From 2009.igem.org



| August
|
| M | T | W | T | F | S | S
|
|
|
|
|
|
| [http://2009.igem.org/User:DavidC/1_August_2009 1]
| [http://2009.igem.org/User:DavidC/2_August_2009 2]
|
| [http://2009.igem.org/User:DavidC/3_August_2009 3]
| [http://2009.igem.org/User:DavidC/4_August_2009 4]
| [http://2009.igem.org/User:DavidC/5_August_2009 5]
| [http://2009.igem.org/User:DavidC/6_August_2009 6]
| [http://2009.igem.org/User:DavidC/7_August_2009 7]
| [http://2009.igem.org/User:DavidC/8_August_2009 8]
| [http://2009.igem.org/User:DavidC/9_August_2009 9]
|
| [http://2009.igem.org/User:DavidC/10_August_2009 10]
| [http://2009.igem.org/User:DavidC/11_August_2009 11]
| [http://2009.igem.org/User:DavidC/12_August_2009 12]
| [http://2009.igem.org/User:DavidC/13_August_2009 13]
| [http://2009.igem.org/User:DavidC/14_August_2009 14]
| [http://2009.igem.org/User:DavidC/15_August_2009 15]
| [http://2009.igem.org/User:DavidC/16_August_2009 16]
|
| [http://2009.igem.org/User:DavidC/17_August_2009 17]
| [http://2009.igem.org/User:DavidC/18_August_2009 18]
| [http://2009.igem.org/User:DavidC/19_August_2009 19]
| [http://2009.igem.org/User:DavidC/20_August_2009 20]
| [http://2009.igem.org/User:DavidC/21_August_2009 21]
| [http://2009.igem.org/User:DavidC/22_August_2009 22]
| [http://2009.igem.org/User:DavidC/23_August_2009 23]
|
| [http://2009.igem.org/User:DavidC/24_August_2009 24]
| [http://2009.igem.org/User:DavidC/25_August_2009 25]
| [http://2009.igem.org/User:DavidC/26_August_2009 26]
| [http://2009.igem.org/User:DavidC/27_August_2009 27]
| [http://2009.igem.org/User:DavidC/28_August_2009 28]
| [http://2009.igem.org/User:DavidC/29_August_2009 29]
| [http://2009.igem.org/User:DavidC/30_August_2009 30]
|
| [http://2009.igem.org/User:DavidC/31_August_2009 31]
|
|
| September
|
| M | T | W | T | F | S | S
|
|
| [http://2009.igem.org/User:DavidC/1_September_2009 1]
| [http://2009.igem.org/User:DavidC/2_September_2009 2]
| [http://2009.igem.org/User:DavidC/3_September_2009 3]
| [http://2009.igem.org/User:DavidC/4_September_2009 4]
| [http://2009.igem.org/User:DavidC/5_September_2009 5]
| [http://2009.igem.org/User:DavidC/6_September_2009 6]
|
| [http://2009.igem.org/User:DavidC/7_September_2009 7]
| [http://2009.igem.org/User:DavidC/8_September_2009 8]
| [http://2009.igem.org/User:DavidC/9_September_2009 9]
| [http://2009.igem.org/User:DavidC/10_September_2009 10]
| [http://2009.igem.org/User:DavidC/11_September_2009 11]
| [http://2009.igem.org/User:DavidC/12_September_2009 12]
| [http://2009.igem.org/User:DavidC/13_September_2009 13]
|
| [http://2009.igem.org/User:DavidC/14_September_2009 14]
| [http://2009.igem.org/User:DavidC/15_September_2009 15]
| [http://2009.igem.org/User:DavidC/16_September_2009 16]
| [http://2009.igem.org/User:DavidC/17_September_2009 17]
| [http://2009.igem.org/User:DavidC/18_September_2009 18]
| [http://2009.igem.org/User:DavidC/19_September_2009 19]
| [http://2009.igem.org/User:DavidC/20_September_2009 20]
|
| [http://2009.igem.org/User:DavidC/21_September_2009 21]
| [http://2009.igem.org/User:DavidC/22_September_2009 22]
| [http://2009.igem.org/User:DavidC/23_September_2009 23]
| [http://2009.igem.org/User:DavidC/24_September_2009 24]
| [http://2009.igem.org/User:DavidC/25_September_2009 25]
| [http://2009.igem.org/User:DavidC/26_September_2009 26]
| [http://2009.igem.org/User:DavidC/27_September_2009 27]
|
| [http://2009.igem.org/User:DavidC/28_September_2009 28]
| [http://2009.igem.org/User:DavidC/29_September_2009 29]
| [http://2009.igem.org/User:DavidC/30_September_2009 30]
|
|
| October
|
| M | T | W | T | F | S | S
|
|
|
|
| [http://2009.igem.org/User:DavidC/1_October_2009 1]
| [http://2009.igem.org/User:DavidC/2_October_2009 2]
| [http://2009.igem.org/User:DavidC/3_October_2009 3]
| [http://2009.igem.org/User:DavidC/4_October_2009 4]
|
| [http://2009.igem.org/User:DavidC/5_October_2009 5]
| [http://2009.igem.org/User:DavidC/6_October_2009 6]
| [http://2009.igem.org/User:DavidC/7_October_2009 7]
| [http://2009.igem.org/User:DavidC/8_October_2009 8]
| [http://2009.igem.org/User:DavidC/9_October_2009 9]
| [http://2009.igem.org/User:DavidC/10_October_2009 10]
| [http://2009.igem.org/User:DavidC/11_October_2009 11]
|
| [http://2009.igem.org/User:DavidC/12_October_2009 12]
| [http://2009.igem.org/User:DavidC/13_October_2009 13]
| [http://2009.igem.org/User:DavidC/14_October_2009 14]
| [http://2009.igem.org/User:DavidC/15_October_2009 15]
| [http://2009.igem.org/User:DavidC/16_October_2009 16]
| [http://2009.igem.org/User:DavidC/17_October_2009 17]
| [http://2009.igem.org/User:DavidC/18_October_2009 18]
|
| [http://2009.igem.org/User:DavidC/19_October_2009 19]
| [http://2009.igem.org/User:DavidC/20_October_2009 20]
| [http://2009.igem.org/User:DavidC/21_October_2009 21]
| [http://2009.igem.org/User:DavidC/22_October_2009 22]
| [http://2009.igem.org/User:DavidC/23_October_2009 23]
| [http://2009.igem.org/User:DavidC/24_October_2009 24]
| [http://2009.igem.org/User:DavidC/25_October_2009 25]
|
| [http://2009.igem.org/User:DavidC/26_October_2009 26]
| [http://2009.igem.org/User:DavidC/27_October_2009 27]
| [http://2009.igem.org/User:DavidC/28_October_2009 28]
| [http://2009.igem.org/User:DavidC/29_October_2009 29]
| [http://2009.igem.org/User:DavidC/30_October_2009 30]
| [http://2009.igem.org/User:DavidC/31_October_2009 31]
|
|
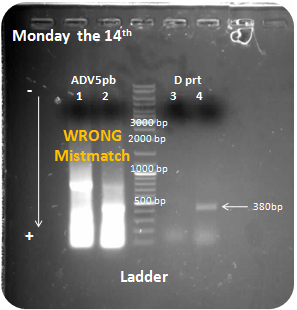
Monday the 14th
Ligation: samples digested on the 13th of September
Ligation pairs: plasmid / insert :
BBa_B0014 / BBa_C0012
First report:
Plasmid = 2µL
Insert = 5µL
Second report:
Plasmid = 1,5µL
Insert = 5µL
Third report:
Plasmide = 1µL
Insert = 7µL
NEB Enzymes
For each samples add sterilized water to obtain a maximum volume equals to 8µL.
Ligation mix (NEB):
1,5µL T4 ligase + 1,5µL T4 ligase buffer.
Add 2µL / tube.
Incubation 1h at RT.
= TAKARA DNA ligation kit
First report:
Solution A (Buffer) = 28µL
Solution B (Enzyme) = 7µL
Incubation 1h at 16°C.
Electroporation
Electroporation cuvettes = 2mm ; inoculums of electrocompetent E.coli DH5alpha= 40µL; pulse = 2,5KVolt ; 1h of incubation.
Spread 1mL of inoculums into a petri dish with LB + ampicillin (50mg/mL) (20/0,02mL).
PCR
D protein
Primers:
Forward : Préfixe ATG BBa
Reverse : Prt D_Rv_Bal1
ADN :
1. Lambda bacteriophage;
2. Purified and amplified D protein.
Cycles:
94°C 2minutes;
(94°C 1 minute;
46°C 1 minutes;
72°C 1 minute(s)) X 35;
72°C 4 minutes;
Penton base from the adenovirus 5
Primers:
Forward : ADV5Fw_Bal1
Reverse : ADV5Rv_Bal1
ADN :
Purified and amplified penton base from plasmid coding for adenovirus 5.
Cycles:
94°C 2minutes;
(94°C 1 minute;
57,3°C 1 minutes;
72°C 2 minute(s)) X 35;
72°C 5 minutes;
DNA electrophoresis
85 Volt, 15 minutes.
105 Volt, 40 minutes.
Ladder fermentas 1 Kb.
Samples: Adenovirus penton base, D protein
Interpretation:
We obtain a small amplification for D protein on the sample 4. But other samples have bad results, we obtain to much DNA for samples 1 and 2 PCR, we also amplified the wrong DNA, maybe we use a bad temperature for primers annealing, finally sample 3 is negative.
 "
"