Team:SupBiotech-Paris/Antitumor action
From 2009.igem.org
Enguerrand (Talk | contribs) |
|||
| Line 1: | Line 1: | ||
{{Template:Supbiotechcss12.css}} | {{Template:Supbiotechcss12.css}} | ||
{{Template:SupbiotechparisEn}} | {{Template:SupbiotechparisEn}} | ||
| + | |||
| + | |||
| + | = Cell targeting = | ||
| + | |||
| + | == Context == | ||
| + | |||
| + | After the [[Team:SupBiotech-Paris/Concept1#drapeau| tissue vector]] action, comes the [[Team:SupBiotech-Paris/Concept2#drapeau| cell vector]], this one is a modified bacteriophage which has the faculty to infect eukaryotic cells. Lambda phage, adapted to a concentrated presence of exogenous proteins, is a good candidate for an eukaryotic because of its high capacity of cloning and a capsid structure [[Team:SupBiotech-Paris/Concept1#drapeau| tissue vector]] design. The penton base originally from the adenovirus capsid appears like a promising candidate for lambda phage targeting. Indeed, it is endowed of several functions like the link to cell receptors, the internalisation of virales particles and the release of the capsid by the endosome.<br> | ||
| + | |||
| + | ==Objective == | ||
| + | |||
| + | Our objectives are to design a [[Team:SupBiotech-Paris/Concept2#drapeau| cell vector]] of Lambda phage type recombined with a penton base from the adenovirus 5 fused at its [[Team:SupBiotech-Paris/Biobricks#drapeau| D protein]]. The [[Team:SupBiotech-Paris/Concept1#drapeau| tissue vector]] should be able to integer the cell, go out of the endosome, transport its DNA to the nucleus of the cell and finally to transcript its [[Team:SupBiotech-Paris/Concept3#drapeau| therapeutic genes]]. <br> | ||
| + | |||
| + | |||
| + | <span style="float: right">[[Team:SupBiotech-Paris/Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | == Experimental approach == | ||
| + | |||
| + | In the framework of recombinant phage gene design we decided to fuse the adenovirus 5 penton base to the [[Team:SupBiotech-Paris/Biobricks#drapeau| D protein]] of the lambda phage. The protein D extraction from Lambda phage genome has been lead by polymerase chain reaction (PCR) with several primers couple. The same strategy has been taken for the adenovirus 5 penton base extraction from which has been extracted a plasmid coding for the virus gently offered by Dr. Karim Benihoud (UMR8121, CNRS/IGR, Villejuif, France). <br> | ||
| + | |||
| + | After fusion protein formation, this one is introduced in a BioBrick plasmid. This plasmid contains a resistivity against an antibiotic for the transfection confirmation of the recombined phage into the bacteria and a reporter gene like GFP with an eukaryotic promoter, the CMV of the <i>Simian virus</i> 40 (SV40), to confirm the transfection in eukaryotic cells. This strategy permits us to prove that the bacteriophage is able to infect eukaryotic cells. <br> | ||
| + | |||
| + | Unfortunately, we have not been able to build the fusion protein in time. However, scientific literature show that the [[Team:SupBiotech-Paris/Concept1#drapeau| tissue vector]] lambda phage type confection is possible by fusion of the penton base with the [[Team:SupBiotech-Paris/Biobricks#drapeau| D protein]] (Stefania Piersanti et al. 2004). The central sequence of the penton base, amino -acids 1 to 571, fused with the bacteriophage offer a transfection in eukaryotic cells, like the use of the RGD fragment responsible for the entry of the virus and the exit of the endosome, fragment 286 to 393. <br> | ||
| + | |||
| + | |||
| + | <span style="float: right">[[Team:SupBiotech-Paris/ Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | == Results == | ||
| + | |||
| + | === Design of the fusion protein === | ||
| + | |||
| + | For the design of the fusion protein, we decided to extract separately the penton base to the [[Team:SupBiotech-Paris/Biobricks#drapeau| D protein]] of the du lambda phage thanks to primers containing a BalI restriction site on the de la [[Team:SupBiotech-Paris/Biobricks#drapeau| protein D]] reverse primer and the penton base forward primer. Moreover the finale fusion protein contains specific BioBricks fragments to its ends. <br> | ||
| + | For these 2 genes extraction we used the following primers: <br> | ||
| + | |||
| + | |||
| + | First and second pair’s genes extraction: <br> | ||
| + | |||
| + | |||
| + | D protein of the Lambda phage: <br> | ||
| + | |||
| + | Forward : ATG-ACG-AGC-AAA-GAA-ACC-TT; <br> | ||
| + | Reverse : AAA-AAA-ATC-CCG-TAA-AAA-AAG-C. <br> | ||
| + | |||
| + | Adénovirus 5 penton Base : <br> | ||
| + | |||
| + | Forward : AAT-GGC-CAA-TGC-GGC-GCG-CGG-CGA-TG <br> | ||
| + | Reverse : CTG-CAG-CGG-CCG-CTA-CTA-GTA-TCA-AAA-AGT-GCG-G <br> | ||
| + | |||
| + | |||
| + | Third pair for extension of the BalI restriction site and the BioBrick prefix only for the [[Team:SupBiotech-Paris/Biobricks#drapeau|D protein]] (already done for the penton base). <br> | ||
| + | |||
| + | |||
| + | Forward : CGA-AAA-AAA-TGC-CCT-AAA-AAA-AAC-CGG-T <br> | ||
| + | Reverse : AAT-GGC-CAA-AAA-AAA-TCC-CGT-AAA-AAA-AGC <br> | ||
| + | |||
| + | |||
| + | Fourth pair for the D protein fusion amplification after ligation of the two fragments. <br> | ||
| + | |||
| + | |||
| + | Forward : CTT-AAG-CGC-CGG-CGA-AGA-TC <br> | ||
| + | Reverse : CTG-CAG-CGG-CCG-CTA-CTA-GTA <br><br> | ||
| + | |||
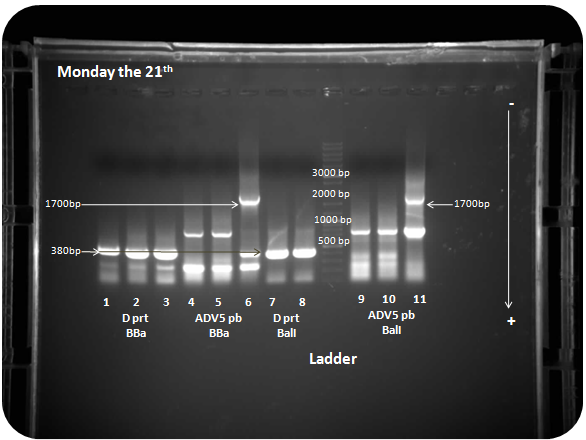
| + | PCR results are presented in figure X. We check that there is the right amplification size fragment 1715bp for the penton base of the sample number 9 and 385bp for the D protein in samples 5 and 6. However there is lots of mismatching during amplification cycles. This can have a negative effect on the result of the final amplification. <br> | ||
| + | |||
| + | [[image:M2109.png|center]] | ||
| + | |||
| + | <i>Figure 1: PCR of D protein BioBricks (1, 2, 3) and the penton base (4, 5, 6), D protein (5 and 6) and penton base (7, 8, 9) with BalI sites </i><br> | ||
| + | |||
| + | |||
| + | <span style=«float: right»>[[Team:SupBiotech-Paris/ Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | === Transfection of eukaryotic cells by the lambda phage recombined with the penton base fused to the D protein (Stefania Piersanti et al., 2004) === | ||
| + | |||
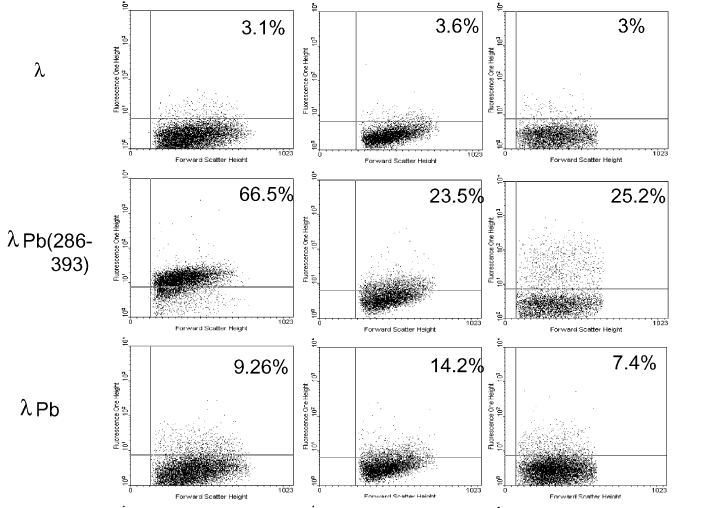
| + | A cytofluorimetric study has been done to analyze the transfection rate of recombined lambda phages. Figure X show cytofluorimetric results of COS-1 cells analyze after to have been exposed to a concentration of 10^6 PFU/cells of recombinants phages, Pb (1-571) or Pb (286-393). | ||
| + | |||
| + | [[image:VT1.png|center]] | ||
| + | |||
| + | [[image:VT2.png|center]] | ||
| + | |||
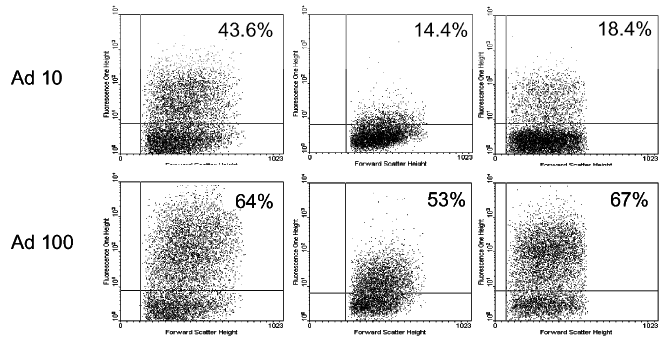
| + | <i> Figure 2 : Analyze of the GFP fluorescence on non recombined lambda phages (Lambda), recombined lambda phages with the fragment 286-393 of the penton base (LambdaPb286-393), recombined lambda phages with the complete penton base (1-571), GFP tagged adenovirus (Ad10 and Ad100)</i><br> | ||
| + | |||
| + | |||
| + | Firstly, we observe that the recombined phage show a tag difference independently of the fragment of the penton base used compared to the non transformed bacteriophage. Secondly, the recombined phage with the RGD fragment alone (286-393) has a higher fluorescence than the phage with a complete fragment and closer to the adenovirus one (figure X). <br> | ||
| + | |||
| + | |||
| + | <span style=«float: right»>[[Team:SupBiotech-Paris/ Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | == Discussion == | ||
| + | |||
| + | Even if the tissue vector has not been finished, the scientific literature shows that a recombinant phage creation with a protein coding the adenovirus penton base is possible. It demonstrated as well that fragments coding for RGD sequences alone have a higher capacity to infect eukaryotic cells compared to the penton base complete fragment de la (figure 2). In the case of our application it is possible to use a recombined lambda phage to insert our therapeutic gene. <br> | ||
| + | |||
| + | |||
| + | <span style=«float: right»>[[Team:SupBiotech-Paris/ Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | == Conclusion == | ||
| + | |||
| + | To conclude the RGD fragment of the penton base alone has a higher efficiency of interaction with integrines of eukaryotic cells, however for our project it was more judicious to use the complete sequence of the penton base (fragment 1-571) because with the use of the [[Team:SupBiotech-Paris/Concept2#drapeau| cell vector]] and the induction system by doxycycline give an injection very fast and targeted of our bacteriophages. The use of a highly efficient transfection system is not advised because phages have not the time to disperse properly and will infect several times the same cell. The use of the complete fragment of the penton base is sufficient for the phage to infect properly eukaryotic cells and let it time to have a bigger dispersion. <br> | ||
| + | |||
| + | |||
| + | <span style=«float: right»>[[Team:SupBiotech-Paris/ Antitumor_action #drapeau|Back to top]]</span> | ||
| + | |||
| + | |||
| + | |||
| + | = Application = | ||
| + | == Context == | ||
| + | |||
| + | In non-small cell lung cancer, or NSCLC, like in all other cancers, the loss of apoptotic capacity of tumor cells is due to the functional loss of various tumor suppressors incoming in the apoptotic pathway.<br> | ||
| + | |||
| + | The [[Team:SupBiotech-Paris/Introduction1En#drapeau|DVS]] application in the anticancer fight is based on the reactivation of this apoptotic pathway by bringing in tumor cells wild type genes coding for non-functional tumor suppressors.<br> | ||
| + | |||
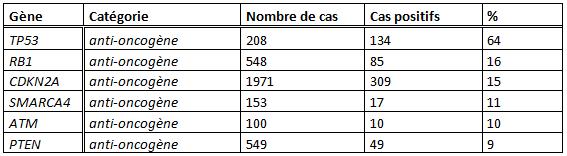
| + | The [http://www.sanger.ac.uk/genetics/CGP/cosmic/ COSMIC project] from [http://www.sanger.ac.uk/ Sanger institute] permits us to determine which genes to bring to the [[Team:SupBiotech-Paris/Concept3En#drapeau|therapeutic plasmid]] in the non-small cell lung cancer case. This project sums up all detected mutations for each type of cancer in function of their appearance frequency. So, from their data, the loss of apoptotic capacity of tumor cells for lung cancer can be due to the functional loss of proteins from the following genes :<br> | ||
| + | |||
| + | |||
| + | [[image: gènes mutés.jpeg]] | ||
| + | |||
| + | |||
| + | These different genes, that play a predominant role in the application of the apoptotic process and which are the most susceptible to be mutated in the lung cancer case, compose the [[Team:SupBiotech-Paris/Concept3En#drapeau|therapeutic plasmid]].<br> | ||
| + | |||
| + | == The objective == | ||
| + | |||
| + | The objective of this study is to check that if a wild type version of a tumor suppressor gene inside the tumor cell for which the own version is mutated, induce or not the apoptotic phenomenon.<br> | ||
| + | |||
| + | == Experimental approach == | ||
| + | |||
| + | |||
| + | === Cancer cell line and reported gene === | ||
| + | |||
| + | We select between several cell lines we had at disposal, a cancer cell line which the cancer origin is due to the mutation of a tumor suppressor gene. We possess the wild type of TP53 gene, the prostatic cancer p53 mutated DU-145 line retains our attention. <br> | ||
| + | So, we will test if bringing a wild type version of the p53 protein (p53wt) in the DU-145 cell line permits to induce an apoptotic process.<br> | ||
| + | |||
| + | |||
| + | <i>Cell culture protocol : </i><br> | ||
| + | <ol> | ||
| + | <li>Take out ampoule from liquid nitrogen<br> | ||
| + | <li>Place the ampoule in 37°C water bath for 5 minutes<br> | ||
| + | <li>In a 50 ml Falcon tube, put 9 ml of 10% MEM + 1 ml of ampoule<br> | ||
| + | <li>Harvest 5 min at 1200 rpm<br> | ||
| + | <li>Discard the supernatant without touching pellet cells (DMSO elimination) <br> | ||
| + | <li>Resuspend pellet in 1 ml of media<br> | ||
| + | <li>Put the suspension in a new T25 containing 5 ml of media<br> | ||
| + | <li>Incubate at 37°C<br> | ||
| + | <li>Do not forget to change the media the day after to eliminate all DMSO traces <br> | ||
| + | <li>One week later, cells are at 100% confluence<br> | ||
| + | </ol> | ||
| + | |||
| + | |||
| + | === TP53 gene incorporation === | ||
| + | |||
| + | Incorporation of the plasmid containing p53wt, pcDNA3 CMV+p53wt, insideDU-145 cells is done by electroporation. <br> | ||
| + | |||
| + | |||
| + | |||
| + | <i>Material :</i> <br> | ||
| + | <ul> | ||
| + | <li> DU-145 cells <br> | ||
| + | <li>pcDNA3 CMV+p53wt plasmid<br> | ||
| + | <li> Electrocompetent culture media<br> | ||
| + | <li>Trypsin<br> | ||
| + | <li>PBS<br> | ||
| + | <li>Icebox | ||
| + | <li>Electrotransfer Cuvette | ||
| + | <li>Centrifuge | ||
| + | <li>Incubator | ||
| + | <li>Electroporator (cliniporator) | ||
| + | </ul> | ||
| + | |||
| + | |||
| + | <i>Protocol: </i> <br> | ||
| + | <ol> | ||
| + | <li>Discard the media of T25 containing DU-145<br> | ||
| + | <li>Rinse with PBS<br> | ||
| + | <li>Add 500 µl of trypsin and let it acts for 3 minutes at room temperature <br> | ||
| + | <li>Add 5 ml of 10% MEM to neutralize trypsin<br> | ||
| + | <li>Suspend cells<br> | ||
| + | <li>Recover media containing DU-145 in a tube and harvest at 1000rpm for 10 minutes<br> | ||
| + | <li> Discard the supernatant and resuspend the pellet in Xµl (X= 90µl x Number of cuvettes) of electrocompetent media (around 5x105 cells per cuvettes) <br> | ||
| + | <li>Suspend your DNA solution in electrocompetent media (18x10-2g/L) <br> | ||
| + | <li>Add 10µl DNA solution per cuvette<br> | ||
| + | <li> Add 90µl of the cell suspension <br> | ||
| + | <li>Put cuvettes in ice<br> | ||
| + | <li>Pass cuvettes to the electroporator (cliniporator) and save each result <br> | ||
| + | <li>Incubate cuvettes at 37°C for 30 minutes<br> | ||
| + | <li>Put the content of each cuvette in a sterile tube, add 3ml of MEM 10% culture media, and incubate at 37°C for the necessitate time (until the annexin V assay) <br> | ||
| + | </ol> | ||
| + | |||
| + | |||
| + | === Apoptosis detection === | ||
| + | |||
| + | Detection of apoptotic cells is done by the annexin V assay: <br> | ||
| + | |||
| + | In the early stage of the apoptosis, we observe the phosphatidyl-serine translocation outside the cell membrane. These is highlighted by the specific fixation of the annexin V coupled with a fluorophore and analysed by flow cytometry. <br> | ||
| + | |||
| + | |||
| + | |||
| + | <i>Material :</i><br> | ||
| + | <ul> | ||
| + | <li> Propidium iodide 1 mg/ml Invitrogen stored cold in the fridge, diluted 10 times<br> | ||
| + | <li>Annexin V<br> | ||
| + | <li>Annexin buffer<br> | ||
| + | </ul> | ||
| + | |||
| + | |||
| + | Work as much as possible in the dark (fluorophores are photolabile) <br> | ||
| + | |||
| + | |||
| + | <i>Protocol : </i><br> | ||
| + | <ol> | ||
| + | <li>Recover culture media (3 ml), put it in a Falcon tube of 50 ml<br> | ||
| + | <li>Rinse the culture with 3 ml of PBS, and dispose it in the Falcon tube<br> | ||
| + | <li>Remove cells with trypsin, and dispose them in the Falcon tube<br> | ||
| + | <li>Harvest<br> | ||
| + | <li>Resuspend the pellet in 0.5 or 1 ml of cold PBS in function of the confluence level<br> | ||
| + | <li>Take 10 µl to count and harvest<br> | ||
| + | <li>Re-suspend the pellet in annexin buffer at a concentration of 1*106 cell/ml<br> | ||
| + | <li>Take 2 aliquots of 100 µl in 2 FACS tubes <br> | ||
| + | <li>Add in each tube 5 µl of annexin V and 1 µl of propidium iodide<br> | ||
| + | <li>Incubate 15 min at RT<br> | ||
| + | <li>Stop the reaction by put tubes in melting ice <br> | ||
| + | <li>Add 400 µl of annexin V buffer<br> | ||
| + | <li>Read in FACS as quick as possible and let tubes in the ice<br> | ||
| + | </ol> | ||
| + | |||
| + | == The sequence of studies == | ||
| + | |||
| + | The time of the plasmid expression in DU-145 cell line were not known so, we realized a kinetic monitoring of the apoptosis induction by making an annexin V assay every 6 hours for 48h after its electroporation. By the way, by coupling apoptosis rate of the population control (blank electroporation) and the population assay (electroporation with plasmid) with their respective growth rate, we will be able to determine the p53wt impact on apoptosis induction. The population control permits to eliminate cell death due to electroporation and to the culture transfer. <br> | ||
| + | |||
| + | Because we had not a continuous access to the cytometer, we grouped the all 48h analyses in 2 cytometry runs. Each time slot of the study is represented by a distinct cell population. So, we realized 14 electroporations corresponding to the 7 time slots: +6h, +12h, +18h, +24h, +30h, +36h and +48h (two by slot: population assay+ population control). <br> | ||
| + | |||
| + | |||
| + | Here is the allocation planning of electroporations: <br> | ||
| + | |||
| + | |||
| + | [[image:planning.jpeg]] | ||
| + | |||
| + | |||
| + | Three cell populations were respectively electropored 12h, 24h et 36h before the first cytometry run (in red, at 9h, day 3), four others 6h, 18h, 30h and 48h before the second run (in green, at 16h, day 3). <br> | ||
| + | |||
| + | The first cytometric analyze permits us to obtain data for the monitoring at +12h, +24h and +36h, while the second one, permits us to obtain data for the monitoring at +6h, 18h, +30h and +48h. <br> | ||
| + | |||
| + | By coupling all these data, we obtain a monitoring on 48h of the apoptosis induction after p53wt electroporation. <br> | ||
| + | |||
| + | == Results == | ||
| + | |||
| + | Each cell population, which represents different time range of the monitoring, has been subjected to an annexin V assay at the instant looked for. Unfortunately, a wrong dilution of the annexin buffer caused the death of each cell populations during the test. Even if results were convincing for the monitoring at +24h, +30h and +48h by simple comparison between the control and the test population in the microscope (figure 1), we could not confirm it by cytometric analyse. <br> | ||
| + | |||
| + | <center> | ||
| + | [[image:figure 1bis.jpeg]]<br> | ||
| + | <font size="1"><i>Figure 1</i> : cells morphology with or without p53 wild-type incorporation </font><br> | ||
| + | </center> | ||
| + | |||
| + | |||
| + | Because we could only start DU-145 culture at the beginning of October, the two weeks needed to reach the necessary confluence did not let the place to a second chance… <br> | ||
| + | |||
| + | |||
| + | However, several studies showed that the fact to bring p53 wild type into tumor mutated cells launched the apoptosis process. It is notably the case of the study leaded by Chunlin Yang in 1995, who were working, like us, on mutated p53 prostatic cancer cells (Tsu-pr1). The p53 wild type transfection were not realized by electroporation but by infecting tumor cells by non replicatives adenoviruses containing p53wt (AdCMV.p53). 48 hours after to have infected a tumor population with AdCMV.p53, a high expression of p53 is correlated with an important rate of cell death. If control populations (non-infected cells and cells infected with adenovirus containing lacZ gene, AdCMV.NLSßgal) show a similar and healthy morphology, condensation and cell detachment are observed in p53 infected population. To check if the death process followed by cells correspond to the apoptotic way, a migration on agaraose gel of their genome were realized.<br> | ||
| + | |||
| + | |||
| + | |||
| + | [[image:figure 2bis.jpeg|float|left]]<br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | <font size="1"><i>Figure 2 </i>: Electrophoresis on agar gel of isolated non-infected DNA cells (a), infected by AdCMV.NLSßgal (b) and AdCMV.p53 (c).</font> <br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | Infected by AdCMV.p53 cells show multiple bands (laddering pattern) while non-infected cells or AdCMV.NLSßgal infected cells show a unique one at high molecular weight. These results indicate that the cell induced by p53 wild type is from apoptotic origin with the observation of the genome division, consequence of the CAD (Caspase Activated DNase) activity, a specific endonuclease to the apoptoctic process. <br> | ||
| + | |||
| + | A MTT test permitted to quantify the effect induced by the p53 wild type expression into infected cells.<br> | ||
| + | |||
| + | [[image:figure3bis.jpeg|float|right]]<br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | <font size="1"><i>Figure 3 </i>: AdCMV.p53 effect on cell survive. Control and AdCMV.p53 infected cells were incubated in serum-free media after 1h of infection.</font> <br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | In serum absence, non-infected and ßgal infected cells continue to proliferate. In contrast, for p53 infected cells, proliferation is stopped and followed by an important fall of population. After 72h, nearly the totality of p53 infected cells are dead (figure 3). <br> | ||
| + | |||
| + | |||
| + | <u><i>According to this study, it appears clearly that the fact to bring a p53 wild-type version into the p53 mutated cell population induce the apoptosis phenomenon and decrease significantly the tumor population.</i></u><br> | ||
| + | |||
| + | |||
| + | |||
| + | Similar results were reported by the study leaded by Corrado Cirielli (in 1999) but this time on the U251 cancer strain from a glioma. Same types of analyses than these realized during the previous study were done. <br> | ||
| + | |||
| + | |||
| + | <dt> Morphologic analyse of AdCMV.p53 infected cells (a), non-infected (b) or infected by AdCMV.NULL (c) : <br> | ||
| + | |||
| + | <dd>[[image:figure4bis.jpeg]]<br> | ||
| + | <font size="1"><i>Figure 4</i> : morphology AdCMV.p53 infected cells (a), non-infected (b) or infected by AdCMV.NULL (c), after one week infection. </font><br> | ||
| + | |||
| + | |||
| + | |||
| + | Control populations (b and c) proliferate and form a cell layer one week after the beginning of experiences while the control population (a) show very few adherent cells (important cell loss) and a consequent morphologic change: cells are spherical.<br> | ||
| + | |||
| + | |||
| + | <dt> AdCMV.p53 effect on DNA division :<br> | ||
| + | <dd>[[image:figrue5bis.jpeg|float|right]]<br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | <font size="1"><i>Figure 5 </i>: electrophoresis on agar gel of isolated DNA of non-infected cells, infected by AdCMV.NULL and AdCMV.p53.</font> <br> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | After AdCMV.p53 infection, U-251 cells show a division of their genome characteristic of the apoptosis process.<br> | ||
| + | |||
| + | |||
| + | <dt>Monitoring of the non-infected cells and infected by AdCMV.p53 or AdCMV.NULL cells by a MTT test:<br> | ||
| + | |||
| + | <dd><center>[[image:figure6bis.jpeg]]</center><br> | ||
| + | <font size="1"><i>Figure 6</i> : Control population proliferation (non-infected or AdCMV.NULL infected) and the test population by monitoring of the optical density after a MTT test.</font><br> | ||
| + | |||
| + | |||
| + | <dd> Non-infected cells and AdCMV.NULL infected cells proliferate in a significant way during the week of analysis while AdCMV.p53 infected cells present a total absence of proliferation and a continuous decrease of their population.<br> | ||
| + | |||
| + | |||
| + | <dd><u><i>This study show one more time that the fact to bring a p53wild-type version into a mutated p53 cell population induce cell death by apoptosis.</i></u><br> | ||
| + | |||
| + | |||
| + | == Conclusion == | ||
| + | |||
| + | |||
| + | |||
Revision as of 03:55, 21 October 2009
Contents |
Cell targeting
Context
After the tissue vector action, comes the cell vector, this one is a modified bacteriophage which has the faculty to infect eukaryotic cells. Lambda phage, adapted to a concentrated presence of exogenous proteins, is a good candidate for an eukaryotic because of its high capacity of cloning and a capsid structure tissue vector design. The penton base originally from the adenovirus capsid appears like a promising candidate for lambda phage targeting. Indeed, it is endowed of several functions like the link to cell receptors, the internalisation of virales particles and the release of the capsid by the endosome.
Objective
Our objectives are to design a cell vector of Lambda phage type recombined with a penton base from the adenovirus 5 fused at its D protein. The tissue vector should be able to integer the cell, go out of the endosome, transport its DNA to the nucleus of the cell and finally to transcript its therapeutic genes.
Experimental approach
In the framework of recombinant phage gene design we decided to fuse the adenovirus 5 penton base to the D protein of the lambda phage. The protein D extraction from Lambda phage genome has been lead by polymerase chain reaction (PCR) with several primers couple. The same strategy has been taken for the adenovirus 5 penton base extraction from which has been extracted a plasmid coding for the virus gently offered by Dr. Karim Benihoud (UMR8121, CNRS/IGR, Villejuif, France).
After fusion protein formation, this one is introduced in a BioBrick plasmid. This plasmid contains a resistivity against an antibiotic for the transfection confirmation of the recombined phage into the bacteria and a reporter gene like GFP with an eukaryotic promoter, the CMV of the Simian virus 40 (SV40), to confirm the transfection in eukaryotic cells. This strategy permits us to prove that the bacteriophage is able to infect eukaryotic cells.
Unfortunately, we have not been able to build the fusion protein in time. However, scientific literature show that the tissue vector lambda phage type confection is possible by fusion of the penton base with the D protein (Stefania Piersanti et al. 2004). The central sequence of the penton base, amino -acids 1 to 571, fused with the bacteriophage offer a transfection in eukaryotic cells, like the use of the RGD fragment responsible for the entry of the virus and the exit of the endosome, fragment 286 to 393.
Results
Design of the fusion protein
For the design of the fusion protein, we decided to extract separately the penton base to the D protein of the du lambda phage thanks to primers containing a BalI restriction site on the de la protein D reverse primer and the penton base forward primer. Moreover the finale fusion protein contains specific BioBricks fragments to its ends.
For these 2 genes extraction we used the following primers:
First and second pair’s genes extraction:
D protein of the Lambda phage:
Forward : ATG-ACG-AGC-AAA-GAA-ACC-TT;
Reverse : AAA-AAA-ATC-CCG-TAA-AAA-AAG-C.
Adénovirus 5 penton Base :
Forward : AAT-GGC-CAA-TGC-GGC-GCG-CGG-CGA-TG
Reverse : CTG-CAG-CGG-CCG-CTA-CTA-GTA-TCA-AAA-AGT-GCG-G
Third pair for extension of the BalI restriction site and the BioBrick prefix only for the D protein (already done for the penton base).
Forward : CGA-AAA-AAA-TGC-CCT-AAA-AAA-AAC-CGG-T
Reverse : AAT-GGC-CAA-AAA-AAA-TCC-CGT-AAA-AAA-AGC
Fourth pair for the D protein fusion amplification after ligation of the two fragments.
Forward : CTT-AAG-CGC-CGG-CGA-AGA-TC
Reverse : CTG-CAG-CGG-CCG-CTA-CTA-GTA
PCR results are presented in figure X. We check that there is the right amplification size fragment 1715bp for the penton base of the sample number 9 and 385bp for the D protein in samples 5 and 6. However there is lots of mismatching during amplification cycles. This can have a negative effect on the result of the final amplification.
Figure 1: PCR of D protein BioBricks (1, 2, 3) and the penton base (4, 5, 6), D protein (5 and 6) and penton base (7, 8, 9) with BalI sites
Transfection of eukaryotic cells by the lambda phage recombined with the penton base fused to the D protein (Stefania Piersanti et al., 2004)
A cytofluorimetric study has been done to analyze the transfection rate of recombined lambda phages. Figure X show cytofluorimetric results of COS-1 cells analyze after to have been exposed to a concentration of 10^6 PFU/cells of recombinants phages, Pb (1-571) or Pb (286-393).
Figure 2 : Analyze of the GFP fluorescence on non recombined lambda phages (Lambda), recombined lambda phages with the fragment 286-393 of the penton base (LambdaPb286-393), recombined lambda phages with the complete penton base (1-571), GFP tagged adenovirus (Ad10 and Ad100)
Firstly, we observe that the recombined phage show a tag difference independently of the fragment of the penton base used compared to the non transformed bacteriophage. Secondly, the recombined phage with the RGD fragment alone (286-393) has a higher fluorescence than the phage with a complete fragment and closer to the adenovirus one (figure X).
Discussion
Even if the tissue vector has not been finished, the scientific literature shows that a recombinant phage creation with a protein coding the adenovirus penton base is possible. It demonstrated as well that fragments coding for RGD sequences alone have a higher capacity to infect eukaryotic cells compared to the penton base complete fragment de la (figure 2). In the case of our application it is possible to use a recombined lambda phage to insert our therapeutic gene.
Conclusion
To conclude the RGD fragment of the penton base alone has a higher efficiency of interaction with integrines of eukaryotic cells, however for our project it was more judicious to use the complete sequence of the penton base (fragment 1-571) because with the use of the cell vector and the induction system by doxycycline give an injection very fast and targeted of our bacteriophages. The use of a highly efficient transfection system is not advised because phages have not the time to disperse properly and will infect several times the same cell. The use of the complete fragment of the penton base is sufficient for the phage to infect properly eukaryotic cells and let it time to have a bigger dispersion.
Application
Context
In non-small cell lung cancer, or NSCLC, like in all other cancers, the loss of apoptotic capacity of tumor cells is due to the functional loss of various tumor suppressors incoming in the apoptotic pathway.
The DVS application in the anticancer fight is based on the reactivation of this apoptotic pathway by bringing in tumor cells wild type genes coding for non-functional tumor suppressors.
The COSMIC project from Sanger institute permits us to determine which genes to bring to the therapeutic plasmid in the non-small cell lung cancer case. This project sums up all detected mutations for each type of cancer in function of their appearance frequency. So, from their data, the loss of apoptotic capacity of tumor cells for lung cancer can be due to the functional loss of proteins from the following genes :
These different genes, that play a predominant role in the application of the apoptotic process and which are the most susceptible to be mutated in the lung cancer case, compose the therapeutic plasmid.
The objective
The objective of this study is to check that if a wild type version of a tumor suppressor gene inside the tumor cell for which the own version is mutated, induce or not the apoptotic phenomenon.
Experimental approach
Cancer cell line and reported gene
We select between several cell lines we had at disposal, a cancer cell line which the cancer origin is due to the mutation of a tumor suppressor gene. We possess the wild type of TP53 gene, the prostatic cancer p53 mutated DU-145 line retains our attention.
So, we will test if bringing a wild type version of the p53 protein (p53wt) in the DU-145 cell line permits to induce an apoptotic process.
Cell culture protocol :
- Take out ampoule from liquid nitrogen
- Place the ampoule in 37°C water bath for 5 minutes
- In a 50 ml Falcon tube, put 9 ml of 10% MEM + 1 ml of ampoule
- Harvest 5 min at 1200 rpm
- Discard the supernatant without touching pellet cells (DMSO elimination)
- Resuspend pellet in 1 ml of media
- Put the suspension in a new T25 containing 5 ml of media
- Incubate at 37°C
- Do not forget to change the media the day after to eliminate all DMSO traces
- One week later, cells are at 100% confluence
TP53 gene incorporation
Incorporation of the plasmid containing p53wt, pcDNA3 CMV+p53wt, insideDU-145 cells is done by electroporation.
Material :
- DU-145 cells
- pcDNA3 CMV+p53wt plasmid
- Electrocompetent culture media
- Trypsin
- PBS
- Icebox
- Electrotransfer Cuvette
- Centrifuge
- Incubator
- Electroporator (cliniporator)
Protocol:
- Discard the media of T25 containing DU-145
- Rinse with PBS
- Add 500 µl of trypsin and let it acts for 3 minutes at room temperature
- Add 5 ml of 10% MEM to neutralize trypsin
- Suspend cells
- Recover media containing DU-145 in a tube and harvest at 1000rpm for 10 minutes
- Discard the supernatant and resuspend the pellet in Xµl (X= 90µl x Number of cuvettes) of electrocompetent media (around 5x105 cells per cuvettes)
- Suspend your DNA solution in electrocompetent media (18x10-2g/L)
- Add 10µl DNA solution per cuvette
- Add 90µl of the cell suspension
- Put cuvettes in ice
- Pass cuvettes to the electroporator (cliniporator) and save each result
- Incubate cuvettes at 37°C for 30 minutes
- Put the content of each cuvette in a sterile tube, add 3ml of MEM 10% culture media, and incubate at 37°C for the necessitate time (until the annexin V assay)
Apoptosis detection
Detection of apoptotic cells is done by the annexin V assay:
In the early stage of the apoptosis, we observe the phosphatidyl-serine translocation outside the cell membrane. These is highlighted by the specific fixation of the annexin V coupled with a fluorophore and analysed by flow cytometry.
Material :
- Propidium iodide 1 mg/ml Invitrogen stored cold in the fridge, diluted 10 times
- Annexin V
- Annexin buffer
Work as much as possible in the dark (fluorophores are photolabile)
Protocol :
- Recover culture media (3 ml), put it in a Falcon tube of 50 ml
- Rinse the culture with 3 ml of PBS, and dispose it in the Falcon tube
- Remove cells with trypsin, and dispose them in the Falcon tube
- Harvest
- Resuspend the pellet in 0.5 or 1 ml of cold PBS in function of the confluence level
- Take 10 µl to count and harvest
- Re-suspend the pellet in annexin buffer at a concentration of 1*106 cell/ml
- Take 2 aliquots of 100 µl in 2 FACS tubes
- Add in each tube 5 µl of annexin V and 1 µl of propidium iodide
- Incubate 15 min at RT
- Stop the reaction by put tubes in melting ice
- Add 400 µl of annexin V buffer
- Read in FACS as quick as possible and let tubes in the ice
The sequence of studies
The time of the plasmid expression in DU-145 cell line were not known so, we realized a kinetic monitoring of the apoptosis induction by making an annexin V assay every 6 hours for 48h after its electroporation. By the way, by coupling apoptosis rate of the population control (blank electroporation) and the population assay (electroporation with plasmid) with their respective growth rate, we will be able to determine the p53wt impact on apoptosis induction. The population control permits to eliminate cell death due to electroporation and to the culture transfer.
Because we had not a continuous access to the cytometer, we grouped the all 48h analyses in 2 cytometry runs. Each time slot of the study is represented by a distinct cell population. So, we realized 14 electroporations corresponding to the 7 time slots: +6h, +12h, +18h, +24h, +30h, +36h and +48h (two by slot: population assay+ population control).
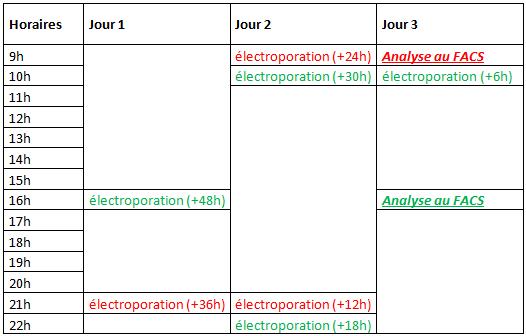
Here is the allocation planning of electroporations:
Three cell populations were respectively electropored 12h, 24h et 36h before the first cytometry run (in red, at 9h, day 3), four others 6h, 18h, 30h and 48h before the second run (in green, at 16h, day 3).
The first cytometric analyze permits us to obtain data for the monitoring at +12h, +24h and +36h, while the second one, permits us to obtain data for the monitoring at +6h, 18h, +30h and +48h.
By coupling all these data, we obtain a monitoring on 48h of the apoptosis induction after p53wt electroporation.
Results
Each cell population, which represents different time range of the monitoring, has been subjected to an annexin V assay at the instant looked for. Unfortunately, a wrong dilution of the annexin buffer caused the death of each cell populations during the test. Even if results were convincing for the monitoring at +24h, +30h and +48h by simple comparison between the control and the test population in the microscope (figure 1), we could not confirm it by cytometric analyse.
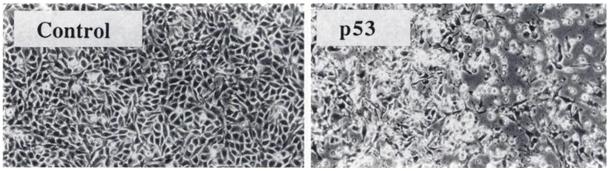
Figure 1 : cells morphology with or without p53 wild-type incorporation
Because we could only start DU-145 culture at the beginning of October, the two weeks needed to reach the necessary confluence did not let the place to a second chance…
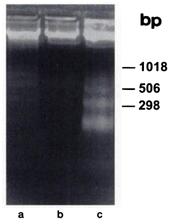
However, several studies showed that the fact to bring p53 wild type into tumor mutated cells launched the apoptosis process. It is notably the case of the study leaded by Chunlin Yang in 1995, who were working, like us, on mutated p53 prostatic cancer cells (Tsu-pr1). The p53 wild type transfection were not realized by electroporation but by infecting tumor cells by non replicatives adenoviruses containing p53wt (AdCMV.p53). 48 hours after to have infected a tumor population with AdCMV.p53, a high expression of p53 is correlated with an important rate of cell death. If control populations (non-infected cells and cells infected with adenovirus containing lacZ gene, AdCMV.NLSßgal) show a similar and healthy morphology, condensation and cell detachment are observed in p53 infected population. To check if the death process followed by cells correspond to the apoptotic way, a migration on agaraose gel of their genome were realized.
Figure 2 : Electrophoresis on agar gel of isolated non-infected DNA cells (a), infected by AdCMV.NLSßgal (b) and AdCMV.p53 (c).
Infected by AdCMV.p53 cells show multiple bands (laddering pattern) while non-infected cells or AdCMV.NLSßgal infected cells show a unique one at high molecular weight. These results indicate that the cell induced by p53 wild type is from apoptotic origin with the observation of the genome division, consequence of the CAD (Caspase Activated DNase) activity, a specific endonuclease to the apoptoctic process.
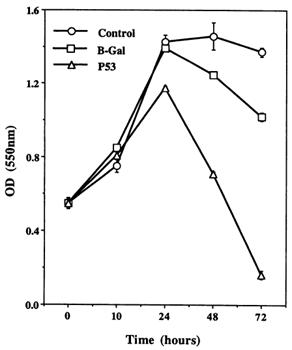
A MTT test permitted to quantify the effect induced by the p53 wild type expression into infected cells.
Figure 3 : AdCMV.p53 effect on cell survive. Control and AdCMV.p53 infected cells were incubated in serum-free media after 1h of infection.
In serum absence, non-infected and ßgal infected cells continue to proliferate. In contrast, for p53 infected cells, proliferation is stopped and followed by an important fall of population. After 72h, nearly the totality of p53 infected cells are dead (figure 3).
According to this study, it appears clearly that the fact to bring a p53 wild-type version into the p53 mutated cell population induce the apoptosis phenomenon and decrease significantly the tumor population.
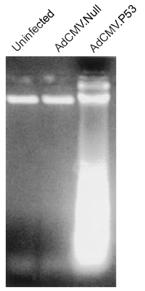
Similar results were reported by the study leaded by Corrado Cirielli (in 1999) but this time on the U251 cancer strain from a glioma. Same types of analyses than these realized during the previous study were done.
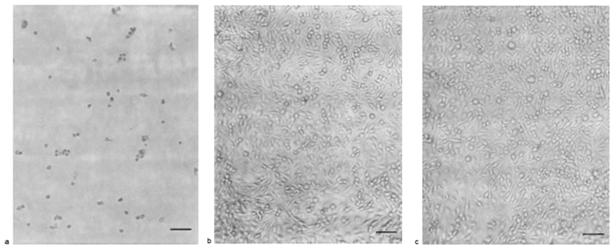
Figure 4 : morphology AdCMV.p53 infected cells (a), non-infected (b) or infected by AdCMV.NULL (c), after one week infection.
Control populations (b and c) proliferate and form a cell layer one week after the beginning of experiences while the control population (a) show very few adherent cells (important cell loss) and a consequent morphologic change: cells are spherical.
Figure 5 : electrophoresis on agar gel of isolated DNA of non-infected cells, infected by AdCMV.NULL and AdCMV.p53.
After AdCMV.p53 infection, U-251 cells show a division of their genome characteristic of the apoptosis process.
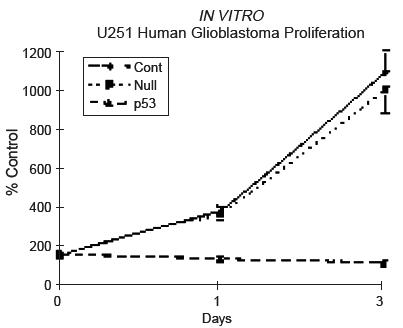
Figure 6 : Control population proliferation (non-infected or AdCMV.NULL infected) and the test population by monitoring of the optical density after a MTT test.
Conclusion
 "
"