Team:Cambridge/Notebook/Week4
From 2009.igem.org
MikeDavies (Talk | contribs) (→Dry Work) |
MikeDavies (Talk | contribs) (→Progress on carotenoid (Tuesday)) |
||
| Line 111: | Line 111: | ||
********************PHOTO**************** | ********************PHOTO**************** | ||
| + | [[Image:Cambridge gel1.JPG|left|360px]] | ||
| + | [[Image:Cambridge gel2.JPG|right|360px]] | ||
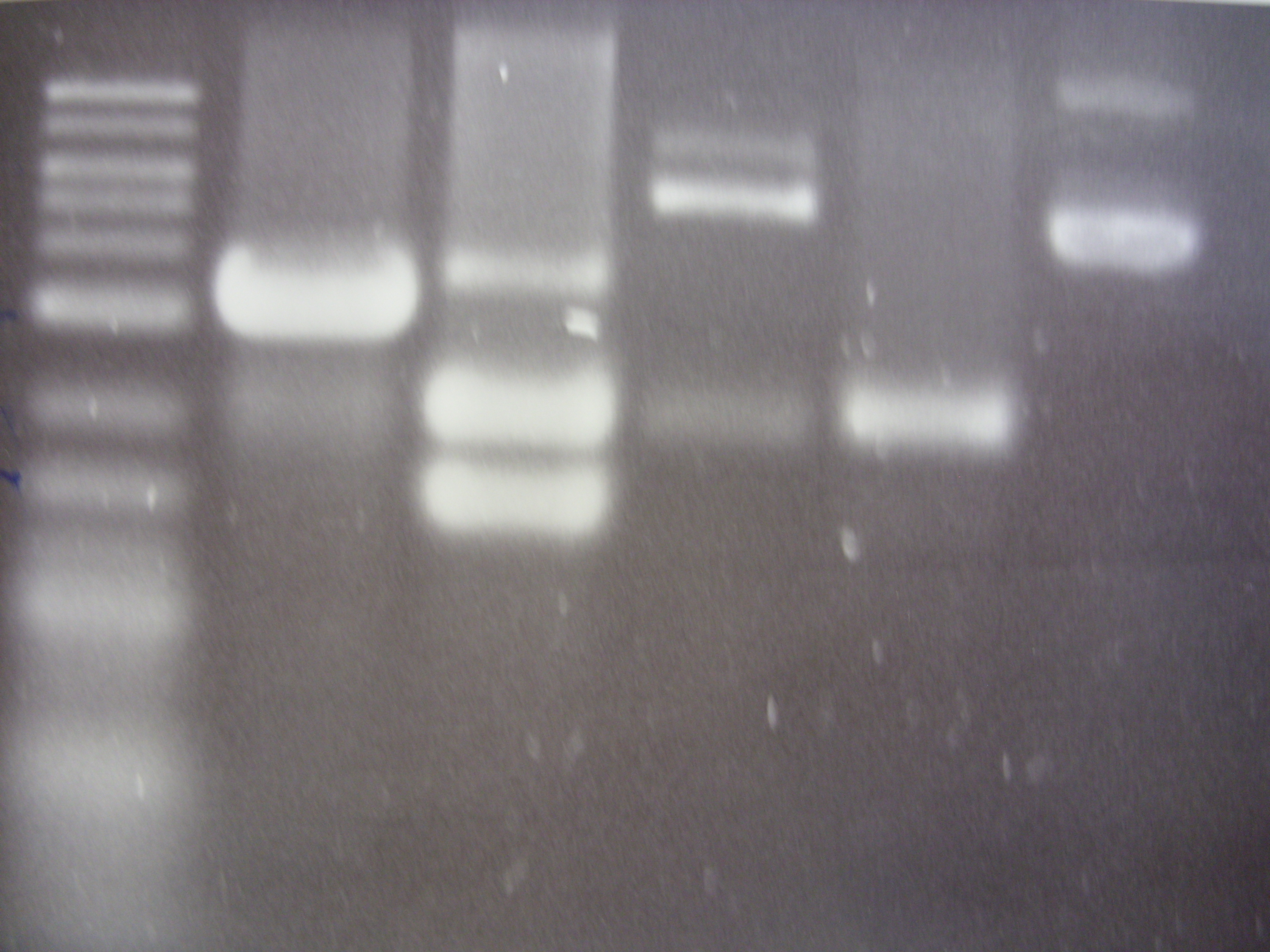
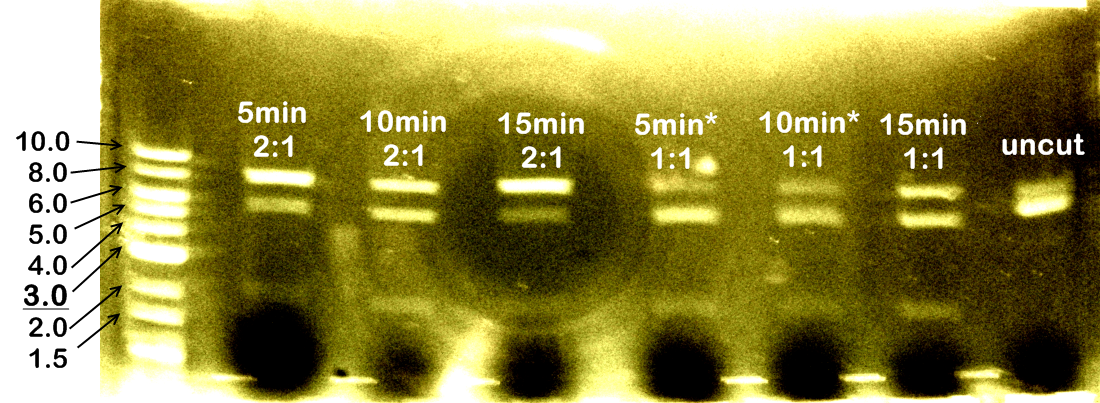
The common bands at 2097bp are the plasmid backbone of pSB1A2. Other than that, each lane (except DNA ladder and negative control) shows at least 2 more bands: the gene insert, and the singly-cut / uncut plasmid. More importantly, the amount of singly-cut/uncut plasmid is significant, indicating that 5 minutes are probably insufficient for FastDigest. We then run restriction digest experiments at 5 minutes, 10 minutes, and 15 minutes, each with either equal amounts of XbaI and PstI, or 2:1 ratio of PstI to XbaI (this ratio is recommended on website of Fermentas for double digestion using PstI and XbaI). Restriction products will be run on agarose gel for analysis tomorrow. | The common bands at 2097bp are the plasmid backbone of pSB1A2. Other than that, each lane (except DNA ladder and negative control) shows at least 2 more bands: the gene insert, and the singly-cut / uncut plasmid. More importantly, the amount of singly-cut/uncut plasmid is significant, indicating that 5 minutes are probably insufficient for FastDigest. We then run restriction digest experiments at 5 minutes, 10 minutes, and 15 minutes, each with either equal amounts of XbaI and PstI, or 2:1 ratio of PstI to XbaI (this ratio is recommended on website of Fermentas for double digestion using PstI and XbaI). Restriction products will be run on agarose gel for analysis tomorrow. | ||
Revision as of 13:26, 5 August 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Week 4 - Development
Monday
Wet Work
Melanin
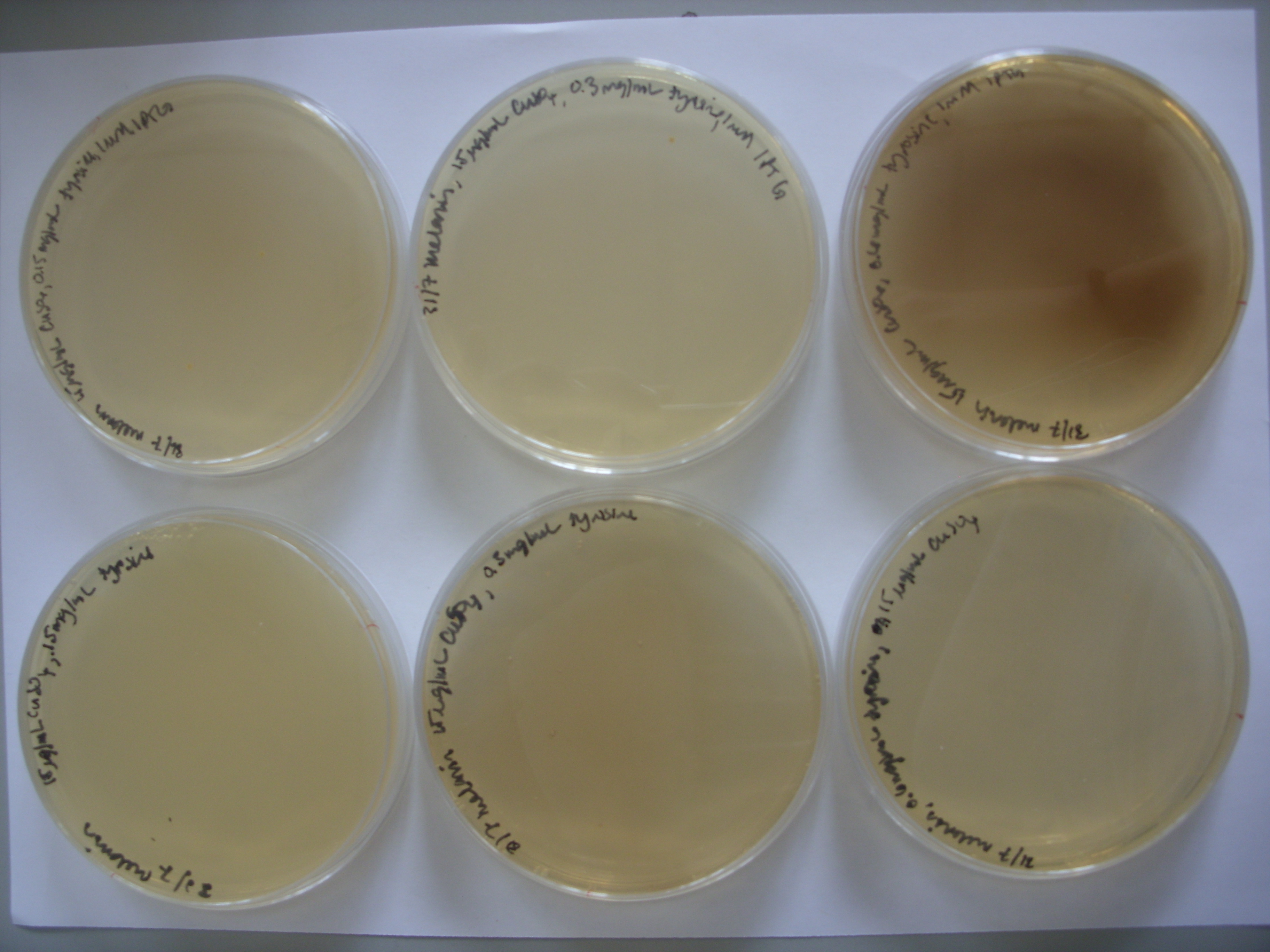
- Results from weekend plates:
From left to right, top row: 1mM IPTG with 0.075 mg/mL tyrosine, 1mM IPTG and 0.3 mg/mL tyrosine, and then 1mM IPTG and 0.6 mg/mL tyrosine From left to right, bottom row: 0.075 mg/mL tyrosine, 0.3 mg/mL tyrosine, and then 0.6 mg/mL tyrosine.
It appears that the bacteria were much too high a concentration; they formed lawns. As we saw on the first plates, pigment production is reduced at high bacterial concentrations. However, it is encouraging that the darkest plate is the one with the greatest tyrosine concentration and IPTG.
To follow up, the experiment was repeated with 1/100 and 1/1000 dilutions of bacterial culture on the following plates:
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.6 mg/mL tyrosine
100ug/ml Ampicillin, 7.5 ug/ml CuSO4, 0.075 mg/mL tyrosine, 0.5mM IPTG
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine, 1mM IPTG
100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.6 mg/mL tyrosine, 1mM IPTG
- Unfortunately, the overnight cultures for the plate readers were contaminated, so a single colony was once again incubated overnight in 10 mL of 100ug/ml Ampicillin, 15 ug/ml CuSO4, 0.3 mg/mL tyrosine, 1mM IPTG in preparation for pigment characterisation using the plate reader.
Violacein
As described in the resource cited below, violacein can be extracted using butanol. A single colony was incubated overnight in LB with trimethoprim. We hope to achieve pigment production in liquid cultures and then extract pigment later in the week.
P.R. August, T.H. Grossman, C. Minor, M.P. Draper, I.A. MacNeil, J.M. Pemberton, K.M. Call, d. Holt, and M. S. Osbourne, Sequence Analysis and Functional Characterization of the Violacein Biosynthetic Pathway from Chromobacterium violaceum, J. Mol. Microbiol. Biotechnol. (2000) 2(4): 513-519. [http://www.horizonpress.com/jmmb/v2/v2n4/26.pdf]
Amplification
- Chose 4 activator / promoter combinations to continue work with:
- P2 activator with Psid promoter (I746374)
- P2 activator with PLL promoter (I746375)
- PSP3 activator with PF promoter (I746380)
- phiR73 with PO promoter (I746391)
- Of the above, on plates all showed leaky expression of GFP and RFP, suggesting that last weeks transformation was successful.
- In preparation for a plate reader assay tomorrow, incubated single colonies overnight of the above constructions as well as the following controls
- I3540 (pBad and GFP) - taken from 2007 glycerol stocks
- I3620 (pBad and RFP) - taken from 2007 glycerol stocks
- untransformed Arabinose strain colony
- Plate reader assay will have triple replicates of all of the above constructs induced with arabinose at various concentrations.
Progress on carotenoids (Monday)
We attempt to start assembling individual Biobricks (K118014/CrtE, K118006/CrtB and K118014/CrtI) into a composite construct (CrtEBI), which is expected to produce reddish/pink lycopene.
We have access to Fermentas FastDigest enzymes. XbaI, SpeI and PstI were used in pair to carry out double digest. In our preliminary trial, the following combinations were used:
- SpeI and PstI on K118006/CrtB
- XbaI and PstI on K118014/CrtI
- XbaI and PstI on K152005/CrtEBIY
- SpeI and PstI on R0011/Constitutive Promoter
First lane on the left was loaded with 2-log DNA ladder. Last lane on the right was loaded with undigested K152005/CrtEBIY as negative control.
In each case, 1 ug of DNA sample was used, with 1 ul of each enzymes, 2 ul of 10X FastDigest buffer, and appropriate amount of water to make up 20 ul final volume. The protocol suggested keeping at 37 oC for 5 minutes for fast digestion, then heat inactivation at 80 oC for 20 minutes to stop enzymatic reaction.
Restriction digestion products were loaded into agarose gel with GelGreen stain and run at 80 V (constant) for 45 minutes.
Dry Work
Promoter response to input concentration
- The pBAD promoter currently used in the amplification system is sensitive to different concentrations of arabinose, although this effect could be considered limited, so this is probably not the best promoter to use to get a sequence of outputs at different concentrations [].
Codon Optimisation for B.Subtilis and E.Coli
A comparison table for codon usage in the two species was made. For each amino acid, a codon was classed as Low, Medium or High use depending on the relative occurence in the genome. The intention is to create an optimised table for both species. A more rigorous and possibly more generally useful approach is under development.
BioBricks
The MelA gene and natural rbs was added to the registry as a BioBrick part. This part will be constructed, ligated into a vector, and sent to the registry as soon as the primers arrive.
Tuesday
Wet Work
Amplification
Transformed the following biobricks into the arabinose strain bacteria, plated overnight
- I746313 - pBad with activator P2 ogr and mRFP (kan resistance, low copy plasmid)
- I746314- pBad with activator PSP3 pag and mRFP (kan resistance, low copy plasmid)
- I746315- pBad with activator phiR73 delta and mRFP (kan resistance, low copy plasmid)
- I746320- PF promoter followed by GFP (high copy plasmid)
- I746321- PO promoter followed by GFP (high copy plasmid)
- I746324- Psid promoter followed by GFP (high copy plasmid)
- I746325- PLL promoter followed by GFP (high copy plasmid)
- I746370 - (failed over the weekend) P2 ogr inducing PF promoter
Also overnight, ran the plate reader to take fluorescence readings for GFP and RFP as well as OD readings of the following cultures induced with arabinose at 10mM, 1mM, 100 uM, 10 uM, and 1uM (as well as no arabinose)
- I746374
- I746375
- I746380
- I746391
Progress on carotenoid (Tuesday)
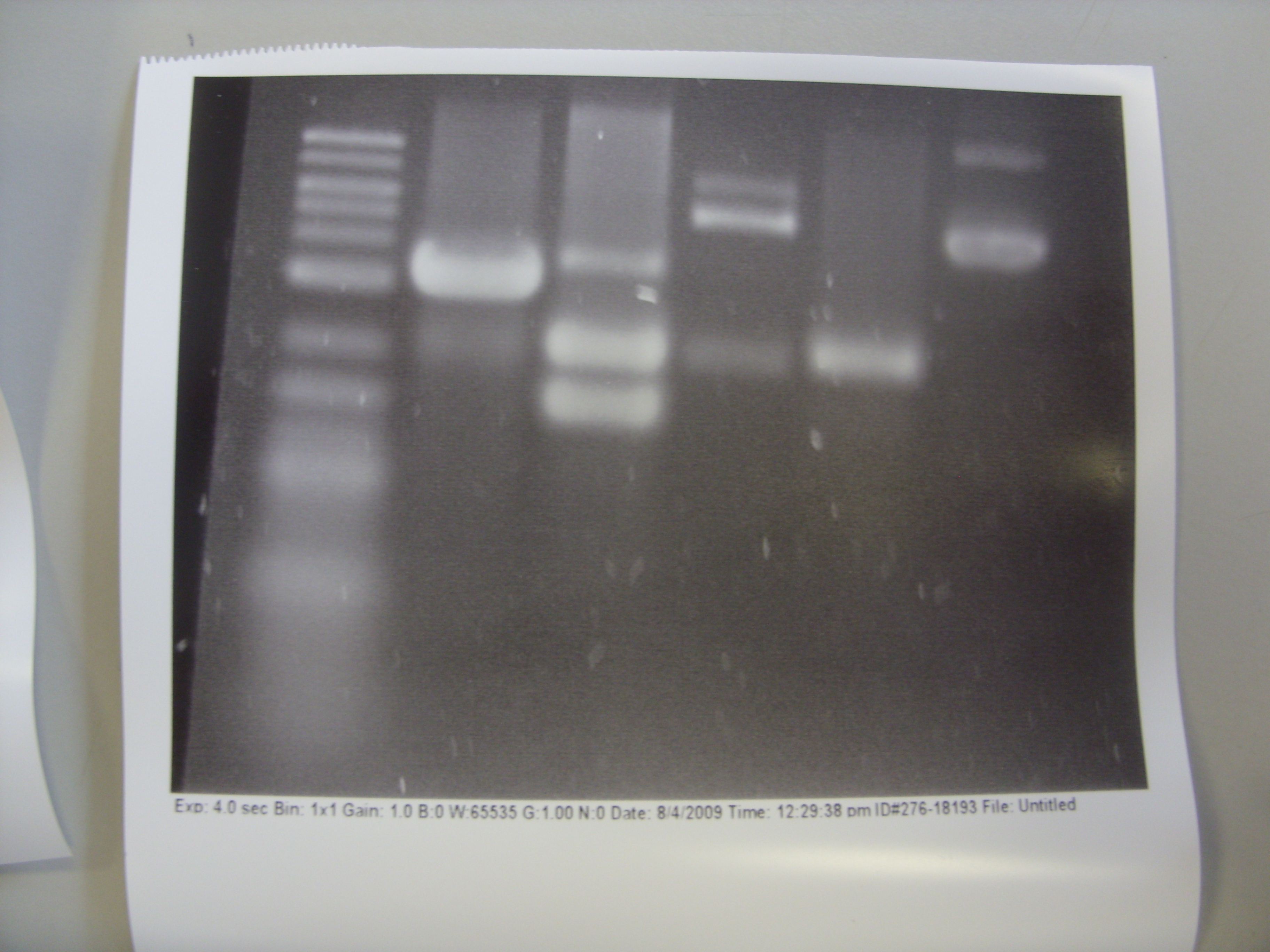
The restriction digest products of yesterday were run on 1% agarose gel with GelGreen stains for 45 minutes. 1 ug of DNA were used for each sample, together with ladder and uncut plasmid (as negative control). However, no bands were visible under UV or Blue light illumination.
To find out the cause of the problem, we put the agarose gel in EtBr solution for 1 hour staining (post-gel staining). The bands then showed up under UV illumination. This suggested that the problem might reside in GelGreen. We might have used the wrong illumination frequency, or GelGreen added was insufficient (protocol recommended 1 ul GelGreen for every 1 ml of agarose gel). Consequently, we will run an agarose gel tomorrow and then do post-gel staining with 3X GelGreen to check if the problem persists.
Results of restriction digest are shown below (refer to yesterday's notebook entry for detailed description of restriction condition):
- PHOTO****************
The common bands at 2097bp are the plasmid backbone of pSB1A2. Other than that, each lane (except DNA ladder and negative control) shows at least 2 more bands: the gene insert, and the singly-cut / uncut plasmid. More importantly, the amount of singly-cut/uncut plasmid is significant, indicating that 5 minutes are probably insufficient for FastDigest. We then run restriction digest experiments at 5 minutes, 10 minutes, and 15 minutes, each with either equal amounts of XbaI and PstI, or 2:1 ratio of PstI to XbaI (this ratio is recommended on website of Fermentas for double digestion using PstI and XbaI). Restriction products will be run on agarose gel for analysis tomorrow.
To make sure that transformation colonies growing on Week 1 plates (Amp) are still viable and contain our plasmid insert, 8 colonies were picked from each Week 1 plate and transfered to freshly made Amp plates. They will be ready for mini-prep on Thursday.
Dry Work
Melanin
Received four of the six primers required to make melA into a BioBrick. The primers recieved were B/C/D and E, the four needed to cut out the restriction sites. A and F were slightly longer plasmids and so should arrive tomorrow.
Violacein
We decided to get the violacein gene synthesised in four sections, as the whole operon is too large to be economically feasible for one synthesis run. Gene designer from DNA2.0 was used to prepare the genes to be sent away for synthesis.
Wednesday
Wet Work
Amplification
All of last nights transformations worked except I746324- Psid promoter followed by GFP (high copy plasmid) - will try transforming it again tonight.
Dry Work
MelA Biobrick
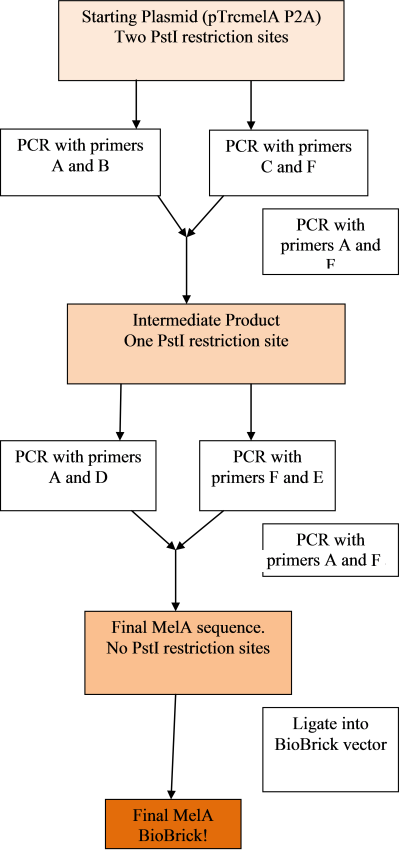
The remaining two plasmids were delivered, so a plan was made for creating the melanin biobrick and removing the PstI sites:
The PCR rounds are designed to cut the melA gene away from it's current surrounding plasmid, and change the two PstI restriction sites from CTGGAC to CTGCAA. This should not affect the coding ability of the gene. The primers will also attach the BioBrick prefix and suffix to the gene, allowing it to be easily ligated into the vector.
 "
"