Team:PKU Beijing/Project/AND Gate 1/Inducible System Result
From 2009.igem.org
(→IPTG Sensor) |
(→IPTG Sensor) |
||
| Line 39: | Line 39: | ||
The result is similar to the tetR system. The colonies which contain the IPTG sensor - GFP plasmid expressed GFP even without induction, and the GFP flourescence didn't increase even after IPTG induction. The evidence that this construct is leaky without induction is on Fig2, and the data after induction are not showed here. | The result is similar to the tetR system. The colonies which contain the IPTG sensor - GFP plasmid expressed GFP even without induction, and the GFP flourescence didn't increase even after IPTG induction. The evidence that this construct is leaky without induction is on Fig2, and the data after induction are not showed here. | ||
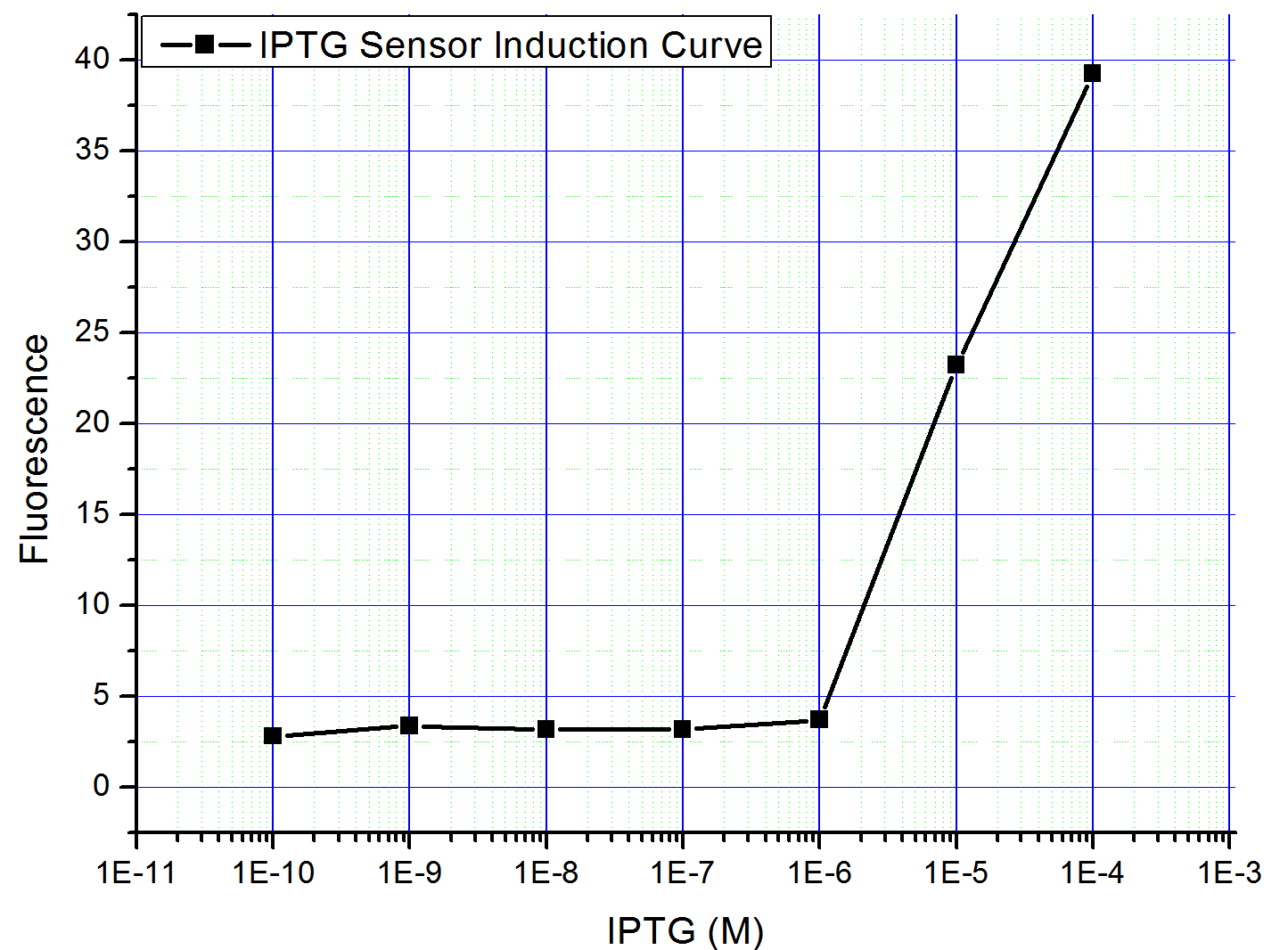
| - | Nevertheless, we found another way to bypass the problem | + | Nevertheless, we found another way to bypass the problem: take advantage of the lacIq on the F plasmid of ''E. coli'' strain [http://ecoliwiki.net/colipedia/index.php/JM109 JM109]. Since the F plasmid is one copy per cell, pLac (<partinfo>BBa_R0010</partinfo>) should be on a low copy plasmid, such as pSB4K5. The pLac can be repressed by endogenous lacI even without its represssor coexpressed on the same plasmid. Thus we constructed the simplified system (Part: <partinfo>K228821</partinfo>) and induced it by different concentration of IPTG. The result is shown in Fig3, judging from the induction curve, pLac promoter may be qualified in constructing the AND Gate. |
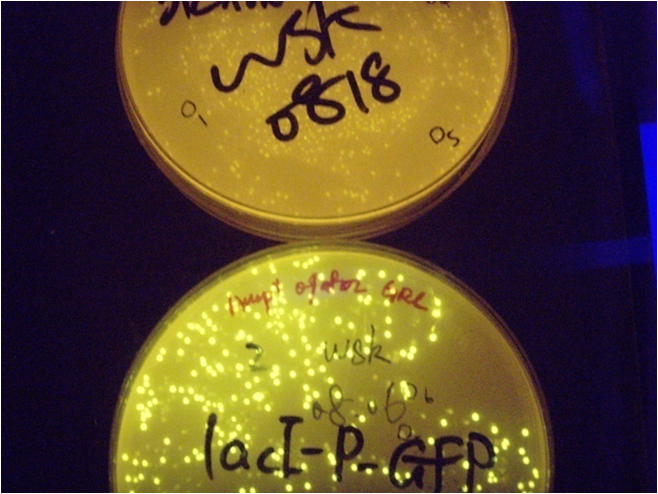
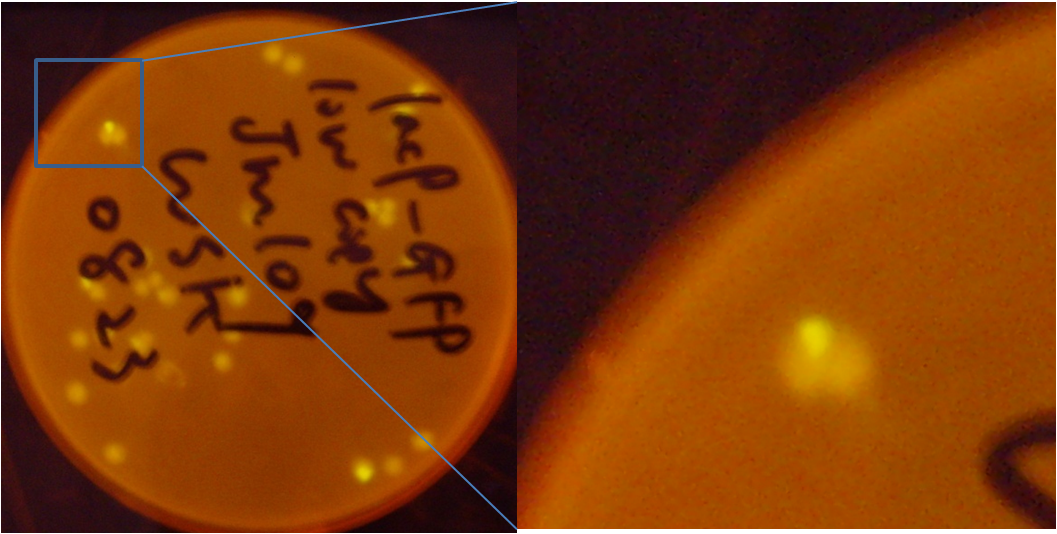
[[Image:PKU_lac_0.png|300px|left|thumb|Fig2. the result of pLac promoter system. The colonies are green under blue light, even without induction, the upper plate is for comparison.]] | [[Image:PKU_lac_0.png|300px|left|thumb|Fig2. the result of pLac promoter system. The colonies are green under blue light, even without induction, the upper plate is for comparison.]] | ||
| Line 45: | Line 45: | ||
[[Image:PKU_Lac_1.png|300px|left|thumb|Fig3. lac promoter induction curve.]] | [[Image:PKU_Lac_1.png|300px|left|thumb|Fig3. lac promoter induction curve.]] | ||
| - | However, there is still another problem in this simplified system: the loss of F plasmid of JM109 may lead to activation of promoter pLac without induction. From the plate (without induction), we found that part( | + | |
| + | |||
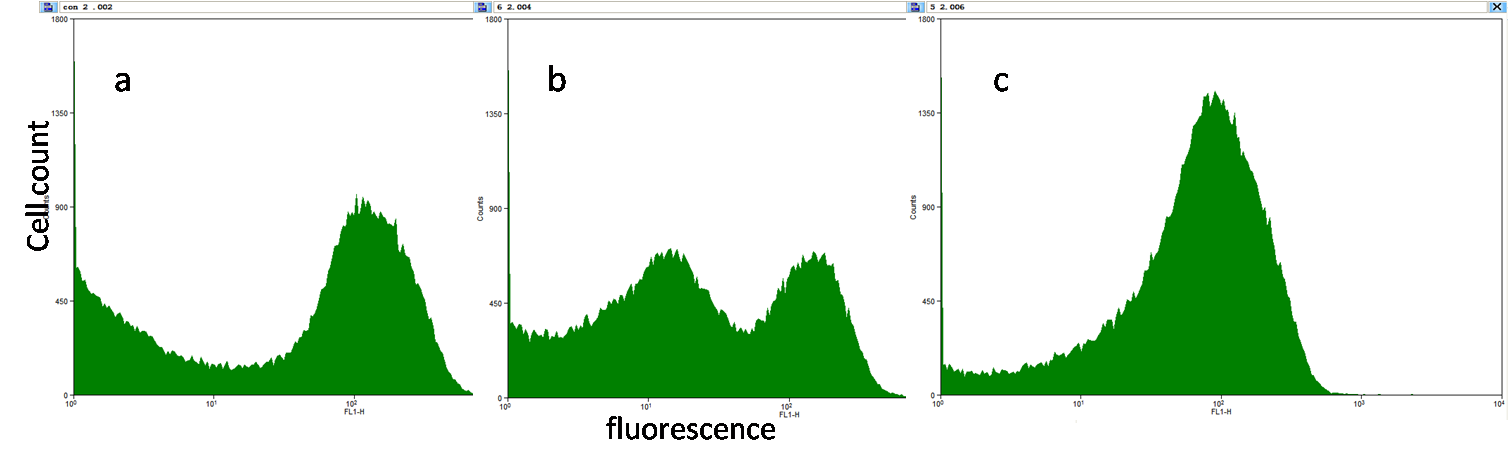
| + | However, there is still another problem in this simplified system: the loss of F plasmid of JM109 may lead to activation of promoter pLac without induction. From the plate (without induction), we found that part(especially the central part) of a singal colony turned green, while they are supposed to stop express GFP in the presence of lacIq. A stronger evidence is the data from flowcytometry: The strength of GFP fluorescence obviously shows double peaks. All these suggest that some of the cell retains the F plasmid while the the others lost it. | ||
[[Image:PKU_lac_1.png|500px|center|thumb|Fig4. the JM109 colonies that contain low copy pLac-GFP plasmid. Center of the colonies turns green, while in the other area GFP expression is suppressed. It suggests that some E.coli lost their F plasmids]] | [[Image:PKU_lac_1.png|500px|center|thumb|Fig4. the JM109 colonies that contain low copy pLac-GFP plasmid. Center of the colonies turns green, while in the other area GFP expression is suppressed. It suggests that some E.coli lost their F plasmids]] | ||
| Line 51: | Line 53: | ||
[[Image:PKU_lac_2.png|600px|center|thumb|Fig5a, b & c. The fluorescence was measured by flowcytometry to see the fluorescence of each cell in a population. <br>Fig5a illustrates that there are leaky cells and normal cells without induction. Fig4b shows the data from the cells induced by 10^-6M IPTG, and Fig4c show those induced by 10^-5M IPTG. Double peaks can be observed on both 4a and 4b, which means there are two groups of E.coli, but as the induction saturates, two peaks merge into one peak.]] | [[Image:PKU_lac_2.png|600px|center|thumb|Fig5a, b & c. The fluorescence was measured by flowcytometry to see the fluorescence of each cell in a population. <br>Fig5a illustrates that there are leaky cells and normal cells without induction. Fig4b shows the data from the cells induced by 10^-6M IPTG, and Fig4c show those induced by 10^-5M IPTG. Double peaks can be observed on both 4a and 4b, which means there are two groups of E.coli, but as the induction saturates, two peaks merge into one peak.]] | ||
| - | In a word, the | + | In a word, the pLac promoter is not a perfect promoter to construct AND Gate. However, if a more stable source of lacI can be supplied, we believe it would be better. |
==='''HSL Sensor'''=== | ==='''HSL Sensor'''=== | ||
Revision as of 10:17, 20 October 2009
|
|||||||||||||
|
|||||||||||||
 "
"