Team:Cambridge/Project/Violacein
From 2009.igem.org
(→Construct composite Biobrick X) |
(→Violacein Pigment) |
||
| Line 6: | Line 6: | ||
{{Template:Cambridgetemplatetop}} | {{Template:Cambridgetemplatetop}} | ||
[[#Introduction | Introduction ]] | [[#Introduction | Introduction ]] | ||
| - | [[# | ]] | + | [[#Previous Work | Previous Work]] |
| - | [[# | ]] | + | [[#1. Test for Violacein Pigment production | Violacein Pigment Production ]] |
| - | [[# | ]] | + | [[#2. Biobrick Construction | Biobrick Construction]] |
[[# | ]] | [[# | ]] | ||
{{Template:Cambridgetemplatebottom}} | {{Template:Cambridgetemplatebottom}} | ||
Revision as of 14:22, 4 August 2009
Categories :
Project :
-
Overview
Sensitivity Tuner
--- Characterisation
--- Modelling
Colour Generators
--- Carotenoids (Orange/Red)
--- Melanin (Brown)
--- Violacein (Purple/Green)
The Future
Safety
Notebook :
Team Logistics :
Violacein Pigment
Introduction
Violacein Biosynthesis
The Violacein pigment is produced from L-tryptophan via a pathway involving four enzymes, VioA-D. This scheme is shown below:
From P.R. August, T.H. Grossman, C. Minor, M.P. Draper, I.A. MacNeil, J.M. Pemberton, K.M. Call, d. Holt, and M. S. Osbourne, Sequence Analysis and Functional Characterization of the Violacein Biosynthetic Pathway from Chromobacterium violaceum, J. Mol. Microbiol. Biotechnol. (2000) 2(4): 513-519. [1]
Further, as module 5 is Aqua, controlling VioA, VioB, VioC, and VioD allows us to generate two different pigments, aqua and violet.
Vio Operon
Our VioA-D genes are from Chromobacterium voilaceum.
Previous Work
Duncan Rowe provided us with the VioA-D operon on the pPSX-Vio+ plasmid, a very low copy number plasmid. We transformed Top10 E. coli with pPSX-Vio+ and made our own plasmid stocks.
Action plan of our team
Our action plan is as follows:
1. Test for violacein pigment production
2. Synthesize the violacein operon without any forbidden restriction sites to make it bio-brick compatible, and bio-brick the individual genes of the operon.
3. Produce aqua pigment.
1. Test for Violacein Pigment production
Proof of pigment production
Successful Pigment Production
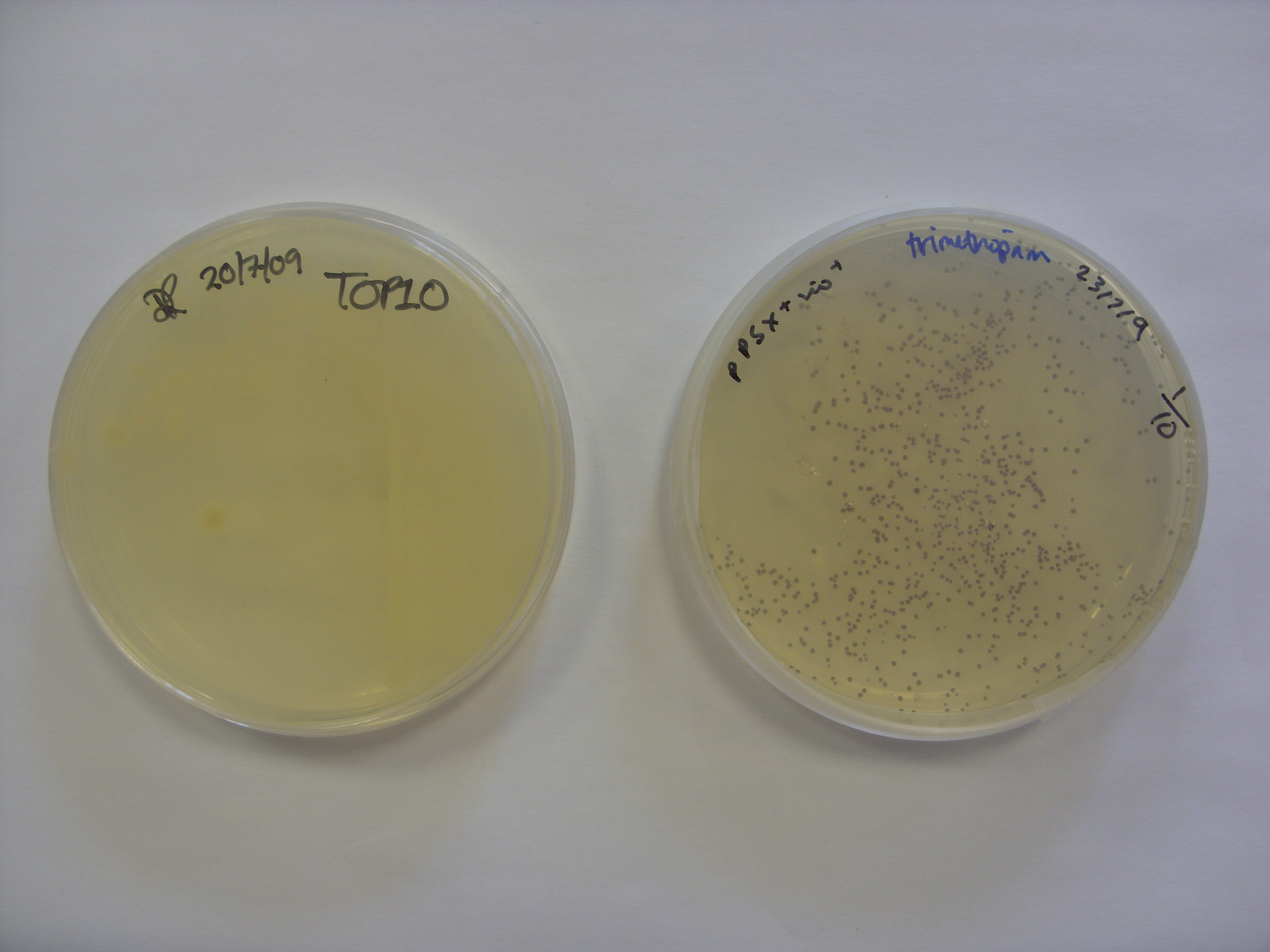
We transformed Top10 with pPSX-Vio+. After three colour eventually appeared, as shown below. Interestingly, the pigment appears to remain within the bacteria, with little or no bleeding into the media. We took the violacein pigment bacteria (right plate in photo) out of the fridge to find that the purple colour had started to develop. They were therefore left at room temperature overnight. The colour appears to be within the bacteria, with little or no bleeding into the media. The control plate (left plate) is the untransformed TOP10 E. coli.
Left: control plate - untransformed TOP10 E. coli, Right: Top10 transformed with pPSX-Vio+.
Pigment production efficienty
The Vio operon is currently on a very low copy number plasmid; moving it onto a higher copy number plasmid may accelerate pigment production.
Violacein Pigment Characterization
2. Biobrick Construction
The Vio operon had numerous forbidden restriction sites. We thus had to synthesize it, removing these restriction sites and optimizing codon usage for E. coli.
 "
"