Team:Newcastle/Labwork/25 August 2009
From 2009.igem.org
(→Stochastic Switch team) |
(→Preparing the DNA ladder) |
||
| Line 27: | Line 27: | ||
===Preparing the DNA ladder=== | ===Preparing the DNA ladder=== | ||
| - | We diluted the Lambda DNA/HindIII marker 10 times with the loading dye as below | + | We diluted the Lambda DNA/HindIII marker 10 times with the loading dye as below and placed the tube containing the mix into the fridge |
* 360ul loading dye | * 360ul loading dye | ||
* 40ul Lambda DNA/HindIII | * 40ul Lambda DNA/HindIII | ||
Revision as of 12:54, 26 August 2009
Contents |
Stochastic Switch team
Summary
Today we decided we should redo the minipreps from last weeks biobricks using the overnight cultures we set up yesterday. For the miniprep procedure we followed the extra guidelines that Chris advised us about. The final rehydrated plasmid minipreps were put into the -20 freezer in the 'iGEM plasmid minipreps' box. We also did a restriction digest of the two miniprep samples for biobrick C0178 and ran this on a 0.8% agarose gel. This went well, both samples having the fragment sizes expected. Tomorrow we will be carrying out another restriction digest of todays minipreps, however to save time tomorrow, we poured a gel and put it into the fridge ready to load the samples tomorrow. In order to make sure the prepared gel did not dry out, a small amount of 1% TAE was poured onto the gel, which was wrapped in cling film, clearly labelled and put into the fridge.
Restriction digest
Today we did restriction digest for a miniprep prepared for BBa_C0178. We prepared DNa ladder to load to the gels. At the end of the day, we prepared our gel to run tomorrow.We followed the restriction digest protocol for our two miniprep prepared for BBa_C0178 and used the followings to prepare the solutions.
- H20, 6.5 ul
- 10x Buffer, 2ul
- DNA from minipreps, 10ul
- EcoRI, 0.5 ul
- PstI, 0.5 ul
- BSA, 0.5 ul (It was diluted 100 times)
We gave a quick pulse to mix everything in the tubes before we put them into 37C incubator for an hour.
Running the gel
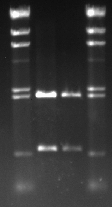
Metal container team had already prepared the gel for us. We placed the gel tray into the gel electrophoresis device and top it up with 1xTAE buffer. 9ul of DNA from each tube is combined with 1ul of buffer and loaded into the wells. DNA ladder was loaded into the 1st and the 4th wells, DNAs were loaded into the 2rd and the 3rd wells. Restriction digest should have cut the plasmid(pSB1A2) into two pieces, plasmid and the the biobrick, from EcoRI and PstI sites. pSB121A2 is 2079bp and BBa_C0178 is 609bp long. As it can be seen from the results below, they match with the HindIII markers.
Preparing the DNA ladder
We diluted the Lambda DNA/HindIII marker 10 times with the loading dye as below and placed the tube containing the mix into the fridge
- 360ul loading dye
- 40ul Lambda DNA/HindIII
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"