Notebook > AND Gate 1 > Input > IPTG Sensor
2009.7.30
Measure the concentrations of Ligation products of reverse lacI + promoter
| Sample | Concentration(×50µg/mL)
|
| 1 | 1.6536
|
| 2 | 2.4225
|
| 3 | 2.7404
|
| 4 | 2.8690
|
| 5 | 2.4858
|
| 6 | 2.5618
|
Enzyme digestion to test the ligation product
| Plasmid | 8µl
|
| EcoRI | 1.5 µl
|
| PstI | 1.5µl
|
| 10×H Buffer | 2µl
|
| ddH2O | 7µl
|
| ∑ | 20µl
|
The result is wrong, so MiniPrep plasmids containing reverse lacI or promoter respectively and measure its concentration
| Sample | Concentration(×50µg/mL)
|
| Promoter | 2.5671
|
| Reverse lacI | 3.9938
|
Digest the above plasmids with EcoRI and XbaI or SpeI , 37 centigrade overnight
| Plasmid(promoter) | 10µl | Plasmid(reverse lacI) | 10µl
|
| EcoRI | 1.5 µl | EcoRI | 1.5 µl
|
| XbaI | 1.5µl | SpeI | 1.5µl
|
| 10×M Buffer | 2µl | 10×H Buffer | 2µl
|
| ddH2O | 5µl | ddH2O | 5µl
|
| ∑ | 20µl | ∑ | 20µl
|
2009.7.31
Using CIAP to recycle the vector
| CIAP | 0.5µl
|
| Buffer | 2.5 µl
|
| Digestion product | 20µl
|
| ddH2O | 2µl
|
| ∑ | 25µl
|
37 centigrade, incubate for 30 min
Recycle the vector and fragment
Ligation of reverse lacI fragment into vector containing the promoter
| Buffer | 1µl
|
| Ligase | 1 µl
|
| Vector(promoter) | 2µl
|
| Fragment(reverse lacI) | 6µl
|
| ∑ | 10 µl
|
Transform ligation product into competent cells
Procedure:
| Ice bath | 30 min
|
| Heat shock | 90 s
|
| Ice bath | 2-3 min
|
| Add 100 µl non-resistant LB |
|
| Shaker, 37 centigrade | 45 min
|
| Plate on Amp plate |
|
2009.8.1
9:00
Pick up the colony and shake in the incubator
2009.8.2
11:00
Pick up 21 colonies and identify the results by PCR
It turns out that all the plasmids are self-ligation of the vector
21:00
ligate reverse lacI insert with lacI promoter again
2009.8.3
01:00
transformaiton
14:01
do the ligation again using the vector provided by WSK
20:00
transform the ligation product into DH5a
2009.8.4
Colony PCR
Lane1-39: my sample
Other lanes: WSK’s sample
The number of the right-sized insert: 1,2,3,5,11,12,17,18,19,21,22,23,25,26,27,31,32,33,35,37
22:25
pick up the colonies 11,12,17,21,25,31,35 and shake in the 37 centigrade incubator
Since the vector used for ligation this time is enzyme digested overnight without addition of BSA by WSK, the PCR results finally has the right insert band, about 1.5 kb.
2009.8.5
Miniprep plasmid with No 11,12,17,21,25,31 and measure their concentration
| Sample | Concentration(×50µg/mL)
|
| 11 | 3.7421
|
| 12 | 3.4852
|
| 17 | 1.9614
|
| 21 | 2.8062
|
| 25 | 5.3108
|
| 31 | 3.4985
|
Double digestion the plasmid
| Plasmid | 5µl
|
| EcoRI | 1.5 µl
|
| PstI | 1.5µl
|
| 10×H Buffer | 2µl
|
| ddH2O | 10µl
|
| ∑ | 20µl
|
12:30
bath in 37 centigrade water
2009.8.6
Add SpeI and continue digestion
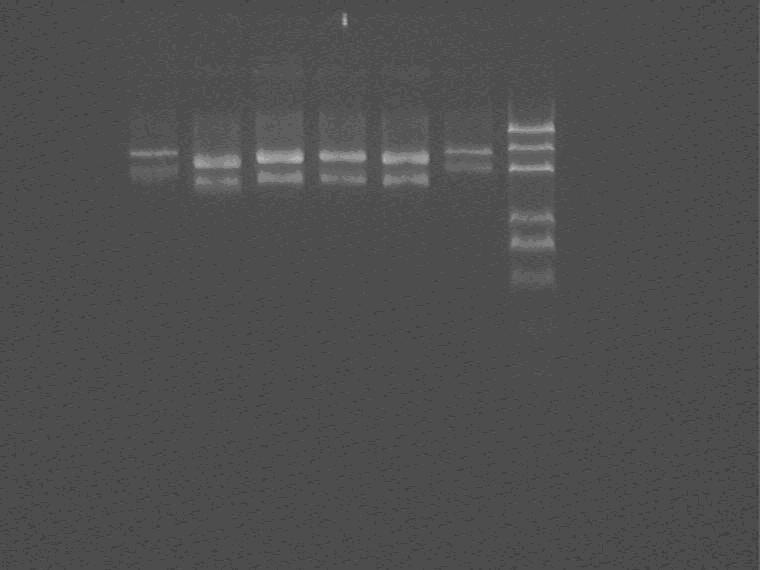
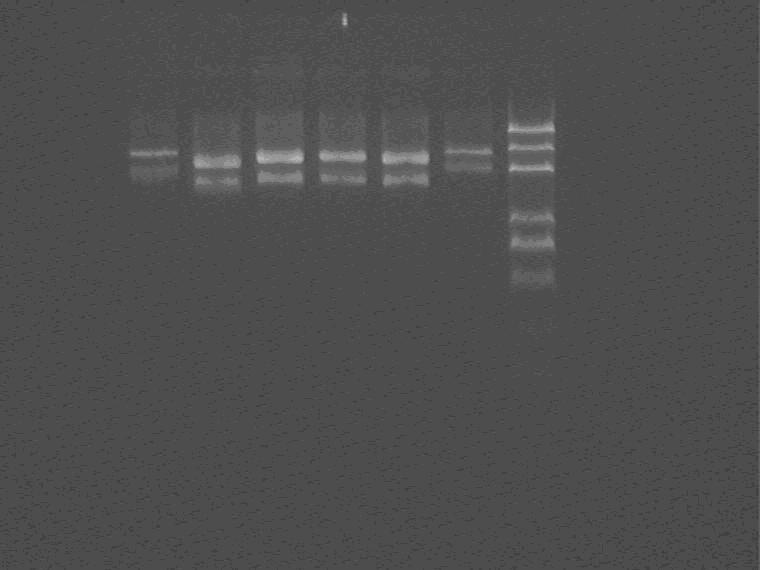
2009.8.7
Electrophoresis and cut the lacI insert band for recycle, measure the recycle product concentration
| Sample | Concentration(×50µg/mL)
|
| 12 | 0.4524
|
| 17 | 0.5731
|
15:30
Ligation of lacI+promoter (insert) with GFP or supD
2009.8.8
18:00
colony PCR: 12lacI-supD, 17lacI-supD
| 2×superMix | 5µl
|
| Universal Primer (Formard) | 0.25 µl
|
| Universal Primer (Reverse) | 0.25µl
|
| ddH2O | 4.5µl
|
| ∑ | 10µl
|
21:30
Electrophoresis to check results
23:00
ligate the insert (reverse lacI+promoter) with vector (containing supD)
2009.8.9
The plate 12lacI-GFP has one colony, colony PCR to check the result
Miniprep 12-2supD and 12-5supD
| Sample | Concentration(×50µg/mL)
|
| 2 | 1.3526
|
| 5 | 2.9598
|
| Plasmid 12-2supD | 6µl
|
| EcoRI | 1.5 µl
|
| PstI | 1.5µl
|
| 10×H Buffer | 2µl
|
| ddH2O | 9µl
|
| ∑ | 20µl
|
2009.8.10
Enzyme digest plasmid 5 to get the insert: reverse lacI+promoter
Digestion confirmation
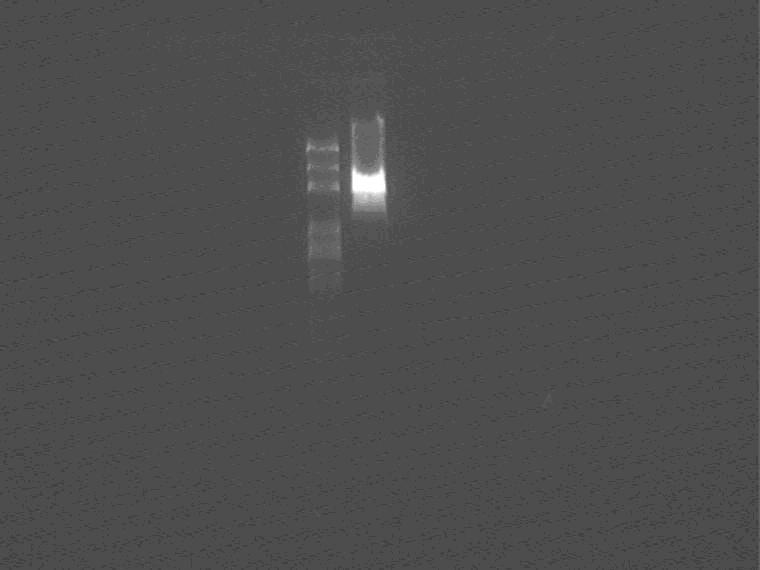
As shown in the figure, there are two bands, vector and insert respectively, it should be right
Recycle the insert in the gel and ligate it with GFP vector, then transform into DH5a
2009.8.11
Pick up nine colonies from the plate and PCR to check the results
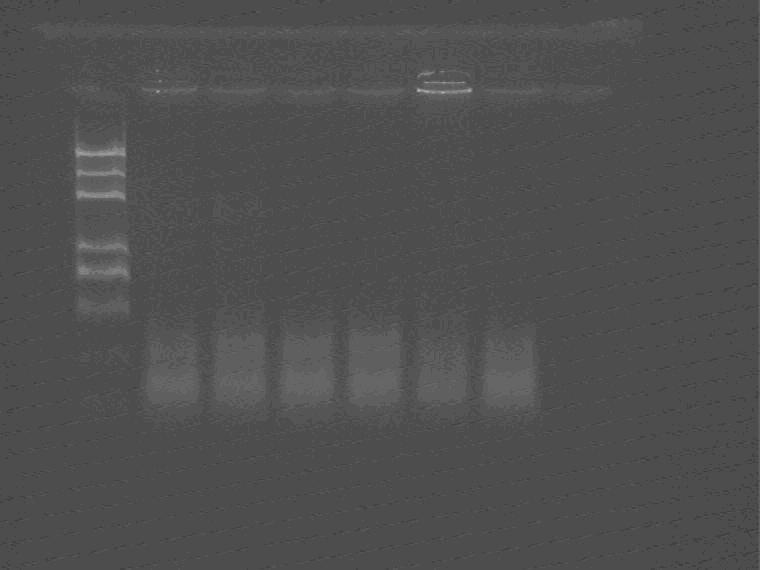
As shown in the figure, 5.1-5.6 are all achromatic, but electrophoresis results show that they are self-ligation.
Shake colonies 5.7-5.9 which are green and shake them in the incubator, then MiniPrep plamid after 10 hours.
| Sample | Concentration(×50µg/mL)
|
| 5.7 | 1.4882
|
| 5.8 | 1.9782
|
| 5.9 | 1.5369
|
The results indicate that lacI with degradation tag LVA may not inhibit the expression of GFP
Use synthesized primer and universal reverse primer to PCR tetR-GFP plasmid, electrophoresis and cut gel to recycle the insert.
2009.8.12
Double enzyme digestion 5.7~5.9 plasmid, electrophoresis. It seems that only 5.8 insert is right
Use delete LVA primer (forward primer as control) to PCR plasmid 5, 5.7, 5.8.
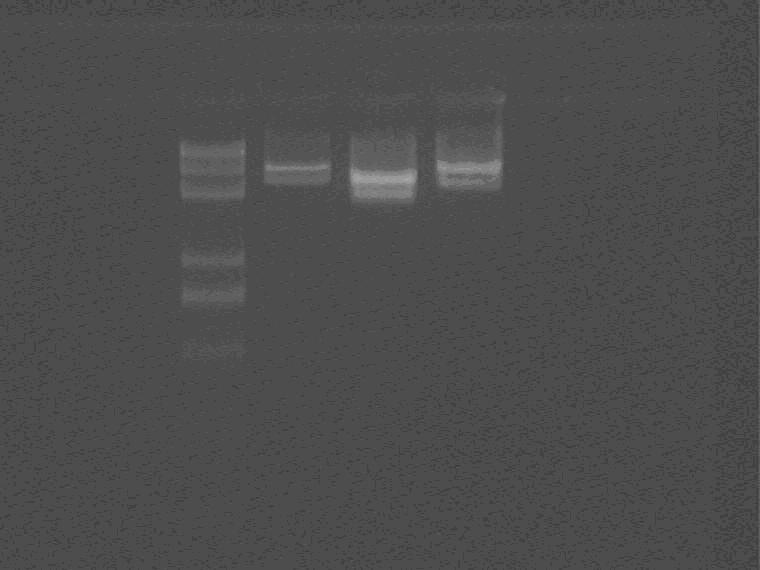
As shown in the figure, to my surprise, 5F also shows positive result. Gel recycle of 5R. Since elongation time is short for 5.7 and 5.8, both results cannot be judged.
2009.8.13
Use dLVA primer to PCR plamid 5(no GFP), enzyme digestion and gel recycle the lacI insert, then ligate it with GFP vector.
Use dLVA primer to PCR plasmid 5.7 and 5.7(contain GFP), gel recycle to get the insert.
2009.8.16
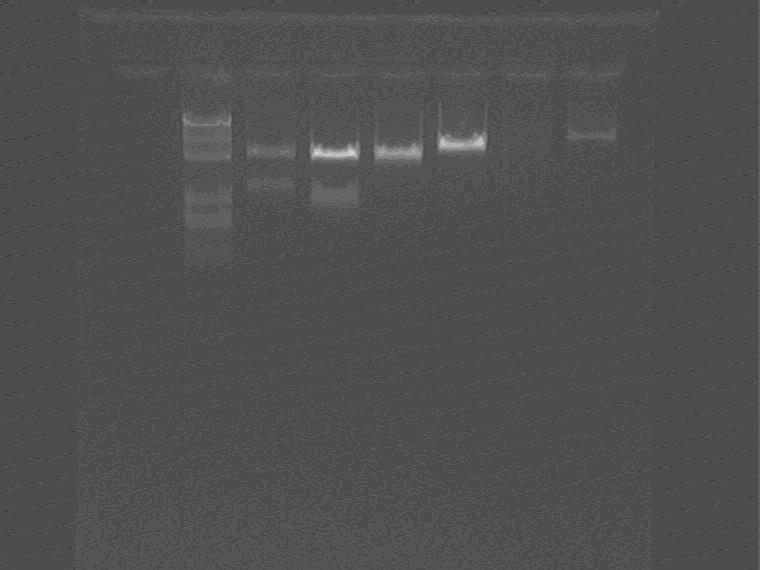
Double enzyme digestion: lacI+promoter+GFP delete LVA plasmid
^Top
|