Team:Newcastle/Metals
From 2009.igem.org
(→Metal Sequester) |
(→Lab Work Done) |
||
| Line 110: | Line 110: | ||
|'''[https://2009.igem.org/Team:Newcastle/Labwork/17_September_2009#Metal_Sensor_Team 17th September 2009]''' | |'''[https://2009.igem.org/Team:Newcastle/Labwork/17_September_2009#Metal_Sensor_Team 17th September 2009]''' | ||
| Midi-prepped potential ''cotC-GFP-smtA'' transformant ''E. coli'' culture and digested with ''BamHI'' and ''HindIII'' | | Midi-prepped potential ''cotC-GFP-smtA'' transformant ''E. coli'' culture and digested with ''BamHI'' and ''HindIII'' | ||
| + | |- | ||
| + | |'''[https://2009.igem.org/Team:Newcastle/Labwork/24_September_2009#Metal_Sensor_Team 24th September 2009]''' | ||
| + | | | ||
|- | |- | ||
|'''[https://2009.igem.org/Team:Newcastle/Labwork/18_September_2009#Metal_Sensor_Team 18th September 2009]''' | |'''[https://2009.igem.org/Team:Newcastle/Labwork/18_September_2009#Metal_Sensor_Team 18th September 2009]''' | ||
Revision as of 03:53, 22 October 2009
Metal Sequester
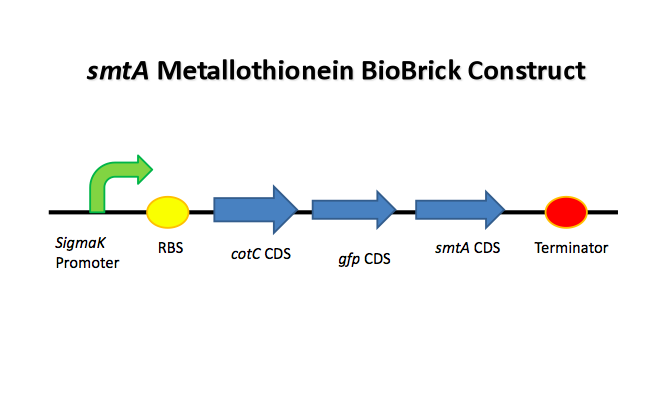
In our system, the team have used the metallothionein SmtA (which usually mops up zinc ions) and have linked it to the CotC spore coat protein - this means that, should the stochastic switch decide, any cadmium mopped up could be sequestered into spores thereby making it unavailable. Please click on the following links to learn more:
- Introduction
- Novelty in this sub-project
- BioBrick constructs
- Lab Work Strategies
- Lab Work Done
- References
Introduction
In order to take up and keep the cadmium from the soil efficiently, we needed to think about a way to cross-link the metal ions to intra-cellular proteins which would end up for the most of it in the spore. If the heavy metal ions were not cross linked to a "sponge protein", it could be lethal to the cell at lower concentration and the cell could end up sporulating too early, or even bursting in the soil, releasing all the heavy metal it has taken up.
From our meeting with Prof. Nigel Robinson on the 18th of March 2009, it was evident that the best way of getting cadmium into the spore is to express a metallothionein, which would 'soak up' the cadmium, which in turn would be trapped within the protein in the spore.
Novelty in this sub-project
To increase the efficiency of our system in sequestering the heavy metal cadmium from the soil, we divised a plan to make sure that most of the metal ions that have been taken in our B.subtilis cell is then rendered bio-unavailable by incorporating it into smtA metallothionein.
SmtA metallothionein protein from E. coli can bind to heavy metals [1,2,3]. They have a tendency to bind to cationic metal ions such as cadmium, copper, arsenic, mercury, silver.
It has been shown that in B. subtilis, in order to express a specific protein in the spore coat, it is possible to make a fusion protein with a spore coat protein called CotC, and a group have successfully expressed antigen proteins which are about the same size as our smtA metallothionein in order to make a vaccine[4]. By fusing CotC spore coat protein from Bacillus subtilis, our smtA metallothionein can be localized to the spore coat, hence we hoped to successfully trap most of the metals ions into the bacterial spores.
CotC was also fused with GFP reporter gene in order to detect the expression of the fusion protein into the spores using a fluorescence microscopy.
Because we want most of the metals to go into the spore rather than the vegetative cell, we designed our fusion protein so that it is controlled by the native cotC promoter sigK which is activated in sporulation conditions. Therefore, our smtA metal sequester would only be expressed once the cell have made the decision to become a metal container and sporulate, and only be expressed in the spore.
Finally, our construct integrated the B. subtilis genome through homologous recombination at the native CotC gene. However, as we will be using pMutin4 integration vector, there will be two different copies of the cotC gene in our engineered B.subtilis genome: a native copy of cotC and our engineered CotC-GFP-smtA fusion copy. We chose not to knock-out the native CotC gene because the lack of native CotC might lead to defects in spore coat formation.
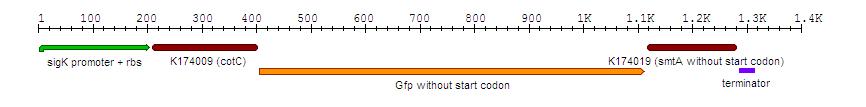
BioBrick constructs
In the parts registry, BBa_K174008:
Lab Work Strategies
a) Construction
Synthesised by GeneArt, fragment: 1385 bp DNA
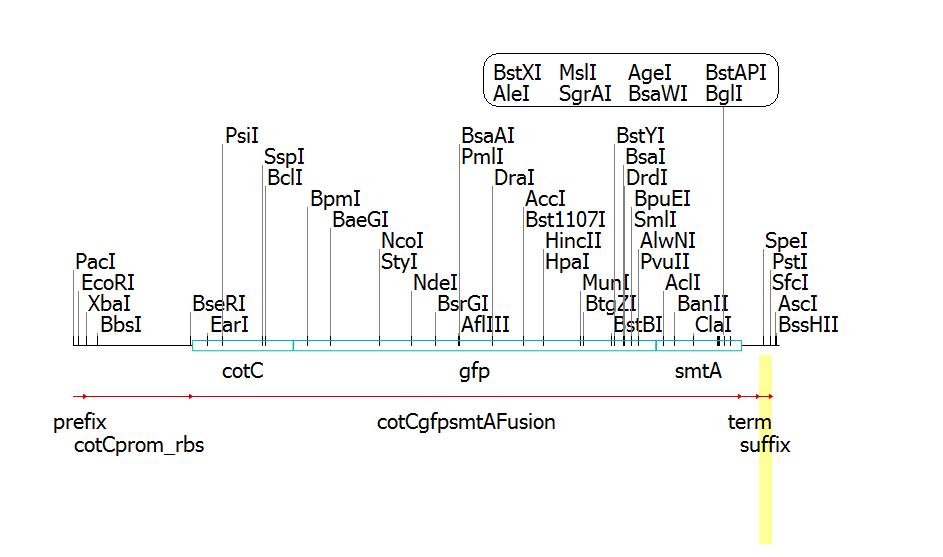
Clone Manager construct and cut map:
Sequencher construct:
b) Cloning and Integration
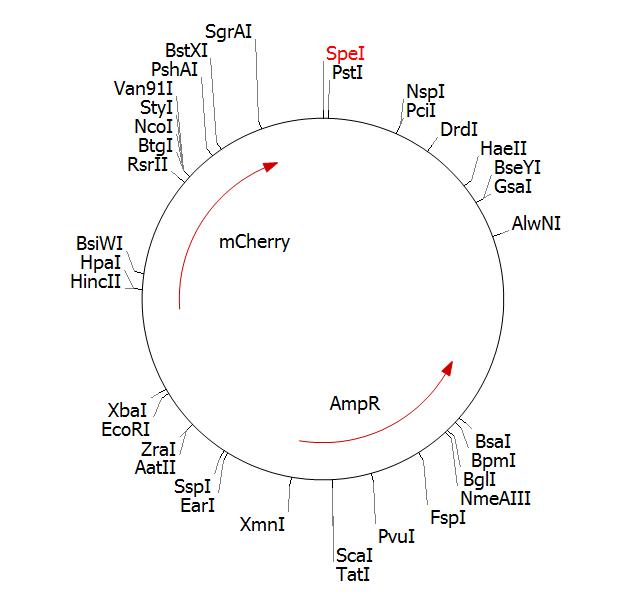
GeneArt cloned the vector into a standard biobrick vector. We sent them Plasmid pSB1AT3 with part BBa_J04450 (mCherry).
BBa_J04450 in pSB1AT3 Clone Manager plamid map:
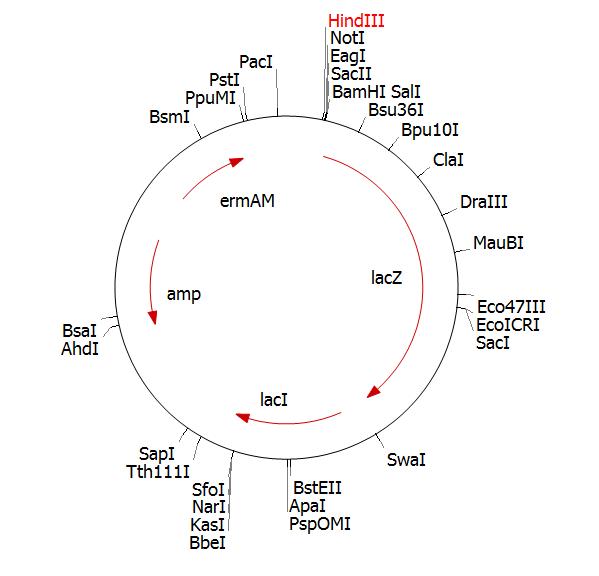
Once we received this fragment cloned in pSBAT3 we amplified the part by PCR. Primer 1 will incorporate a HinDIII site and primer 2 will incorporate a BamHI. Checked that these enzymes don’t cut the fragment:
We cut the PCR fragment and clone into pMutin4 cut with the same enzymes (diagram below is pMutin2 but this is essentially the same)
We then integrate into the 168 chromosome using homology between our cotC fusion and the native copy of cotC.
c) Testing and Characterisation
Once transformed the spores of the mutant will need to be tested for fluorescence. Two obvious methods:
1) Fluorescence microscopy: grow the cells in sporulation medium and look under the microscope for fluorescent spores.
2) Purify spores and measure their fluorescence in a fluorescence plate reader.
3) We would also test whether the strain is able to absorb cadmium in the spores when compared to the wild-type.
Lab Work Done
| Summary of Lab Sessions for Metal Sequestering | |
|---|---|
| | |
| 9th September 2009 | Arrival of cotC-GFP-smtA BioBrick - transformed this into DH5-alpha E. coli cells |
| 10th September 2009 | Used colonies of cotC-GFP-smtA transformants to inoculate LB media in preparation for mini and midi-preps |
| 11th September 2009 | Mini and midi-prepped cotC BioBrick and PCR amplified cotC BioBrick, giving it restriction ends; |
| 14th September 2009 | Ligated cotC into pMUTIN4 plasmid and attempted E. coli transformation |
| 15th September 2009 | Transformations seemed to have work; picked 12 colonies from 200ul plate and inoculated 12 tubes of 5ml LB + ampicillin |
| 16th September 2009 | Mini-preps of 12 picked colonies and restriction enzyme digest analysis - gel showed consistent bands so prepared midi-prep cultures |
| 17th September 2009 | Midi-prepped potential cotC-GFP-smtA transformant E. coli culture and digested with BamHI and HindIII |
| 24th September 2009 | |
| 18th September 2009 | Analysed digested midi-prep sample through DNA gel electrophoresis - negative results |
| 28th September 2009 | Picked 10 colonies of potential cotC in pMUTIN4 E. coli transformants and inoculated 10 tubes of LB + amp with them |
| 29th September 2009 | Conducted mini-preps of 10 cultures of potential transformants and analysed them through restriction digests and gel electrophoresis - did not work |
| 1st October 2009 | Checked pMUTIN4 and cotC on gel; carried out another ligation |
| 2nd October 2009 | Used ligation products to transform more E. coli cells |
| 5th October 2009 | Attempted transformations failed |
| 6th October 2009 | Conducted pMUTIN4 midi-prep again and transformed E. coli with cotC in pSB1AT3 (newly arrived from GeneArt) |
| 7th October 2009 | Transformations of E. coli with cotC in pSB1AT3 worked - inoculated LB with these colonies for mini and midi-preps. Also PCR amplified the cotC BioBrick with restriction sites. |
| 8th October 2009 | Digested newly midi-prepped pMUTIN4 and also midi-prepped cotC in pSB1AT3 (from GeneArt) |
| 9th October 2009 | Ran digested cotC PCR product (made on 07/10/09) on gel and excised band. |
| 12th October 2009 | Because the cotC-GFP-smtA + pMUTIN4 digests haven't been working, the team plated out some new E. coli + pMUTIN4 cells. |
References
1- Cretì, P., F. Trinchella, et al. "Heavy metal bioaccumulation and metallothionein content in tissues of the sea bream Sparus aurata from three different fish farming systems." Environmental Monitoring and Assessment.
2- Morby, A. P., J. S. Turner, et al. (1993). SmtB is a metal-dependent repressor of the cyanobacterial metallothionein gene smtA: identification of a Zn inhibited DNA-protein complex. 21: 921-925.
3- Waldron, K. J. and N. J. Robinson (2009). "How do bacterial cells ensure that metalloproteins get the correct metal?" Nat Rev Micro 7(1): 25-35.
4- Mauriello, E. M. F., L. H. Duc, et al. (2004). "Display of heterologous antigens on the Bacillus subtilis spore coat using CotC as a fusion partner." Vaccine 22(9-10): 1177-1187.
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"