|
|
| (19 intermediate revisions not shown) |
| Line 1: |
Line 1: |
| | {{:Team:UNICAMP-Brazil/inc_topo}} | | {{:Team:UNICAMP-Brazil/inc_topo}} |
| | + | __NOTOC__ |
| | =The Coliguard - Differentiation= | | =The Coliguard - Differentiation= |
| - |
| |
| - |
| |
| | | | |
| | ==Introduction== | | ==Introduction== |
| | + | <p style=”text-align:justify;”>Cell differentiation process can be defined as a “Progressive restriction of the developmental potential and increasing specialization of function that leads to the formation of specialized cells, tissues, and organs”, according to NCBI’s MeSH Database(1). Generally, it is a process by which a cell acquires a new morphological/functional type, capable of performing different and brand new tasks. In most multicellular organism, however, we might find a kind of differentiation that results in a type of cell providing support for another one(2). The differentiation mechanism chosen for our project relies on this perspective, and the first attempt at its implementation as a synthetic biology tool was made by the Paris 2007 iGEM team(3).</p> |
| | + | <p style=”text-align:justify;”>The differentiation system adopted for our project was designed focusing the need of creating two subpopulations with distinct characteristics and, mainly, the need of controlling the proportions between both subpopulations. Therefore, we need a differentiation system whose rate could be controlled.</p> |
| | + | <p style=”text-align:justify;”>Accordingly to our project objectives, the final results of this differentiation system must be:</p> |
| | | | |
| - | Cell differentiation process can be defined as a “Progressive restriction of the developmental potential and increasing specialization of function that leads to the formation of specialized cells, tissues, and organs”, according to NCBI’s MeSH Database(1). Generally, it is a process by which a cell acquires a new morphological/functional type, capable of performing different and brand new tasks. In most multicellular organism, however, we might find a kind of differentiation that result in a type of cell providing support to another one(2). The differentiation mechanism chosen for our project relies on this perspective, whose attempt to implement this tool into synthetic biology was first started by Paris 2007 iGEM team(3).
| + | • '''A Worker subpopulation''' |
| | + | This lineage is the one responsible for producing the valuable products, being unable to recognize and destroy contaminants present in the culture medium. In an attempt to maximize productivity, it would be interesting to keep the largest proportion of this subpopulation in the culture media. |
| | | | |
| - | The differentiation system adopted for our project was designed focusing the need of creating two subpopulations with distinct characteristics and, mainly, the need of controlling the proportions between both subpopulations. Therefore, we need a differentiation system whose rate could be controlled.
| + | • '''A Killer subpopulation''' |
| | + | This lineage is the one responsible for recognizing and destroying contaminants present in the culture media. This subpopulation is also able to produce the compound of interest, but since its metabolism must be relocated for maintaining the recognition and destruction systems, it should produce the compound in a much lower concentration. To guarantee maximum efficiency of our system, this subpopulation must remain in a basal low proportion, which would be greatly increased in the presence of contaminants. Once they are fully eliminated, our basal proportions must be automatically restored. |
| | | | |
| - | Accordingly to our project objectives, the final results of this differentiation system must be:
| + | ==The mechanism== |
| | | | |
| - | • [[A Worker subpopulation]]
| + | In order to design the suggested differentiation system, we will need to modify the expression pattern of several genes, introduce new constructions, and associate two different systems previously proposed by iGEM teams: the Slippage random mechanism by team Caltech 2008(4) and the Cre-recombinase mechanism proposed by team Paris 2007(3). |
| - | The one responsible for producing the industry compounds of interest, being unable to recognize and destroy contaminants present in the culture medium. In an attempt to maximize productivity, it would be interesting to keep the largest proportion of this subpopulation into the media.
| + | |
| | | | |
| - | • [[A Killer subpopulation]]
| |
| - | The one responsible for recognizing and destroying contaminants present in the culture media. This subpopulation also has productivity capacity, yet it could be much lower than the Worker subpopulation since a part of its energetic metabolism must be relocated for maintaining the recognition and destruction systems. To guarantee maximum efficiency of our system, this subpopulation must remain in a basal low proportion, which would be greatly increased in the presence of contaminants. Once they are fully eliminated, our basal proportions must be automatically restored.
| |
| | | | |
| | + | '''The slippage mechanism controls the basal proportions of both lineages''' |
| | | | |
| - | ==The mechanism==
| + | Slippage is just one of the numerous errors that can be made by DNA Polymerase, and in which the number of tandem repeats of a short sequence (AGTC) could be changed during the transcriptional process. |
| | + | Our construction will consist of 10 repetitions of the sequence AGTC, with an ATG start codon on the upstream position and the Cre-recombinase coding gene on the downstream position, everything under control of a constitutive promoter. In most cases, the slippage error won’t occur and the 10 repetitions of AGTC will leave the CRE-recombinase out of frame from the ATG, thus resulting in no expression of this recombinase. The absence of this expression in most cases will result in the expression of the Worker lineage characteristics, through mechanism that will be explained later. In the few cases in which the slippage error occurs and leaves a number of repeats that can be divided by three, i.e. from 10 to 9, the ATG and CRE-recombinase gene will be in the same reading frame, thus resulting in CRE-recombinase’s expressions and lead to Killer cells differentiation. |
| | | | |
| - | In order to design the suggested differentiation system, we will need to modify the expression pattern of several genes, introduce new constructions, and associate two different systems previously proposed by iGEM teams: the Slippage random mechanism by Caltech 2008(4) and the Cre-recombinase’s Paris 2007(3).
| |
| | | | |
| - | The slippage mechanism taking control of basal proportions
| + | ==How will CRE-recombinase’s expression result in Killer cell differentiation?== |
| | | | |
| - | Slippage is just one kind of the numerous DNA Polymerase’s mistakes, by which the number of tandem repeats of a determined sequence (AGTC) could be changed during transcriptional process. | + | CRE-recombinase is an enzyme that promotes an unidirectional recombination event between the lox66 and lox71 recombination sites(5). Therefore, we intend to adapt Paris 2007’s construction cassettes flanked by both lox sites made by team Paris 2007 by inserting our genes of interest into it. |
| - | Our construction will consist of 10 repetitions of AGTC, with an ATG start codon on the upstream position and the Cre-recombinase coding gene on the downstream position, everything under control of a constitutive promoter. In mostly cases, when slippage mistake doesn’t occurs, the (AGTC)10 wouldn’t let the ATG and Cre-recombinase into the same reading frame, hence resulting in no expression of this recombinase. The absence of this expression, found in mostly cases, would result in the Worker cells characteristics, through mechanism that will be explained later. As for the few cases in which the slippage occurs, the number of repetitions could easily turn from 10 to 9. This would let the ATG and Cre-recombinase gene into the same reading frame, thus resulting in Cre-recombinase’s expressions and into Killer cells differentiation. | + | According to the characteristics of our two subpopulations, the following genes might be necessary in order to promote cell differentiation and must be included in this construction: |
| | | | |
| | + | • GFP and Resistance Genes: as reporter, necessary only to assure whether or not the construction is inserted into genomic DNA. |
| | | | |
| - | ==How Cre-recombinase’s expression would result into Killer cells differentiation?==
| + | • ''ftsK'': essential gene for the maintenance of the full cell cycle. The importance of the cell cycle to our project will be explained later. |
| | | | |
| - | Cre-recombinase is an enzyme described to produce an unidirectional recombination between lox66 and lox71 recombination sites(5). Therefore, we intend to suit Paris 2007’s construction cassette flanked by both lox sites, in order to insert genes of our interest into it.
| + | • ''finOP'': conjugation inhibition system, related with the recognition and destruction system of the killer lineage. |
| - | According to the characteristics of our two subpopulations, the following genes might be necessary to be included into this construction:
| + | |
| | | | |
| - | • GFP and Resistance Genes: as a reporter, necessary only to assure whether or not the construction is inserted into genomic DNA.
| + | The general idea is that in the absence of CRE-recombinase expression (most cases), the resulting Worker cell has this construction fully operational, resulting in a cell capable of maintaining its cell cycle but unable to conjugate due to the presence of the ''finOP'' genes (Worker cell’s characteristics). On the other hand, the expression of the CRE-recombinase will result in the excision of this entire construction, resulting in a cell unable to maintain a complete cell cycle, but capable of conjugation (Killer cell’s characteristics). |
| | | | |
| - | • ftsK: essential gene on maintaining the full cell cycle, whose importance in our project will be later explained.
| |
| | | | |
| - | • finOP: conjugation inhibition system, related with the recognition and destruction cell capacity.
| + | ==Why is this difference related to conjugation capacity?== |
| - | Therefore, when we had the absence of Cre-recombinase expression (mostly cases), the resulting cell may has this construction fully operating, hence resulting in a cell capable of maintaining its cell cycle and unable to conjugate (Worker cell’s characteristics). The expression of Cre-recombinase, on the other hand, results in the cleavage of this entire construction, hence resulting in a cell unable to maintain its cell cycle but capable of conjugate (Killer cell’s characteristics).
| + | |
| | | | |
| - | | + | Conjugation plays an important role in recognition and destruction systems (see the specific sections for further information). In view of this, it’s very important to inhibit conjugation of the Worker cells, since they must concentrate on metabolite production and be unable to recognize and eliminate contaminants. |
| - | ==Why this difference related to conjugation capacity?==
| + | The conjugation inhibition will involve a system knows as ''finOP''. Briefly, this system consists of an antisense RNA (''finP'') and a small protein (''finO'')(9). The antisense RNA prevents ''traJ'' transcription, which lies immediately upstream of the operon and, in turn, is essential in activating the entire tra operon transcription(10). As for ''finO'', it binds to ''finP'' and ''traJ'', thereby allowing the duplex formation(11). Thus, for the correctly function of this system, both ''finO'' and ''finP'' must be expressed. |
| - | | + | |
| - | Conjugation plays an important role in recognition and destruction systems (see the specific sections for further information). Seeing that, it’s quite important to inhibit conjugation on Worker cells since it must not be able to recognize and eliminate contaminants. | + | |
| - | The conjugation inhibition will involve a system called finOP. Briefly, this system consists of an antisense RNA (finP) and a small protein (finO)(9). The antisense RNA prevents traJ transcription, which lies immediately upstream of the operon and, in turn, is essential in activating the entire tra operon transcription(10). As for finO, it binds to finP and traJ, thereby allowing the duplex formation(11). Thus, for the correctly function of this system, both finO and finP must be expressed. | + | |
| | | | |
| | | | |
| Line 54: |
Line 51: |
| | (1) It maintains a low basal proportion of Killer cells, in the absence of contaminants. | | (1) It maintains a low basal proportion of Killer cells, in the absence of contaminants. |
| | (2) It guarantees a quickly return to the basal proportion after the elimination of contaminants. | | (2) It guarantees a quickly return to the basal proportion after the elimination of contaminants. |
| - | As discussed previously, it’s quite important that the proportion of Killer cells could be increased in the presence of contaminants. On the other hand, it’s even more important that this proportion could quickly be restored to the basal levels since the contaminants were eliminated. The inability of completing cell cycles makes the Killer cells proportion directly and almost exclusively dependent to the specific stimulus trigged by the presence of contaminants. Thus, as soon as this stimulus ceases, the proportion of Killer cells would be drastically reduced since they are unable to reproduce and proliferate | + | |
| | + | As discussed previously, it’s very important that the proportion of Killer cells could be increased in the presence of contaminants. On the other hand, it’s even more important that this proportion can be quickly restored to the basal levels after contaminant elimination. The inability of completing cell cycles makes the Killer cells proportion directly and almost exclusively dependent to the specific stimulus trigged by the presence of contaminants. Thus, as soon as this stimulus ceases, the proportion of Killer cells would be drastically reduced, given that they would be unable to reproduce and proliferate. |
| | | | |
| | | | |
| - | ==How the basal proportion will change in the presence of contaminants?== | + | ==How will the basal proportion change in the presence of contaminants?== |
| | + | |
| | + | The answer is AI2 (auto-inducer 2). AI2 is a member of signaling molecules used in bacteria quorum sensing, produced by both gram-positive and gram-negative bacteria(6, 7 and 8). We thought that, since we could silence the ''AI2'' coding gene on our lineage, any detectable presence of this molecule would be due to the presence of contaminants, thereby working as a trigger signal. |
| | + | Thus, we decided to place the Cre-recombinase coding gene under the control of a promoter with sensibility to AI2. We will use ''AI2'' self promoter for that purpose, since there is a negative feedback control related to AI2 production. |
| | + | In short, when contaminants are present they are likely to produce AI2, which will trigger CRE-recombinase expression. This expression, as previously discussed, is responsible for Killer cells differentiation. |
| | | | |
| - | The answer is AI2 (auto-inducer 2). AI2 is a member of signaling molecules used in bacteria quorum sensing, produced by both gram-positive and gram-negative bacteria(6, 7 and 8). We thought that, since we could silence the AI2 coding gene on our lineage, any detectable presence of this molecule would be due to the presence of contaminants, thereby working as a trigger signal.
| |
| - | Thus, we decided to place the Cre-recombinase coding gene under the control of a promoter with sensibility to AI2. We will use AI2 self promoter for that purpose, since there is a negative feedback control related to AI2 production.
| |
| - | In short, when contaminants are present they probably will produce AI2, which will trigger Cre-recombinase expression. This expression, as previously discussed, is responsible for Killer cells differentiation.
| |
| | | | |
| - |
| |
| | ==Strategy== | | ==Strategy== |
| | | | |
| | As we previously explained, the model that we´ve developed to the differentiation subpart of our project involves the modification and coupling of two strategies previously used by the teams of Paris 2007 and Caltech 2008. | | As we previously explained, the model that we´ve developed to the differentiation subpart of our project involves the modification and coupling of two strategies previously used by the teams of Paris 2007 and Caltech 2008. |
| - | The Paris 2007 team used the FX58 E. coli strain, which has the construction Lox71-FSTK-Lox66 inserted in its genome. The FSTK gene is essential to cell division; its disruption stops cell division. The Lox71 and Lox66 sites are cleaved by a CRE recombinase, excising them and all the DNA sequences located between them. In Paris model, the Cre recombinase gene is under the control of dapA promoter. (for more information about Paris 2007 project, access the link: http://parts.mit.edu/igem07/index.php/Paris) | + | The Paris 2007 team used the ''FX58 E. coli'' strain, which has the construction ''Lox71-FSTK-Lox66'' inserted in its genome. The ''FSTK'' gene is essential to cell division; its disruption stops cell division. The ''Lox71'' and ''Lox66'' sites are cleaved by a CRE recombinase, excising them and all the DNA sequences located between them. In Paris model, the Cre recombinase gene is under the control of ''dapA'' promoter. (for more information about Paris 2007 project, access the link: https://2007.igem.org/Paris) |
| - | The Caltech 2008 team inserted the repetitive sequence AGCT10 between the start codon of the GFP gene and the rest of its ORF. In this case, the ORF is not in frame with its start codon, so the GFP is not corrected translated. However, DNA polymerase may slip when it replicates repetitions of small nucleotide polymers. So, during the replication of this construction, in some cases the AGCT10 is replaced by AGCT9. In this case, the GFP ORF is in frame with its start codon, and the protein is corrected translated. | + | The Caltech 2008 team inserted the repetitive sequence AGCT10 between the start codon of the ''GFP'' gene and the rest of its ORF. In this case, the ORF is not in frame with its start codon, so the ''GFP'' is not corrected translated. However, DNA polymerase may slip when it replicates repetitions of small nucleotide polymers. So, during the replication of this construction, in some cases the AGCT10 is replaced by AGCT9. In this case, the ''GFP'' ORF is in frame with its start codon, and the protein is corrected translated. |
| - | In our model, we aimed to insert two constructions flanking the FSTK gene; a) an upstream construction: LOX71-GFP; and b) a downstream construction: FinO-FinP-Lox66. The presence of finO and FinP genes impede the cell to conjugate. Moreover, we aimed to insert the repetition AGCT10 between the Cre recombinase start codon and the rest of its ORF; when Cre recombinase ORF is in frame with the start codon, it will be translated and act on the Lox based construction, excising the FSTK, FinO and FinP genes; so, the cell will lose its division skills, however it will be able to conjugate. | + | In our model, we aimed to insert two constructions flanking the ''FSTK'' gene; a) an upstream construction: ''LOX71-GFP''; and b) a downstream construction: ''FinO-FinP-Lox66''. The presence of ''finO'' and ''FinP'' genes impede the cell to conjugate. Moreover, we aimed to insert the repetition AGCT10 between the Cre recombinase start codon and the rest of its ORF; when Cre recombinase ORF is in frame with the start codon, it will be translated and act on the ''Lox'' based construction, excising the ''FSTK'', ''FinO'' and ''FinP'' genes; so, the cell will lose its division skills, however it will be able to conjugate. |
| | | | |
| | | | |
| Line 76: |
Line 74: |
| | The constructions we´ve developed to this part of the project are described below. | | The constructions we´ve developed to this part of the project are described below. |
| | | | |
| - | 1) [[BBa_K284043]]: AGTC repetition and CRE recombinase under constitutive promoter | + | 1) [[BBa_K284043]]: AGTC repetition and ''CRE'' recombinase under constitutive promoter |
| - | After the amplification of CRE recombinase by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284031. The following steps were also based on the standard assembly strategy, gathering the biobricks BBa_R0040 (pTet promoter), BBa_284040 (AGCT repetition), BBa_284031 (Cre recombinase without the start codon) and BBa_B0015 (double terminator), resulting in BBa_K284043. | + | After the amplification of ''CRE'' recombinase by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284031. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (pTet promoter), BBa_284040 (AGCT repetition), BBa_284031 (Cre recombinase without the start codon) and BBa_B0015 (double terminator), resulting in BBa_K284043. |
| | | | |
| | [[Image:Imagem6.jpg|center|500px]] | | [[Image:Imagem6.jpg|center|500px]] |
| | | | |
| - | 2) BBa_K284090: Downstream construction flanking FSTK gene | + | 2) BBa_K284090: Upstream construction flanking ''FSTK'' gene |
| - | This construction was developed to be inserted in the downstream region of the FSTK gene. Lox 71 is a cleavage site for CRE recombinase. This biobrick was assembled according to the standard protocol, gathering the previously existing biobricks BBa_I718017 (Lox 71), BBa_R0040 (pTet promoter), RBS (BBa_B0030), BBa_E0040 (green fluorescent protein ORF) and BBa_B0015 (double terminator). | + | This construction was developed to be inserted in the upstream region of the ''FSTK'' gene. Lox 71 is a cleavage site for ''CRE'' recombinase. This biobrick was assembled according to the standard protocol, joining the previously existing biobricks BBa_I718017 (Lox 71), BBa_R0040 (pTet promoter), RBS (BBa_B0030), BBa_E0040 (green fluorescent protein ORF) and BBa_B0015 (double terminator). |
| | | | |
| | | | |
| Line 88: |
Line 86: |
| | | | |
| | | | |
| - | 3) Upstream construction flanking the FSTK gene | + | 3) Downstream construction flanking the ''FSTK'' gene |
| | | | |
| | This construction is a composite part formed by the biobricks BBa_K284060, BBa_K284071 and BBa_I718017. | | This construction is a composite part formed by the biobricks BBa_K284060, BBa_K284071 and BBa_I718017. |
| | | | |
| - | 3.1) [[BBa_K284060]]: FinP under constitutive promoter | + | 3.1) [[BBa_K284060]]: ''FinP'' under constitutive promoter |
| | | | |
| | | | |
| Line 98: |
Line 96: |
| | | | |
| | | | |
| - | After the amplification of FinP (from E. coli F plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284033. The following steps were also based on the standard assembly strategy, gathering the biobricks BBa_R0040 (pTet promoter), BBa_B0030 (RBS), BBa_284033 (FinP form F plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284060. | + | After the amplification of ''FinP'' (from ''E. coli'' F plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284033. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (pTet promoter), BBa_B0030 (RBS), BBa_284033 (''FinP'' form F plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284060. |
| | | | |
| | 3.2) [[BBa_K284071]]: FinO under constitutive promoter | | 3.2) [[BBa_K284071]]: FinO under constitutive promoter |
| - | After the amplification of FinO (from E. coli R100 plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284032. The following steps were also based on the standard assembly strategy, gathering the biobricks BBa_R0040 (pTet promoter), BBa_B0030 (RBS), BBa_284032 (FinO form R100 plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284071. | + | After the amplification of FinO (from E. coli R100 plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284032. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (''pTet'' promoter), BBa_B0030 (''RBS''), BBa_284032 (''FinO'' form R100 plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284071. |
| | | | |
| | | | |
| Line 107: |
Line 105: |
| | | | |
| | | | |
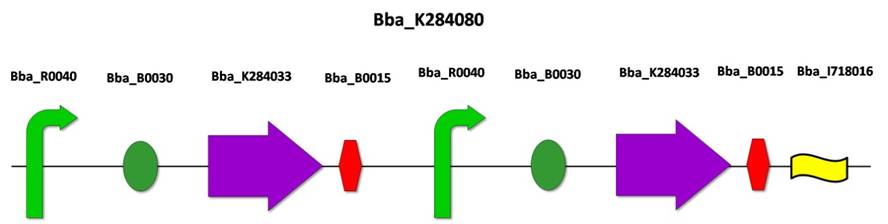
| - | 3.3) [[BBa_K284080]]: Upstream construction flanking FSTK gene | + | 3.3) [[BBa_K284080]]: Downstream construction flanking ''FSTK'' gene |
| - | This construction was developed to be inserted in the upstream region of the FSTK gene. Lox 66 is a cleavage site for CRE recombinase. This biobrick was assembled according to the standard protocol, gathering the biobrick BBa_K284060 (FinP under constitutive promoter), BBa_K284071 (FinO under constitutive promoter) and BBa_I718016 (Lox 66). | + | This construction was developed to be inserted in the downstream region of the ''FSTK'' gene. ''Lox 66'' is a cleavage site for ''CRE'' recombinase. This biobrick was assembled according to the standard protocol, joining the biobrick BBa_K284060 (''FinP'' under constitutive promoter), BBa_K284071 (''FinO'' under constitutive promoter) and BBa_I718016 (''Lox 66''). |
| | | | |
| | | | |
| Line 114: |
Line 112: |
| | | | |
| | | | |
| - | '''REFERENCES'''
| + | ===References=== |
| | + | ---- |
| | | | |
| | 1. http://www.ncbi.nlm.nih.gov/sites/entrez?db=mesh | | 1. http://www.ncbi.nlm.nih.gov/sites/entrez?db=mesh |
| Line 120: |
Line 119: |
| | 2. Weismann, A. The Germ Plasm. 1892 | | 2. Weismann, A. The Germ Plasm. 1892 |
| | | | |
| - | 3. http://parts.mit.edu/igem07/index.php/Paris | + | 3. https://2007.igem.org/Paris |
| | | | |
| | 4. https://2008.igem.org/Team:Caltech | | 4. https://2008.igem.org/Team:Caltech |
| | | | |
| - | 5. Zhang, Z. and Lutz, B. Cre recombinase-mediated inversion using lox66 and lox71: method to introduce conditional point mutations into the CREB-binding protein. Nucleic Acid Research. | + | 5. Zhang, Z. and Lutz, B. Nucleic Acid Research. 2002, 30 (17), 1-5. |
| | | | |
| - | 6. Cao, J.G. & Meighen, E.A. J Biol Chem 264, 21670-21676 (1989) | + | 6. Cao, J.G. & Meighen, E.A. J Biol Chem. 1989, 264, 21670-21676. |
| | | | |
| - | 7. Miller, S.T. et al. Mol Cell 15, 677-687 (2004) | + | 7. Miller, S.T. et al. Mol Cell. 2004, 15, 677-687. |
| | | | |
| - | 8. Miller, M. B.; Bassler, B. L. Annu. Rev. Microbiol. 2001, 55, 165-199 | + | 8. Miller, M. B.; Bassler, B. L. Annu. Rev. Microbiol. 2001, 55, 165-199. |
| | | | |
| - | 9. Gubbins, M.J et al. J. Biol Chem 278, pp. 27663–27671 | + | 9. Gubbins, M.J et al. J. Biol Chem. 2003, 278, 27663–27671. |
| | | | |
| - | 10. van Biesen, T. et al. Mol Microbiology 10, 35-43 | + | 10. Van Biesen, T. et al. Mol Microbiology. 1993, 10(1), 35-43. |
| | | | |
| - | 11. Koraimann, G. et al. Mol. Microbiology 21, 811-821 | + | 11. Koraimann, G. et al. Mol. Microbiology. 1996, 21, 811-821. |
| | | | |
| | | | |
| | | | |
| | {{:Team:UNICAMP-Brazil/inc_rodape}} | | {{:Team:UNICAMP-Brazil/inc_rodape}} |
The Coliguard - Differentiation
Introduction
Cell differentiation process can be defined as a “Progressive restriction of the developmental potential and increasing specialization of function that leads to the formation of specialized cells, tissues, and organs”, according to NCBI’s MeSH Database(1). Generally, it is a process by which a cell acquires a new morphological/functional type, capable of performing different and brand new tasks. In most multicellular organism, however, we might find a kind of differentiation that results in a type of cell providing support for another one(2). The differentiation mechanism chosen for our project relies on this perspective, and the first attempt at its implementation as a synthetic biology tool was made by the Paris 2007 iGEM team(3).
The differentiation system adopted for our project was designed focusing the need of creating two subpopulations with distinct characteristics and, mainly, the need of controlling the proportions between both subpopulations. Therefore, we need a differentiation system whose rate could be controlled.
Accordingly to our project objectives, the final results of this differentiation system must be:
• A Worker subpopulation
This lineage is the one responsible for producing the valuable products, being unable to recognize and destroy contaminants present in the culture medium. In an attempt to maximize productivity, it would be interesting to keep the largest proportion of this subpopulation in the culture media.
• A Killer subpopulation
This lineage is the one responsible for recognizing and destroying contaminants present in the culture media. This subpopulation is also able to produce the compound of interest, but since its metabolism must be relocated for maintaining the recognition and destruction systems, it should produce the compound in a much lower concentration. To guarantee maximum efficiency of our system, this subpopulation must remain in a basal low proportion, which would be greatly increased in the presence of contaminants. Once they are fully eliminated, our basal proportions must be automatically restored.
The mechanism
In order to design the suggested differentiation system, we will need to modify the expression pattern of several genes, introduce new constructions, and associate two different systems previously proposed by iGEM teams: the Slippage random mechanism by team Caltech 2008(4) and the Cre-recombinase mechanism proposed by team Paris 2007(3).
The slippage mechanism controls the basal proportions of both lineages
Slippage is just one of the numerous errors that can be made by DNA Polymerase, and in which the number of tandem repeats of a short sequence (AGTC) could be changed during the transcriptional process.
Our construction will consist of 10 repetitions of the sequence AGTC, with an ATG start codon on the upstream position and the Cre-recombinase coding gene on the downstream position, everything under control of a constitutive promoter. In most cases, the slippage error won’t occur and the 10 repetitions of AGTC will leave the CRE-recombinase out of frame from the ATG, thus resulting in no expression of this recombinase. The absence of this expression in most cases will result in the expression of the Worker lineage characteristics, through mechanism that will be explained later. In the few cases in which the slippage error occurs and leaves a number of repeats that can be divided by three, i.e. from 10 to 9, the ATG and CRE-recombinase gene will be in the same reading frame, thus resulting in CRE-recombinase’s expressions and lead to Killer cells differentiation.
How will CRE-recombinase’s expression result in Killer cell differentiation?
CRE-recombinase is an enzyme that promotes an unidirectional recombination event between the lox66 and lox71 recombination sites(5). Therefore, we intend to adapt Paris 2007’s construction cassettes flanked by both lox sites made by team Paris 2007 by inserting our genes of interest into it.
According to the characteristics of our two subpopulations, the following genes might be necessary in order to promote cell differentiation and must be included in this construction:
• GFP and Resistance Genes: as reporter, necessary only to assure whether or not the construction is inserted into genomic DNA.
• ftsK: essential gene for the maintenance of the full cell cycle. The importance of the cell cycle to our project will be explained later.
• finOP: conjugation inhibition system, related with the recognition and destruction system of the killer lineage.
The general idea is that in the absence of CRE-recombinase expression (most cases), the resulting Worker cell has this construction fully operational, resulting in a cell capable of maintaining its cell cycle but unable to conjugate due to the presence of the finOP genes (Worker cell’s characteristics). On the other hand, the expression of the CRE-recombinase will result in the excision of this entire construction, resulting in a cell unable to maintain a complete cell cycle, but capable of conjugation (Killer cell’s characteristics).
Why is this difference related to conjugation capacity?
Conjugation plays an important role in recognition and destruction systems (see the specific sections for further information). In view of this, it’s very important to inhibit conjugation of the Worker cells, since they must concentrate on metabolite production and be unable to recognize and eliminate contaminants.
The conjugation inhibition will involve a system knows as finOP. Briefly, this system consists of an antisense RNA (finP) and a small protein (finO)(9). The antisense RNA prevents traJ transcription, which lies immediately upstream of the operon and, in turn, is essential in activating the entire tra operon transcription(10). As for finO, it binds to finP and traJ, thereby allowing the duplex formation(11). Thus, for the correctly function of this system, both finO and finP must be expressed.
Why can’t Killer cells complete their cell cycle?
The inability of completing cell cycle works as a security device, as it wouldn’t let the Killer cells to proliferate and take over the entire culture, thus resulting in loss of productivity.
This security system works in two ways:
(1) It maintains a low basal proportion of Killer cells, in the absence of contaminants.
(2) It guarantees a quickly return to the basal proportion after the elimination of contaminants.
As discussed previously, it’s very important that the proportion of Killer cells could be increased in the presence of contaminants. On the other hand, it’s even more important that this proportion can be quickly restored to the basal levels after contaminant elimination. The inability of completing cell cycles makes the Killer cells proportion directly and almost exclusively dependent to the specific stimulus trigged by the presence of contaminants. Thus, as soon as this stimulus ceases, the proportion of Killer cells would be drastically reduced, given that they would be unable to reproduce and proliferate.
How will the basal proportion change in the presence of contaminants?
The answer is AI2 (auto-inducer 2). AI2 is a member of signaling molecules used in bacteria quorum sensing, produced by both gram-positive and gram-negative bacteria(6, 7 and 8). We thought that, since we could silence the AI2 coding gene on our lineage, any detectable presence of this molecule would be due to the presence of contaminants, thereby working as a trigger signal.
Thus, we decided to place the Cre-recombinase coding gene under the control of a promoter with sensibility to AI2. We will use AI2 self promoter for that purpose, since there is a negative feedback control related to AI2 production.
In short, when contaminants are present they are likely to produce AI2, which will trigger CRE-recombinase expression. This expression, as previously discussed, is responsible for Killer cells differentiation.
Strategy
As we previously explained, the model that we´ve developed to the differentiation subpart of our project involves the modification and coupling of two strategies previously used by the teams of Paris 2007 and Caltech 2008.
The Paris 2007 team used the FX58 E. coli strain, which has the construction Lox71-FSTK-Lox66 inserted in its genome. The FSTK gene is essential to cell division; its disruption stops cell division. The Lox71 and Lox66 sites are cleaved by a CRE recombinase, excising them and all the DNA sequences located between them. In Paris model, the Cre recombinase gene is under the control of dapA promoter. (for more information about Paris 2007 project, access the link: https://2007.igem.org/Paris)
The Caltech 2008 team inserted the repetitive sequence AGCT10 between the start codon of the GFP gene and the rest of its ORF. In this case, the ORF is not in frame with its start codon, so the GFP is not corrected translated. However, DNA polymerase may slip when it replicates repetitions of small nucleotide polymers. So, during the replication of this construction, in some cases the AGCT10 is replaced by AGCT9. In this case, the GFP ORF is in frame with its start codon, and the protein is corrected translated.
In our model, we aimed to insert two constructions flanking the FSTK gene; a) an upstream construction: LOX71-GFP; and b) a downstream construction: FinO-FinP-Lox66. The presence of finO and FinP genes impede the cell to conjugate. Moreover, we aimed to insert the repetition AGCT10 between the Cre recombinase start codon and the rest of its ORF; when Cre recombinase ORF is in frame with the start codon, it will be translated and act on the Lox based construction, excising the FSTK, FinO and FinP genes; so, the cell will lose its division skills, however it will be able to conjugate.
Constructions
The constructions we´ve developed to this part of the project are described below.
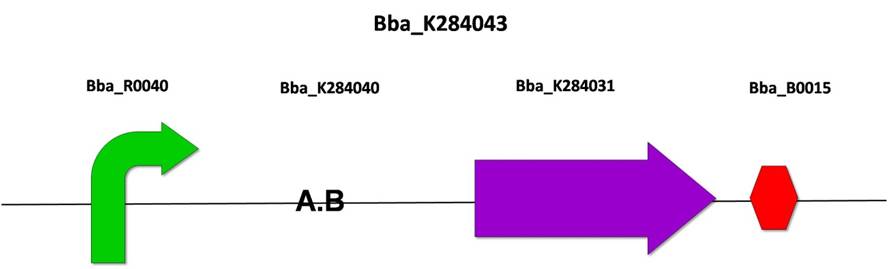
1) BBa_K284043: AGTC repetition and CRE recombinase under constitutive promoter
After the amplification of CRE recombinase by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284031. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (pTet promoter), BBa_284040 (AGCT repetition), BBa_284031 (Cre recombinase without the start codon) and BBa_B0015 (double terminator), resulting in BBa_K284043.
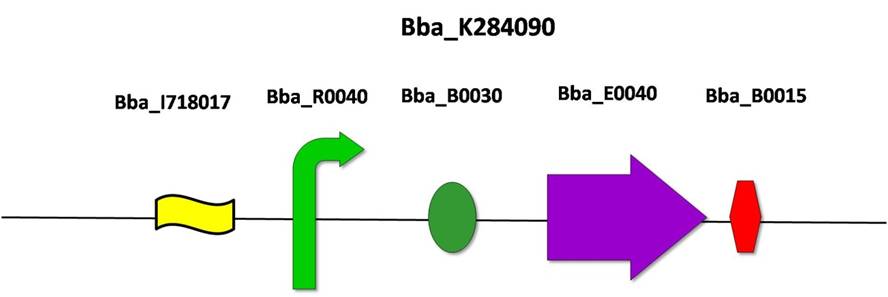
2) BBa_K284090: Upstream construction flanking FSTK gene
This construction was developed to be inserted in the upstream region of the FSTK gene. Lox 71 is a cleavage site for CRE recombinase. This biobrick was assembled according to the standard protocol, joining the previously existing biobricks BBa_I718017 (Lox 71), BBa_R0040 (pTet promoter), RBS (BBa_B0030), BBa_E0040 (green fluorescent protein ORF) and BBa_B0015 (double terminator).
3) Downstream construction flanking the FSTK gene
This construction is a composite part formed by the biobricks BBa_K284060, BBa_K284071 and BBa_I718017.
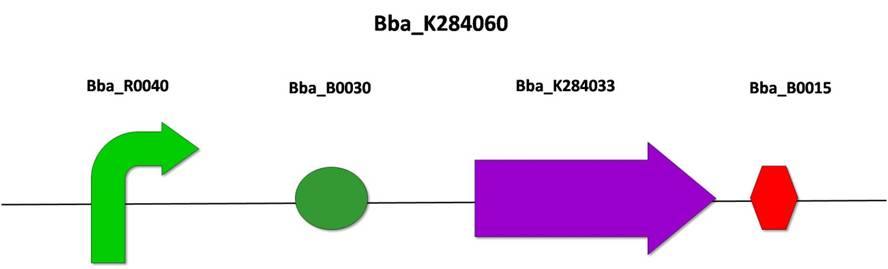
3.1) BBa_K284060: FinP under constitutive promoter
After the amplification of FinP (from E. coli F plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284033. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (pTet promoter), BBa_B0030 (RBS), BBa_284033 (FinP form F plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284060.
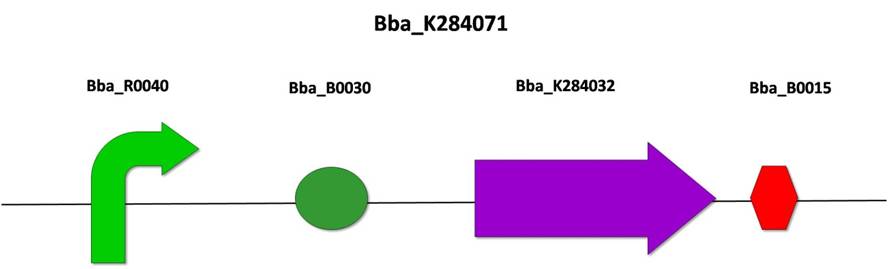
3.2) BBa_K284071: FinO under constitutive promoter
After the amplification of FinO (from E. coli R100 plasmid) by PCR, we cloned it into the PSB1A3 vector, according to the standard assembly strategy, resulting in the biobrick BBa_K284032. The following steps were also based on the standard assembly strategy, joining the biobricks BBa_R0040 (pTet promoter), BBa_B0030 (RBS), BBa_284032 (FinO form R100 plasmid) and BBa_B0015 (double terminator), resulting in BBa_K284071.
3.3) BBa_K284080: Downstream construction flanking FSTK gene
This construction was developed to be inserted in the downstream region of the FSTK gene. Lox 66 is a cleavage site for CRE recombinase. This biobrick was assembled according to the standard protocol, joining the biobrick BBa_K284060 (FinP under constitutive promoter), BBa_K284071 (FinO under constitutive promoter) and BBa_I718016 (Lox 66).
References
1. http://www.ncbi.nlm.nih.gov/sites/entrez?db=mesh
2. Weismann, A. The Germ Plasm. 1892
3. https://2007.igem.org/Paris
4. https://2008.igem.org/Team:Caltech
5. Zhang, Z. and Lutz, B. Nucleic Acid Research. 2002, 30 (17), 1-5.
6. Cao, J.G. & Meighen, E.A. J Biol Chem. 1989, 264, 21670-21676.
7. Miller, S.T. et al. Mol Cell. 2004, 15, 677-687.
8. Miller, M. B.; Bassler, B. L. Annu. Rev. Microbiol. 2001, 55, 165-199.
9. Gubbins, M.J et al. J. Biol Chem. 2003, 278, 27663–27671.
10. Van Biesen, T. et al. Mol Microbiology. 1993, 10(1), 35-43.
11. Koraimann, G. et al. Mol. Microbiology. 1996, 21, 811-821.
|






|
|
|
 "
"