Team:UNICAMP-Brazil/Notebooks/October 14
From 2009.igem.org
(New page: {{:Team:UNICAMP-Brazil/inc_topo}} {{:Team:UNICAMP-Brazil/inc calendar}} __NOTOC__ ==''' YeastGuard '''== ====New strategy: pGEM==== *<p style=”text-align:justify;”>We did miniprep...) |
(→New Strategy: pGEM) |
||
| (13 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
__NOTOC__ | __NOTOC__ | ||
| + | ==''' ColiGuard '''== | ||
| + | |||
| + | ==== PY Promoter - Preparation of electrocompetent cells of the conjugative strain ==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>Now that we have PY1 promoter + RFP inserted in a plasmid with kanamycin resistance we are able to transform the conjugative strain. As we said before, this conjugative strain already has ampicilin resistance, which is why we inserted our construction in a plasmid with a different resistance.</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>Before transforming this strain we need to prepair electrocompetent cells. So today we inoculated a colony of this strain in liquid LB-AMP at 37ºC to prepare the competent cells tomorrow.</p> | ||
| + | |||
| + | ''Fabi and Léo'' | ||
| + | |||
| + | |||
| + | ==== Confirming the New Device ==== | ||
| + | |||
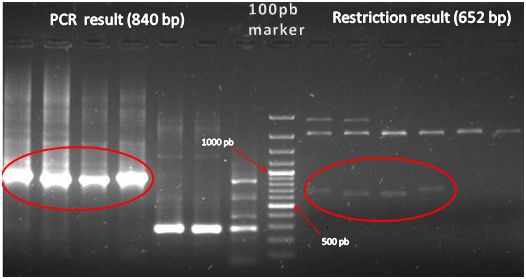
| + | *<p style=”text-align:justify;”>In order to confirm definitively our new part, we made two tests. We made PCR with VF2 and VR primers and we made enzyme restriction analysis with EcoRI and PstI. After run the agarose gel, we finally confirm our new part. We expected 840 bp (for PCR) and 670 pb (Restriction enzyme). We get it!!! Yes!! [[Image:New_biobrick.JPG|500px|center]] | ||
| + | |||
| + | ''Luige, Ane and Marcos'' | ||
==''' YeastGuard '''== | ==''' YeastGuard '''== | ||
| - | ====New | + | ====[https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/pGEMStrategy New Strategy: pGEM]==== |
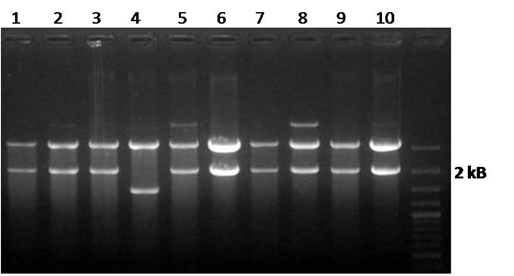
| - | * | + | * We did miniprep of 10 ''ADH1''+Lysozyme colonies, then we digested the ''ADH1''+Lysozyme/biofusion minipreps in order to connect it to the yeast expression vector (YEP358). We used the ''Xba''I and ''Pst''I enzymes. 9 of 10 digestions worked! =) The expected fragment size is 2000bp. |
| - | + | [[Image:20091021_confirmacao_adh1lys_results.png|300px|center]] | |
| - | + | ||
| - | * | + | * We chose two fragments and purified them from the agarose gel. We ligated them to YEP358. We will transform it tomorrow. |
Latest revision as of 03:47, 22 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"