Parts > Parts Characterization > BBa_K228004 > Transfer Function
BBa K228004 - NahR( reverse) - salicylate promoter
Designed by Lin Min Group: iGEM09_PKU_Beijing (2009-09-18)
Input: Salicylate molecules
Output: GFP fluorescence
Description
The transfer function describes the equilibrium relationship between input (salicylate solution at a gradient of concentration) and output (GFP fluorescence) signals.
For the purpose of characterization, we connected [http://partsregistry.org/Part:BBa_E0840 BBa_E0840] downstream of the salicylate promoter to make a composite part, [http://partsregistry.org/Part:BBa_K228850 BBa_K228850]. This report system allows us to use Microplate Reader to test the salicylate inducible promoter indirectly via the fluorescence output of [http://partsregistry.org/Part:BBa_E0840 BBa_E0840], while the input is salicylate solution at a gradient of concentration.
Data
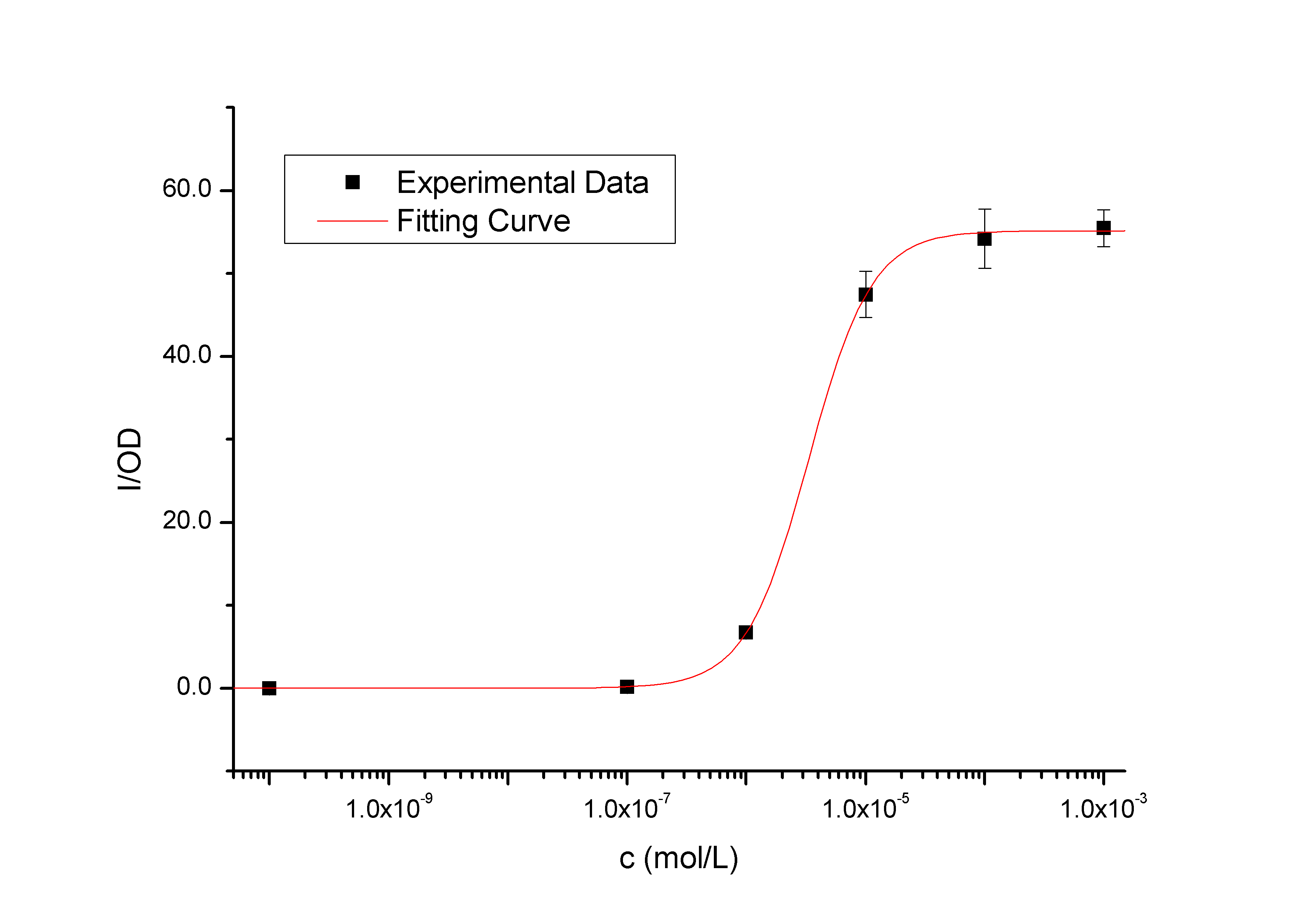 Figure 1 - Transfer function of BBa_K228004. This transfer function is the 120 min time-slice from the time and dose dependent input-output surface. The data points represent the mean of 6 individual measurements. The corresponding error bars represent the 95% confidence interval in the mean of the independent measurements. The solid black curve was calculated by fitting a simple Hill function to the experimental measurements. The Y axis denotes the value of fluorescence normalized by the OD600 value, and the X axis denotes the concentration of the inducer. 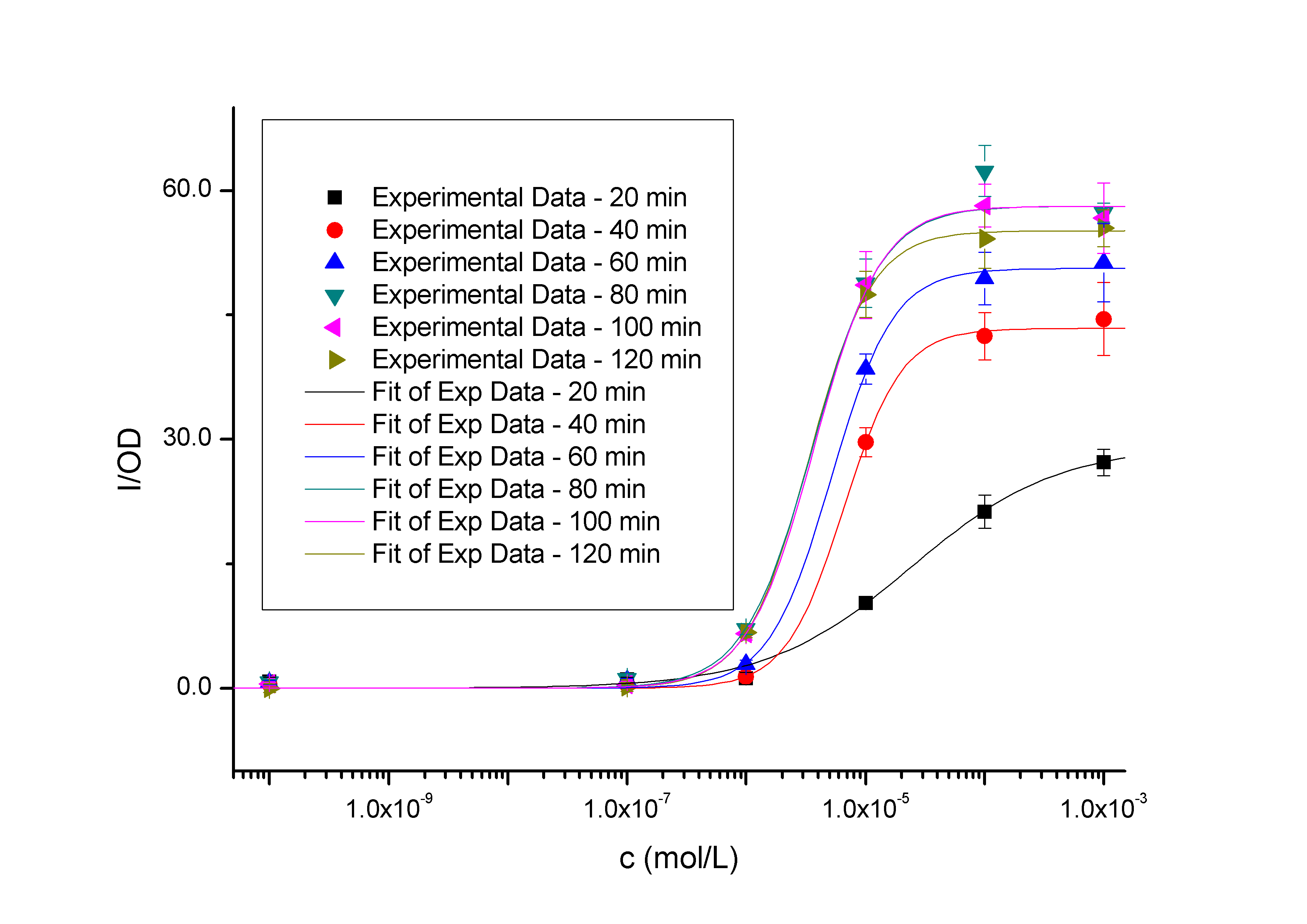 Figure 2 – Transfer functions of BBa_K228004 in different time slices from the time and dose dependent input-output surface. The data points represent the mean of 6 individual measurements. The corresponding error bars denoting the 95% confidence interval in the mean of the independent measurements. These solid curves were calculated by fitting simple Hill functions to the experimental measurements. The Y axis denotes the value of fluorescence normalized by the OD600 value, and the X axis denotes the concentration of the inducer. As we can see, the level of induction triggered by the same concentration of inducer (higher than 10^-7 mol/L, causing concentration of 10^-10 mol/L has no significantly positive differences with the blank) will be enhanced as the induction time being extended, so it is the same with the out-put saturation. Protocol
- A subset of inducer solution with a concentration gradient of 10^-7, 10^-5, 10^-3, 10^-2;
- Bacterial colonies;
- Phosphate Buffered Solution (PBS).
1. One 40 ml culture of LB medium and antibiotic (Ampicillin, 100ng/ml) was inoculated with a single colony from a LB agar plate containing a [http://partsregistry.org/Part:BBa_K228850 BBa_K228850] (the constructed report system described in the above).
2. Cultures were grown in 1.5 ml test tubes for several hrs at 37°C with shaking at 70 rpm to an OD600 value of 0.530. This growth took on average 4 hrs.
3. Add 1.5 mL of the fresh bacteria culture to 42 new test tubes. Then pipet an appropriate volume of salicylate solution into test tubes to yield 7 different final concentrations (0, 1.0*10^-10, 1.0*10^-7, 1.0*10^-6, 1.0*10^-5, 1.0*10^-4, 1.0*10^-3). Thus 6 replicates were measured for each concentration of salicylate. The concentration 0 group is to measure the fluorescent background.
4. Place the induction system at 37 degree. Every 20 minites, pipet 200 uL of the culture from each 1.5 ml test tube into a new 1.5 ml test tube, respectively, till the incubating time reaches 2 hrs.
5. Pellet bacteria cells by 1min centrifugation at 13000 rpm, and discard the supernatant as possible as we could. Resuspend the pelleted cells in 200ul of PBS. Time between repeated operations was about 20s.
6. Transfer 100 uL of bacteria l resuspention into each well of 96-well plate to test the GFP fluorescence by Microplate Reader.
7. Use spectrophotometer to test the OD600 value of the left 100 ul of bacteria resuspention.
8. We converted the OD600 value into the concentration of cells (/ml). The equation is OD600 1.0=5*10^8 cells/ml. Then we normalized the GFP fluorescence by the OD600 values, to obtain the ratio of GFP fluorescence to the OD600 value. The mean for each concentration-time group was then averaged across 6 replicates to obtain a population mean.
9. The transfer function in Figure 1 is the 120 min time-slice from the time and dose dependent input-output surface. The R^2 is 0.99999. The data points represent the mean of 6 individual measurements. The corresponding error bars denote the 95% confidence interval in the mean of the independent measurements.
10. In Figure 2, the level of induction triggered by the same concentration of inducer (of which concentrations are higher than 10^-7 mol/L, causing concentration of 10^-10 mol/L group has no significantly positive differences with the blank group) will be enhanced as the induction time being extended, so it is the same with the saturation of fluorescence, as expected.
11. To estimate parameters that characterize the measured transfer functions, we used least squares estimation to fit a simple model to the data. Hill equations derived from simple biochemical equations describe the data well (Table 1). Pmax is the maximum output level, K is the switch point, and n is the hill coefficient describing the steepness of the transition from low output to high output.
Table 1. Parameters of the measured transfer functions
| | Pmax | K | n | R^2
|
| 20 min | 29.2±3.6 | 2.42E-05±1.07E-05 | 0.710±0.144 | 0.98124
|
| 40 min | 43.4±4.4 | 6.54E-06±1.48E-06 | 1.792±0.275 | 0.97309
|
| 60 min | 50.6±6.6 | 5.13E-06±1.71E-06 | 1.677±0.335 | 0.96184
|
| 80 min | 58.1±2.5 | 3.59E-06±1.16E-06 | 1.520±0.373 | 0.99088
|
| 100 min | 58.1±3.2 | 3.70E-06±1.05E-06 | 1.567±0.316 | 0.98898
|
| 120 min | 55.2±0.3 | 3.32E-06±8.29E-08 | 1.649±0.032 | 0.99999
|
^Top
|