Back to Calendar
ColiGuard
Another PCR colony of BBa K112806 + BBa B0015, another failure...
A lot of colonies have grown again, we selected 15 to do the Colony PCR, but none of them was positive.
We are really worried with this, our time is finishing. We decided to chenge radically our strategy. We will change the terminator to BBa B0014 instead of BBa B0015 because the digestion of B0015 is not working. We let B0014 digesting ON with EcoRI and XbaI
Marcos
finO and finP with pGEM - confirmation
- We decided that we are going to confirm such ligations by performing:
-A Digestion: with XbaI and SpeI restriction enzymes, in order to release our parts from pGEM plasmid;
-A PCR: performed with the specific forward primer for our insert and with the reverse primer for pGEM plasmid (M13), in order to confirm that, once our inserts are indeed in there, they are also in the correct frame position.
- Seeing that, today we performed the digestion with EcoRI and SpeI for all of our minipreps samples that actually worked. Digestion lasted 3 hours.
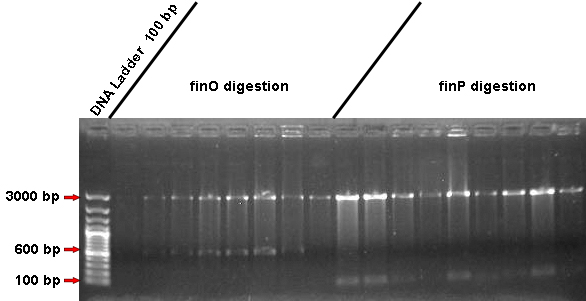
- According to the picture, the digestion excised a fragment of expected size (600 bp for finO and 120 bp for finP) in almost all samples! That's an important clue on confirming our inserts's correctly ligation!
Gabriel and Marcelo
YeastGuard
New strategy: pGEM
GEL
YFP+Terminator
pADH1+YFP (new biobrick)
Today we started the experiments to obtain a new device in biobrick format. It’s the ADH1+YFP biobrick.
First of all, we did these digestions: 1)YFP digestion using the enzymes XbaI and PstI; 2)pADH1 (biofusion) digestion using the enzymes SpeI and PstI.
Wesley and Gleidson
YEP
We transformed E. coli with YEP plasmid (Protocol 3)
|






|