Team:Newcastle/Labwork/18 September 2009
From 2009.igem.org
(→Analysing midi-prep digests) |
(→Analysing midi-prep digests) |
||
| Line 46: | Line 46: | ||
<font color="purple"><u>'''Just as a recap:'''</u> we had attempted to ligate the ''cotC'' BioBrick into ''pMUTIN4'' and had attempted to transform ''DH5-alpha E. coli'' with this product. Several colonies were found on the LB + chloramphenicol plate the trasnformants had been plated onto which suggested both the ligations and the transformations had worked well. Mini-preps were then carried out for 12 colonies in the [https://2009.igem.org/Team:Newcastle/Labwork/16_September_2009#a.29_Mini-preps_of_cotC-GFP-smtA_E._coli_transformants 16/09/09 Lab Session] and digested. When ran on gel, the 2 bands created did not match the expected sizes but there was consistency for most of the samples. This led us to think that it was the restriction enzymes to blame and ruled out contamination. | <font color="purple"><u>'''Just as a recap:'''</u> we had attempted to ligate the ''cotC'' BioBrick into ''pMUTIN4'' and had attempted to transform ''DH5-alpha E. coli'' with this product. Several colonies were found on the LB + chloramphenicol plate the trasnformants had been plated onto which suggested both the ligations and the transformations had worked well. Mini-preps were then carried out for 12 colonies in the [https://2009.igem.org/Team:Newcastle/Labwork/16_September_2009#a.29_Mini-preps_of_cotC-GFP-smtA_E._coli_transformants 16/09/09 Lab Session] and digested. When ran on gel, the 2 bands created did not match the expected sizes but there was consistency for most of the samples. This led us to think that it was the restriction enzymes to blame and ruled out contamination. | ||
| - | Based on this decision, the Metal Sensing team conducted a midi-prep on colony 8 LB culture (seen as colony 8 had the clearest bands) and had digested the DNA (see the [https://2009.igem.org/Team:Newcastle/Labwork/17_September_2009#1.29_Midi-prep_of_potential_cotC-GFP-smtA_transformant_E._coli_cells 17/09/09 Lab Session]) | + | Based on this decision, the Metal Sensing team conducted a midi-prep on colony 8 LB culture (seen as colony 8 had the clearest bands) and had digested the DNA (see the [https://2009.igem.org/Team:Newcastle/Labwork/17_September_2009#1.29_Midi-prep_of_potential_cotC-GFP-smtA_transformant_E._coli_cells 17/09/09 Lab Session]). The team then attempted to run 10ul of the digest on agrose gel through DNA gel electrophoresis but the DNA ladder was faulty and the bands seemed faint. Today's midi-prep analysis is a second attempt of this procedure. |
</font> | </font> | ||
Revision as of 16:44, 13 October 2009
Formal Lab Session - 18th September 2009
Stochastic Switch Team
Today we did midipreps of the ara transformation and digested 10ul to run on a gel (EcoRI + PstI) Two bands could be seen one for the backbone and one correctly sied for the insert (~200bp) so this midiprep can be taken forward in order to do the ara-sspb double clone.
Today we also made up some fresh DNA ladder as we have had problems with degradation.
- 40ul of 500ug/ml promega lambda HindIII digest (20ug in total)
- 280ul H2O
- 80ul loading dye
=400ul which was aliquoted into 4 tubes; 3 put in the freezer 1 in the fridge.
Metal Sensing Team
Introduction
Practical Outline
This is the initial list of tasks which need to be carried out by the end of the day:
- Start the Bacillus subtilis transformation process ("Day of transformation" steps of protocol) in the morning.
- Design primers for checking B. subtilis cotC integration
- Plate out some TPA2 cells for production of competent Dh5-alpha E. coli cells
- Run digests from yesterday (midi-prep digests) on gel
- If successful transform B. subtilis with midi-prep DNA
- If unsuccessful prepare another ligation attempt
- Carry out a second attempt at digesting the 5 midi-prep samples from the 11/09/09 Lab Session, i.e. cotC-GFP-smtA, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3
Procedure
Starting Bacillus subtilis transformations
The team started the first stage of the Bacillus subtilis transformation process by carrying out the beginning steps of the "Day of transformation" section shown in the protocol. If the midi-prep test digest analysis shows that the ligations and the transformation of cotC-GFP-smtA in the pMUTIN4 plasmid have worked then some of the remaining midi-prep DNA can be used to transform B. subtilis when the time is right.
Designing primers for cotC BioBrick integration
It is key that if the team manage to transform Bacillus subtilis with the cotC-GFP-smtA BioBrick in pMUTIN4 plasmid, they can determine that the BioBrick has successfully integrated into the B. subtilis genome. We have designed the BioBrick so that it integrates within the cotC gene of the B. subtilis genome so any primers designed should flank this region. Unfortunately, due to time restrictions, we didn't manage to carry this step out; it will be carried out another day.
Plating out TPA2 cells
It has been noted that the stock of competent DH5-alpha E. coli cells stored in the -80C freezer has been greatly reduced. To generate some more competent E. coli cells, some TPA2 cells (DH5-alpha E. coli) from the stocks freezer were taken and spread on an LB + agar plate. This is to fulfil the first step of the "Pre-culture" stage in the "Preparing Competent Cells" protocol. The plate will then be grown overnight in the 37C incubator.
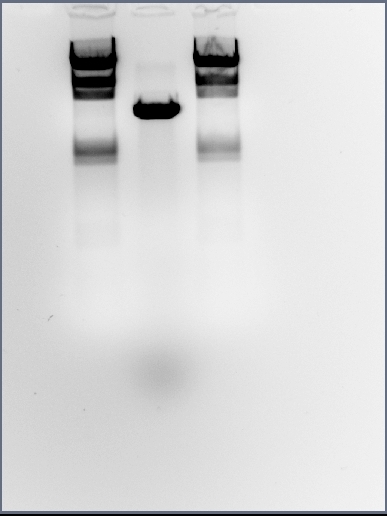
Analysing midi-prep digests
The midi-prep digest, which we are analysing today, was carried out in yesterday's lab session. The team hopes that the midi-prep DNA is that of cotC-GFP-smtA ligated into pMUTIN4 plasmid and only an analytical digest will determine this.
Just as a recap: we had attempted to ligate the cotC BioBrick into pMUTIN4 and had attempted to transform DH5-alpha E. coli with this product. Several colonies were found on the LB + chloramphenicol plate the trasnformants had been plated onto which suggested both the ligations and the transformations had worked well. Mini-preps were then carried out for 12 colonies in the 16/09/09 Lab Session and digested. When ran on gel, the 2 bands created did not match the expected sizes but there was consistency for most of the samples. This led us to think that it was the restriction enzymes to blame and ruled out contamination.
Based on this decision, the Metal Sensing team conducted a midi-prep on colony 8 LB culture (seen as colony 8 had the clearest bands) and had digested the DNA (see the 17/09/09 Lab Session). The team then attempted to run 10ul of the digest on agrose gel through DNA gel electrophoresis but the DNA ladder was faulty and the bands seemed faint. Today's midi-prep analysis is a second attempt of this procedure.
Analysing cotC, kinA, pGFP-rrnB, pMUTIN4 and pSB1AT3 midi-preps
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
News
Events
- 20 – 21 June 2009 - Europe workshop (London)
- 23 – 24 June 2009 - UK iGEM meetup (Edinburgh)
- 23 October Practice Presentation (Newcastle)
- 23 October T-shirts are ready
- 27 October Practice Presentation (Sunderland)
- 27 October Poster is ready
- 30 October – 2 November 2009 - Jamboree (Boston)
Social Net
- Newcastle iGEM Twitter
- [http://www.facebook.com/home.php#/group.php?gid=131709337641 Newcastle on Facebook]
- [http://www.youtube.com/user/newcastle2009igem Newcastle Youtube Channel]
 "
"