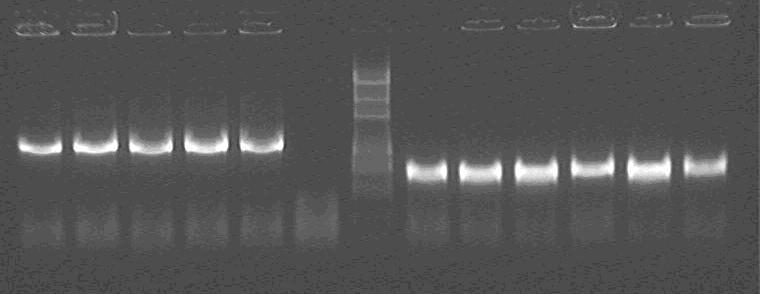
Lane1~5: LG1~5;
Lane6: PCR negative control;
Lane7: Marker
Lane8~13: LS1~6
All colonies are correct!!!
Result
I successfully transferred lacP-SupD-term (K228822) and lacP-GFP (K228821) to low copy plasmid pSB4K5.
^Top
|