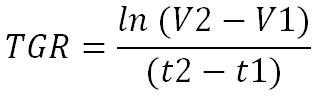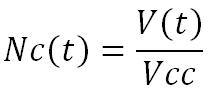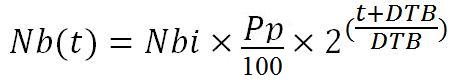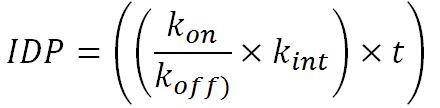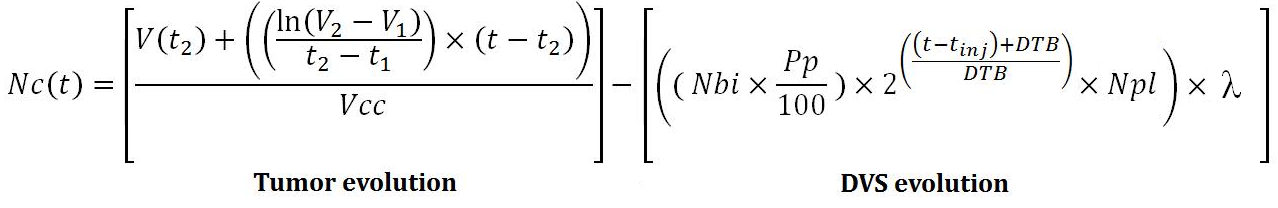Team:SupBiotech-Paris/Treatement modeling
From 2009.igem.org
Contents |
Modelling of DVS efficiency on a lung tumour
Context
Non-small cell lung carcinoma, or NSCLC, is an aggressive cancer, with a relatively high speed growth. Treatments are often ineficient, because the tumor growth is faster than the elimination by the drug.
Objective
We have decided to model our treatment efficacy for this kind of tumor. Therefore we have modelled the tumour progression, our treatment evolution and efficacy.
The objective of the modelling is to verify if our treatment is able to eliminate the entire tumor.
Model segmentation
First, we had to recreate DVS complete mechanism and the tumor evolution. Then, for each step of the treatment, we have identified all the parameters that intervene, their actions and their interactions, in order to determine the model equations.
To simplifly the equation we have devided the mechanism and we have modelled each step separately.
Tumor and DVS evolution versus time
First step : Tumor development according to time
We consider the tumor is non metastatic and its growth is exponential.
Let the tumor have a volume V1 in cm3 at an instant t1.
Let the same tumor, at an instant t2, have a volume V2.
The tumor is considered in exponential growth phase and without metastasis therefore its development equation, Tumor Growth Rate (TGR), is equal to :
Thus, the tumor volume according to the time (V(t)) is equal to :
Finally, knowing the Average volume of a cancerous cell (Vcc) (experimental data), if we regard the tumor as fraught (without cavity or blood vessel), we can determine that the Number of cancerous cells according to time (Nc(t)), without treatment effect, is equal to :
Second step : Development of the tissue vector
The tissue vector is injected to the patient at an instant t, near t2. The Number of injected vectors (Nbi) is 1x10^6. The pulmonary tropism of the vector isn’t perfect, only a Percentage (Pp) goes to the lung. The total number of tissue vector in the body increases, because this vector is bacterial and therefore possesses a Doubling period (DTB).
Thus we can establish that the tissue vector number in the lungs (Nb(t)) is equal to:
The number of Tissue vectors increases until injection of doxycycline. F rom then, tissue vectors lysis releases the cell vectors in the lung.
This injection time is not insignificant. Indeed, si if we wait long enough, tissue vectors number is sufficient to eliminate the tumor or at least to significantly reduce it. On the other hand, if we wait too long, a higher dose of doxycycline (and so potentially toxic) is necessary for cell vector release.
Thus we can use modelling to determine the optimal injection time of doxycyline (Tdox).
Third step : Release of the cell vector
Once the doxycycline injected, the cell vector is released. The cell vectors number is proportional to the tissue vectors number in the lung. And yet, we know the average value of recombinant phage vectors released by M. avium (Npl) is equal to 100.
We can write cell vectors number at the injection instant (Np(Tdox)) is equal to :
The cell vectors number does not increase such as the tissue vectors. Indeed, it decreases with time, because of the phage vector stability and of its cell penetration (to release the therapeutic plasmide).
Its stability in the blood is equal to the phage vector deterioration constant (kdeg) according to time. If we add this constant to the cell vectors number equation according to time (Np(t)) we obtain the following formula :
The phage vector dispersion steps in the tumor and for cell penetration are the steps below Fourth and Fifth) because of their complexity.
DVS Efficiency
So, we determined:
- The size of the tumor versus time (initial volume + growth)
- The amount of tissue vector versus time
- The amount of released cell vectors for a tissue vector
Now, we're going to determine the efficiency of our vectors for penetring cancer cells.
For that we are studying:
- The area of dispersal cell vector
- The importance of the cellular internalization of the vector in cancer cells.
Fourth step : The cell vector dispersion
Here, we are looking for determine the maximum area that a phage can cover. This requires knowing:
- The spread of phages in the bloodstream
- Their diffusion through the walls of blood vessels
- The surface of a cancer cell
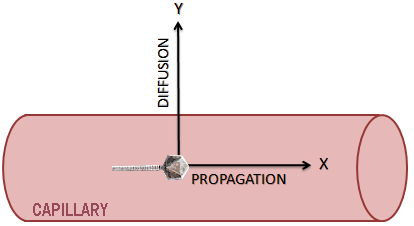
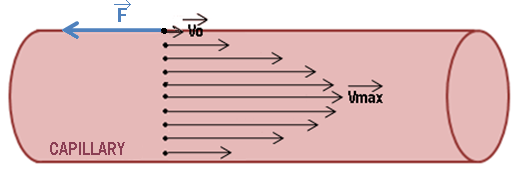
For our modeling, we consider the blood as a Newtonian fluid with a constant velocity Vmax. Here, We neglect the heart-related jolts and turbulent flows caused by the cavities of the blood epithelium.
The cell vector moves along two axes. An X axis in the direction of blood flow and a Y axis orthogonal to the axis X.
The phage propagation in the bloodstream
The movement in X depends solely on the propagation of phages the vessel due to blood flow. Indeed, we neglect the diffusion which takes place also along the X axis because it is 1000 times less than the propagation of particles in the blood (due to the importance of blood flow). The cellular carriers are moving at speeds spread on a dish from, Vmax in the center of the vessel at V0 against the vessel wall.
The speed of phages decrease in approaching the vessel walls due to the friction forces which are opposing to the movement.
We can determine how long the particle (with a Vmax velocity), ie the particles in the center of the ship, reached the end. This gives the time necessary to internalize all the phages of the bacteria.
The diffusion through the walls of blood vessels
The movement in Y is the distribution of phages in the blood (j(n)). It depends on the equation of diffusion of a particle (n) in a fluid (Fick's Law).
With n the number of particles (phages), grad n the difference between the concentrations and D the diffusion coefficient. The cellular distribution of vectors within the blood vessel and then through the wall is a phenomenon of diffusion with output. So, there will always be a strong gradient of concentration of phage in the blood. We can therefore say that the gradient is constant (equal to 1) over time. Thus the diffusion rate (j(n)) is equal to D.
Dispersal area of phage
When we combine moving Y (diffusion rate) and moving in X (blood flow velocity), we obtain, after integration on the perimeter of a blood vessel, the action surface of cell vectors. Then, we are able to determine the number of cancer cells per 100 cell vectors destroyed or 1 tissue vector.
The diffusion rate of the cell vector is equal to 0.5 μm.s-1 and the size of a capillary blood is 10μm in diameter. The particle farthest places so 10s to reach the vessel wall.
With this dissemination length (10s), the blood flow velocity (1x10 ^ 3μm.s-1) in the capillaries, and the surface of one cancer cell (1 micron square), we can determine:
- The length (L) covered by the cell vectors released by one tissue vector.
- The surface (S) occupied by phages in blood vessel diameter of 2r.
- The amount of cancer cells available.
L = 1 x 10^4 µm 2r = 10 µm S = 2 x π x L r = 31.4 x 10^4 µm²
Thus, a tissue vector can potentially target more than 31 000 cancer cells, yet it has that 100 cell vectors. We can make a simplification to say that 100 cell vectors destroy 100 cancer cells. The efficiency of the dispersion is complete.
For phage, once reached the wall, comes in the cellular internalization. This model responds to two courses of action.
Step Five: The cell vector internalization
Once in contact with the cell, the cell vector has two possible ways of action.
- The vector fix then it detaches from the cell.
- The vector fix then it is internalized within the cell.
We can model this according time and the association constant (kon) and dissociation constant (koff) and internalization constant(Kint).
This gives:
With kon = 5.10^3 M-1s-1, koff = 8.10^-3 s-1 and Kint = 5,78.10^-4 s-1. If we calculate the global constant K', such as IDP = K' xt, we obtain K'= 361.5 s-1. Thus more than 360 phages are internalized per second in contact with a wall.
If we compare the time required to internalize a phage compared to waiting times before the cell enters apoptosis in response to the entrance of a vector cell (1 hour), it appears logical to neglect the internalization of cell vectors (IDP = constant = 360 phages/s) in the final equation.
Thus, with a total efficiency of the phage diffusion, and a neglected internalization time, we can say that the efficiency constant λ is equal to 1.
Simultaneous evolution of DVS and tumor
The evolution equation of our model over time is equal to:
With :
- Nc (t), the number of cancer cells depending time,
- V (t), tumor volume,
- V1 and V2, two tumors volumes respectively times t1 and t2,
- Vcc, the volume of a cancer cell,
- Nbi, the number of injected tissue vectors,
- Pp, the lung percentage of tissue vectors relative to the injected dose,
- DTB, the doubling time of tissue vector,
- tinj, injection time of the [[Team:SupBiotech-Paris/Concept1#drapeau|tissue vectors],
- Npl, the number of cell vectors released by bacteria.
We can neglected (differences between the time or space scales) some factors:
- Kdeg, the degradation constant of the phage, because all the phages were internalized in 10s.
Treatment simulation
The DVS needs two injections:
- The DVS injection,
- The activator injection: the doxycycline.
You can find just below a simulator, which calculates how many time after the DVS injection, we have to wait until the doxycycline injection.
 "
"