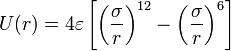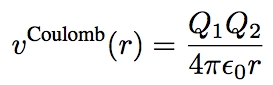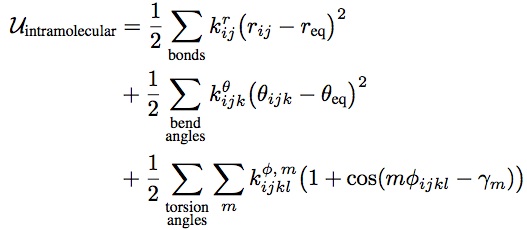Team:EPF-Lausanne/Theory
From 2009.igem.org
(Difference between revisions)
(→the non-bonded interactions:) |
|||
| Line 15: | Line 15: | ||
==the non-bonded interactions:== | ==the non-bonded interactions:== | ||
*The ''Lennard-Jones potential'' is the most commonly used form, with two parameters: σ, the diameter, and ε, the well depth. It takes into account the Van der Waals forces. It represents the non-bonded forces and the total potential energy can be calculated from the sum of energy contributions between pairs of atoms. [[Image:lennard_jones_vdw_forces.jpg|center]] | *The ''Lennard-Jones potential'' is the most commonly used form, with two parameters: σ, the diameter, and ε, the well depth. It takes into account the Van der Waals forces. It represents the non-bonded forces and the total potential energy can be calculated from the sum of energy contributions between pairs of atoms. [[Image:lennard_jones_vdw_forces.jpg|center]] | ||
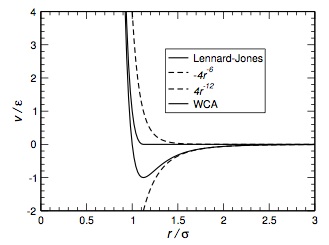
| - | [[Image:Lennard_jones.jpg|center|thumb|Lennard-Jones pair potential showing the r<sup>−12</sup> and r<sup>−6</sup> contributions]] | + | [[Image:Lennard_jones.jpg|center|300px|thumb|Lennard-Jones pair potential showing the r<sup>−12</sup> and r<sup>−6</sup> contributions]] |
*when electrostatic charges are present, we add the ''Coulomb force'', where Q1, Q2 are the charges and ϵ0 is the permittivity of free space | *when electrostatic charges are present, we add the ''Coulomb force'', where Q1, Q2 are the charges and ϵ0 is the permittivity of free space | ||
[[Image:Coulomb.jpg|200px|center]] | [[Image:Coulomb.jpg|200px|center]] | ||
| - | |||
==the bonded interactions:== | ==the bonded interactions:== | ||
Revision as of 07:24, 8 September 2009
Molecular dynamics simulation consists of the numerical, step-by-step, solution of the classical equations of motion. For this purpose we need to be able to calculate the forces acting on the atoms, and these are usually derived from a potential energy. This potential energy can be divided into:
the non-bonded interactions:
- The Lennard-Jones potential is the most commonly used form, with two parameters: σ, the diameter, and ε, the well depth. It takes into account the Van der Waals forces. It represents the non-bonded forces and the total potential energy can be calculated from the sum of energy contributions between pairs of atoms.
- when electrostatic charges are present, we add the Coulomb force, where Q1, Q2 are the charges and ϵ0 is the permittivity of free space
the bonded interactions:
angles, bonds and dihedral angles have to be taken into account
To understand a bit more, you can see the following article:
Introduction to Molecular Dynamics Simulation - Michael P. Allen
 "
"