User:DavidC/6 October 2009
From 2009.igem.org
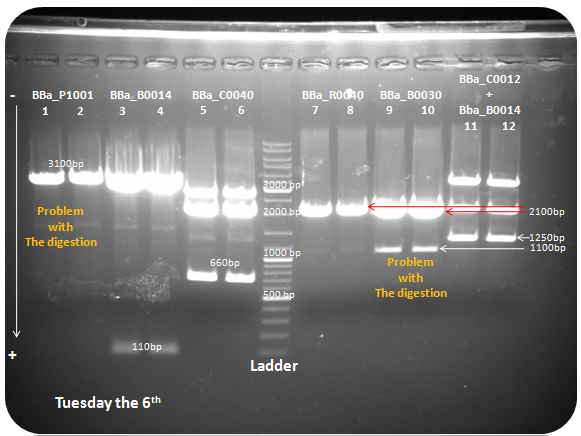
Tuesday the 6th
DNA extraction
Miniprep extraction (promega) of:
BBa_B0014;
BBa_B0030;
BBa_P1001;
BBa_C0040;
BBa_R0040.
Restriction digest
Ligation between BBa_B0014 and BBa_P1001:
Restriction digest of BBa_B0014 by PstI and SpeI (3284pb):
DNA (miniprep) = 30µL
Buffer (NEB) = 5µL
H20 = 13µL
PstI (NEB) = 1µL
Spe I (NEB) = 1µL
1 hour of incubation at 37°C.
Restriction digest of BBa_P1001 by PstI and XbaI (1279bp):
DNA (miniprep) = 30µL
Buffer M (TAKARA) = 4µL
H20 = 4µL
Eco RI (TAKARA) = 1µL
Spe I (TAKARA) = 1µL
1 hour of incubation at 37°C.
Ligation between BBa_C0012 + BBa_B0014 and BBa_B0030
Restriction digest of BBa_C0012 + BBa_B0014 by EcoRI and SpeI (1223bp):
DNA (miniprep) = 30µL
Buffer (NEB) = 5µL
H20 = 13µL
PstI (NEB) = 1µL
Spe I (NEB) = 1µL
1 hour of incubation at 37°C.
Restriction digest of BBa_B0030 by EcoRI and XbaI (2094bp):
DNA (miniprep) = 30µL
Buffer M (TAKARA) = 4µL
H20 = 4µL
Eco RI (TAKARA) = 1µL
Spe I (TAKARA) = 1µL
1 hour of incubation at 37°C.
Restriction digest of BBa_C0040 by Eco RI and Spe I (660bp)
DNA (miniprep) = 30µL
Buffer M (TAKARA) = 4µL
H20 = 4µL
Eco RI (TAKARA) = 1µL
Spe I (TAKARA) = 1µL
1 hour of incubation at 37°C.
Restriction of BBa_R0040 by Eco RI and Xba I (2133bp)
DNA (miniprep) = 30µL
Buffer M (TAKARA) = 4µL
H20 = 4µL
Eco RI (TAKARA) = 1µL
Xba I (TAKARA) = 1µL
1 hour of incubation at 37°C.
DNA electrophoresis
85 Volt, 15 minutes.
105 Volt, 40 minutes.
Ladder fermentas 1 Kb.
Samples: BBa_P1001, BBa_B0014, BBa_C0040, BBa_R0040, BBa_B0030, BBa_C0012 + BBa_B0014.
DNA purification
Kit Qiagen “gel extraction kit”, final volume = 50µL.
Ligation
Ligation of BBa_B0014 with BBa_P1001 (TAKARA, DNA ligation kit) :
First report:
Plasmid (P1001) = 1µL
Insert (B0014) = 7µL
Solution A = 1µL
Solution B = 1µL
Second report:
Plasmid (P1001) = 0,5µL
Insert (B0014) = 4µL
Solution A = 4,5µL
Solution B = 1µL
Ligation between BBa_C0012 + BBa_B0014 and BBa_B0030 (TAKARA, DNA ligation kit) :
First report:
Plasmid (B0030) = 1µL
Insert (C0012 + B0014) = 7µL
Solution A = 1µL
Solution B = 1µL
Second report:
Plasmid (B0030) = 0,5µL
Insert (C0012 + B0014) = 4µL
Solution A = 4,5µL
Solution B = 1µL
Electroporation
Electroporation cuvettes = 2mm ; inoculums of electrocompetent E.coli DH5alpha= 40µL; pulse = 2,5KVolt ; 1h of incubation.
Spread 1mL of inoculums into a petri dish with LB + ampicillin (50mg/mL) (20/0,02mL).
 "
"