Team:UNICAMP-Brazil/Notebooks/October 9
From 2009.igem.org
(→pADH1+YFP (new biobrick)) |
(→Cre-Recombinase - Confirmation: Part I) |
||
| (19 intermediate revisions not shown) | |||
| Line 17: | Line 17: | ||
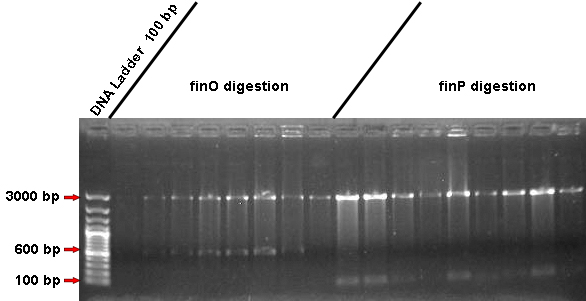
* We decided that we are going to confirm such ligations by performing: | * We decided that we are going to confirm such ligations by performing: | ||
| - | -A Digestion: with | + | -A Digestion: with ''Xba''I and ''Spe''I restriction enzymes, in order to release our parts from pGEM plasmid; |
-A PCR: performed with the specific forward primer for our insert and with the reverse primer for pGEM plasmid (M13), in order to confirm that, once our inserts are indeed in there, they are also in the correct frame position. | -A PCR: performed with the specific forward primer for our insert and with the reverse primer for pGEM plasmid (M13), in order to confirm that, once our inserts are indeed in there, they are also in the correct frame position. | ||
| Line 30: | Line 30: | ||
''Gabriel and Marcelo'' | ''Gabriel and Marcelo'' | ||
| + | ==== PY Promoter - Colony-PCR screening ==== | ||
| + | |||
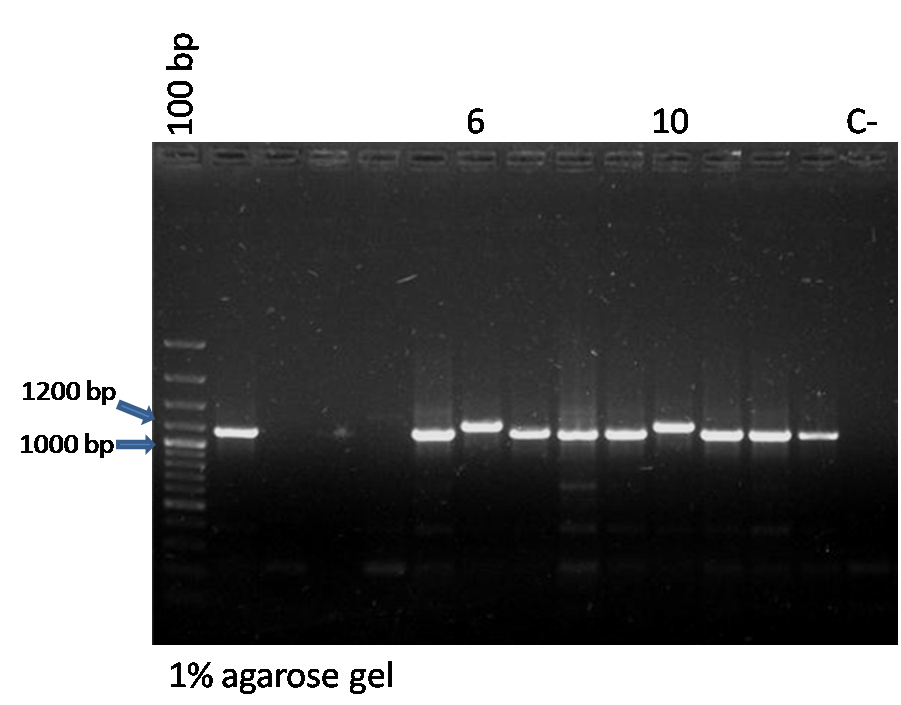
| + | *<p style=”text-align:justify;”>We selected 13 colonies of the transformation we did yesterday to perform a colony-PCR screening. We used 2 pairs of primers and analyzed the results of our PCR in an agarose gel:</p> | ||
| + | <p style=”text-align:justify;”>- VF/VR, the verification primers of BBa_J23100 plasmid:</p> | ||
| + | |||
| + | [[Image:py_gel_4.png|450px|center]] | ||
| + | |||
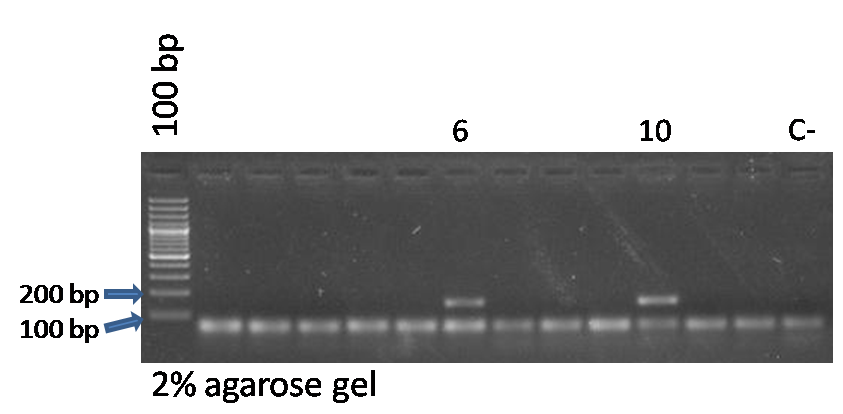
| + | <p style=”text-align:justify;”>- Ppy-F-1 and Ppy-R, the pair of primers that amplify our inserted sequence (PY1):</p> | ||
| + | |||
| + | [[Image:py_gel_5.png|450px|center]] | ||
| + | |||
| + | <p style=”text-align:justify;”>- The expected size for PY1 + BBa_J23100 amplified with VF/VR primers is 1190 bp.</p> | ||
| + | |||
| + | <p style=”text-align:justify;”>- The expected size for BBa_J23100 without our insert amplified with VF/VR primers is 1057 bp.</p> | ||
| + | |||
| + | <p style=”text-align:justify;”>- The expected size for PY1 + BBa_J23100 amplified with PY primers is 133 bp.</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>As we can observe in the gel photo, colonies 6 and 10 presented bands compatible with the size expected for both pairs of primers.</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>So we inoculated these 2 colonies in liquid LB-AMP at 37ºC to perform a mini-prep for plasmid extraction tomorrow.</p> | ||
| + | |||
| + | ''Fabi and Léo'' | ||
| + | |||
| + | ====Cre-Recombinase - Confirmation: Stage I==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>After performing minipreps (on October 5th) for 20 inoculated cultures, possibly harbouring our Cre-Recombinase without ATG's Biobricks, it's time to start the confirmation procudures. Regarding "confirmation procudures", we mean a two stages procedure: (1) perform a specific PCR for Cre-Recombinase and (2) perform a digestion, aiming in excising a fragment of expectable size from the pSB1A3 vector.</p> | ||
| + | *<p style=”text-align:justify;”>Today we did stage I, a PCR reaction using Cre's specific designed primers, running an agarose gel straight after reaction ended.</p> | ||
| + | *<p style=”text-align:justify;”>Thereby, according to Stage I.... we did it! Several samples appears to contain our Cre's Biobrick!</p> | ||
| + | *<p style=”text-align:justify;”>If we succeed in Stage II, we will finally be able to say we got a biobrick! =]</p> | ||
| + | |||
| + | ''Víctor'' | ||
==''' YeastGuard '''== | ==''' YeastGuard '''== | ||
| - | ====New | + | ====[https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/pGEMStrategy New Strategy: pGEM]==== |
| - | *<p style= | + | *<p style="text-align:justify;">The plates containing ''E. coli'' transformed with pDLD didn’t grow. We repeated the ligation reaction and transformed competent ''E. coli'' again.</p> |
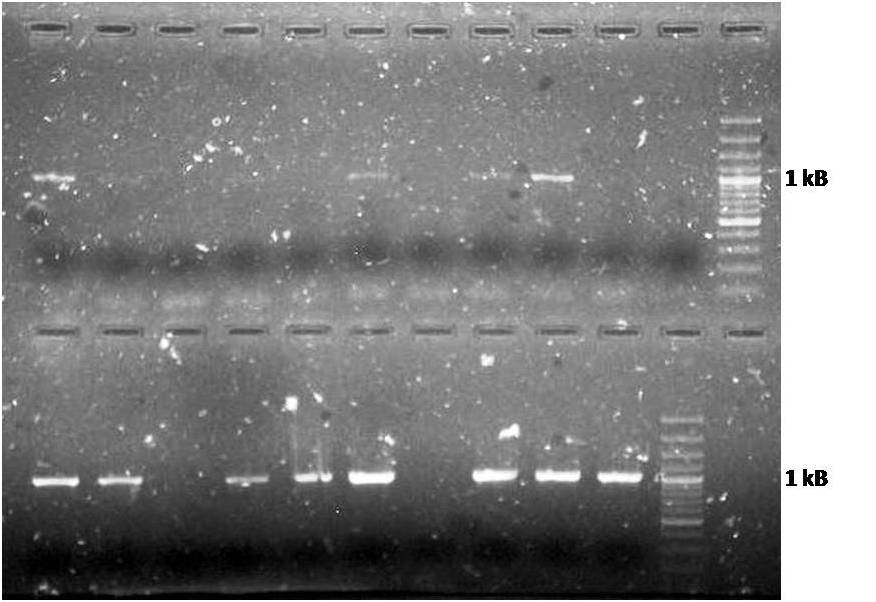
*<p style=”text-align:justify;”>We transformed the ligation between Lysozyme and biofusion, performed yesterday, in competent ''E. coli'' and plated in LB+Amp media.</p> | *<p style=”text-align:justify;”>We transformed the ligation between Lysozyme and biofusion, performed yesterday, in competent ''E. coli'' and plated in LB+Amp media.</p> | ||
*<p style=”text-align:justify;”>The pJEN1 plates showed colonies, we did the colony PCR to find a correct construction of the pJEN1 in Biofusion. We confirmed 12 of 20 colonies.</p> | *<p style=”text-align:justify;”>The pJEN1 plates showed colonies, we did the colony PCR to find a correct construction of the pJEN1 in Biofusion. We confirmed 12 of 20 colonies.</p> | ||
| - | + | [[Image:PJEN1plates.jpg|400px|center]] | |
| Line 45: | Line 76: | ||
====YFP+Terminator==== | ====YFP+Terminator==== | ||
*<p style=”text-align:justify;”>We transformed the ligation between YFP+end and Adh1 promoter, performed yesterday, in competent ''E. coli'' and plated in LB+Amp media.</p> | *<p style=”text-align:justify;”>We transformed the ligation between YFP+end and Adh1 promoter, performed yesterday, in competent ''E. coli'' and plated in LB+Amp media.</p> | ||
| + | |||
| + | * We decided to check over the size of the terminator part at the registry, since we had to much work with it. We submited its sequence to the BLAST algorithm and discovered that this biobrick is the same as YFP one!!! =( We got so desapointed =/ It was the only terminator biobrick to be used in yeasts. | ||
====pADH1+YFP==== | ====pADH1+YFP==== | ||
| Line 50: | Line 83: | ||
*<p style=”text-align:justify;”>First of all, we did these digestions: 1)YFP digestion using the enzymes ''Xba''I and ''Pst''I; 2)pADH1 (biofusion) digestion using the enzymes ''Spe''I and ''Pst''I.</p> | *<p style=”text-align:justify;”>First of all, we did these digestions: 1)YFP digestion using the enzymes ''Xba''I and ''Pst''I; 2)pADH1 (biofusion) digestion using the enzymes ''Spe''I and ''Pst''I.</p> | ||
| - | ''Wesley | + | ''Wesley'' |
| - | ==== | + | ====YEP358==== |
| - | We transformed ''E. coli'' with | + | We transformed ''E. coli'' with YEP358-β galactosidase plasmid ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Electroporation Protocol 3]) |
{{:Team:UNICAMP-Brazil/inc_rodape}} | {{:Team:UNICAMP-Brazil/inc_rodape}} | ||
Latest revision as of 03:20, 22 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"