Team:Cambridge/Notebook/Week2
From 2009.igem.org
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
The Project
Monday 20 July
Finalized our project into a few objectives
- Pigment production
- Transform E. coli to produce pigment
- Hook up the pigmentation genes to inducible promoters
- Shutter mechanism
- build, test a shutter mechanism
- Hook up pigmentation genes under inducible promoters to the shutter mechanism
- Improve on previous iGEM projects
- arsenic sensor?
- bacterial photography?
- Modelling
- shutter mechanism
- Growth rate regulation
Goals for Wet Work
- Inducible pigment production integrated with a shutter mechanism
- Possibly attach the arsenic bio-sensor to our green/red pigment output
Tuesday 21 July
Proceedure to make competant cells started
- One colony of TOP10 E. coli added to 50ml of LB broth. Incubated overnight in the shaking incubator
Planned and prepared for transformation
- Media made up
- Two litres of 0.1 mM HEPES
- One litre of LB broth (for making competant cells)
- 100ml of 10% glycerol
- Ampicillin made up with water to make 8 x 1ml aliquots of 100mg/ml
- 15 LB agar and 100ug/ml Ampicillin plates made up
Dry work
Plan for tomorrow
- prepare trimethoprim stock solution
- make LB agar and trimethoprim plates as well as LB agar and copper + tyrosine plates
- Complete proceedure for making competant cells (transfer bacteria into big flask, wash with HEPES, and aliquot into glycerol)
- Attempt transformations by electroporation
- pPSX plasmid (violet)
- melA plasmid (brown)
- biobricks
Wednesday 22 July
competent cell procedure
- completed (see protocol page for details of the procedure)
planned and prepared for electroporation tomorrow
- made up 4 LB agar and 100ug/ml Ampicillin, 0.5ug/mL tyrosine, 15 ug/mL CuSO4 plates
- plan to transform TOP10 competent bacteria with
- melanin plasmid, pTRCmelA plasmid --plate onto tyrosine/copper/ampicillin plate
- violacein plasmid,Plasmid pPSX-Vio+ -- plate onto trimethoprim plate
- orange biobrick, BBa_K152005 -- plate onto ampicillin plate
- promoter, R0011 -- plate onto ampicillin plate
- more?
- need to make trimethoprim stock solution 10mg/mL acetone, and plate at 50ug/mL
- tried to make the stock solution today, couldn't get the solid trimethoprim to dissolve in acetone
- need to work out exact volumes of plasmid to be added to the cell (concentrations written on the side of the bottles)
Thursday 23 July
Electroporation
- transformed Top10 competent cells, incubated the following plates:
- Top10 + pTRCmelA plasmid on tyrosine/copper/ampicillin plate
- Top10 + Plasmid pPSX-Vio+ on trimethoprim plate
- Top10 + BBa_K152005 on ampicillin plate
- Top 10 + R0011 on ampicillin plate
- 1:10 dilutions of each were also plated
- All plates were stored overnight in the 37 degree incubator
Making BioBricks
With the aim of making a brown-pigmented BioBrick, primers were constructed to cut out the MelA gene from the plasmid, add the prefix and suffix, and remove the two forbidden restriction sites. This would then be pasted into one of the recommended vectors and could be submitted to the registry.
Input and Processing systems
We split into groups to plan and discuss the following possible ideas for input systems:
- Mike and Shuna = Amplifier
- Siming and Alan = Bistable switch
- Crispian and Megan = Population control and growth
- Viv = Coloured spores
Plans for Friday
Meeting at 10am to discuss ideas Meeting with faculty at 2.30pm to further discuss ideas and present work done so far
Wet work = colony PCR (to be explained by James) and observation of (hopefully!) coloured plates
Friday 24 July
Used ApE to examine the melanin plasmid:
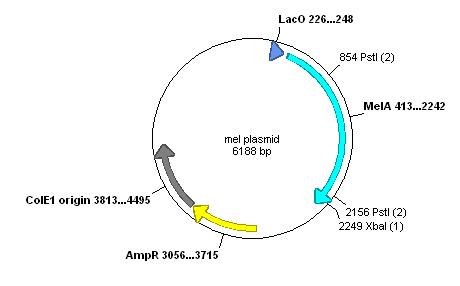
This contains only two forbidden restriction sites within the actual gene. These could be removed by PCR.
Also looked at the vio operon:
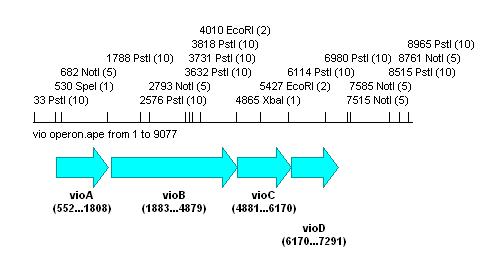
As this contains a large number of the forbidden biobrick restriction sites, it would be impossible to prepare the biobrick using PCR alone. The operon would have to be synthesised with these sites removed to be feasible as a biobrick.
 "
"