Team:TUDelft/Modeling Cascade
From 2009.igem.org
(→Modeling the Transcriptional Cascade) |
|||
| (21 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| - | {{Template: | + | {{Template:TUDelftiGEM2009_menu_Model}} |
| - | =Modeling the Transcriptional Cascade= | + | ='''Modeling the Transcriptional Cascade'''= |
| - | + | The modeling of the transcriptional cascade had several objectives: | |
| - | A full description of the Transcriptional Cascade can be found [https://2009.igem.org/Team:TUDelft/Synthetic_Transcriptional_Cascade here]. | + | * to provide the delay team with design guidelines which would maximize the delay time |
| + | |||
| + | * to asses the affect of parameter variation on the delay time | ||
| + | |||
| + | * to determine areas of instability in the parameter space | ||
| + | |||
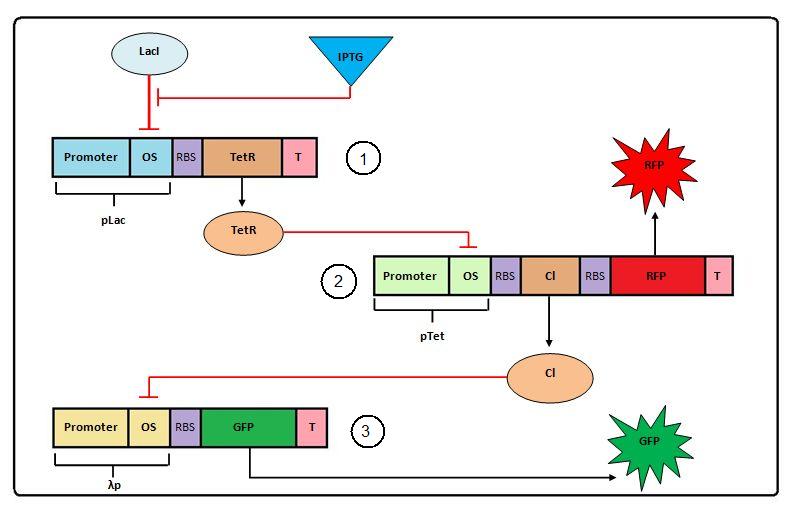
| + | A schematic of the system to be modeled can be seen below. A full description of the Transcriptional Cascade can be found [https://2009.igem.org/Team:TUDelft/Synthetic_Transcriptional_Cascade here]. | ||
| + | |||
| + | [[Image:Negative_Feedforward.jpg|center| <center> Transcriptional cascade assembly and overview </center> | thumb | 550px]]<br> | ||
| + | |||
| + | The system shown above has one input: IPTG and one primary output: GFP. It is characterized by the delay time, which is measured as the time between the induction with IPTG and a certain expression level in the final product (GFP). | ||
==ODEs== | ==ODEs== | ||
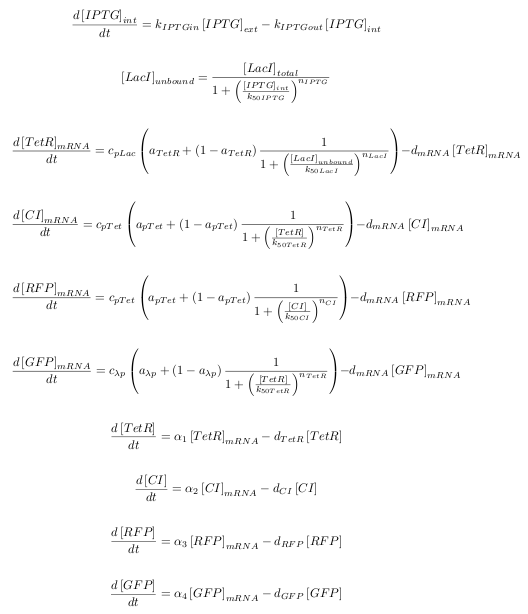
| - | The kinetic equations were written out in a Matlab script for | + | The kinetic equations were written out in a Matlab script. A total of ten equations were used: one for the diffusion of IPTG into the cell, one for the binding of IPTG to LacI, as well as four transcription equations, and four translation equations for the various levels of the cascade. |
[[Image:TUD_eq_cas.png]] | [[Image:TUD_eq_cas.png]] | ||
| + | The notation in this system of equations can be seen in the table below:<br> | ||
| + | <br> | ||
{| border="1" align="center" | {| border="1" align="center" | ||
| <b>Symbol</b> || <b>Definition</b> | | <b>Symbol</b> || <b>Definition</b> | ||
| Line 35: | Line 47: | ||
|- align="left" | |- align="left" | ||
|} | |} | ||
| - | + | <br> | |
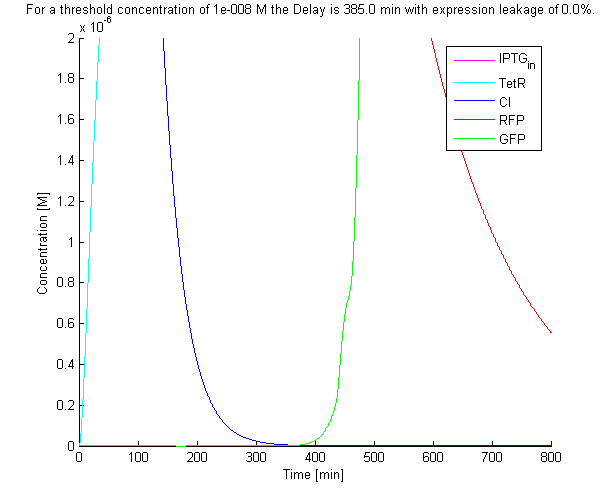
| + | The solution with the [[Team:TUDelft/Modeling_Parameters|default parameters]] of the system of ODEs can be seen below: | ||
| + | <br> | ||
| + | [[Image:TUD ODE default solution.png|thumb|550px]] | ||
| + | <br> | ||
| + | A function was written to determine the point at which the concentration of the final product reached a certain threshold. A threshold of 1E-8 M was used in our simulations, which corresponds to a handful of proteins within a cell, assuming a cell volume of around 1E-15 L. Using the default parameter values, a delay time of 385 min was predicted. | ||
==Sensitivity== | ==Sensitivity== | ||
| + | A sensitivity analysis was done on the system. This looked at how variations in each parameter would influence the delay time. Parameters were swept over a range of values while the change in delay time was observed. The normalized sensitivity for various parameters is shown below, ordered with the most sensitive parameter at the top. | ||
| + | <br> | ||
{| border="1" align="center" | {| border="1" align="center" | ||
| - | | <b>Parameter</b> || <b>Normalized Sensitivity</b> | + | | <b>Parameter</b> || Description || <b>Normalized Sensitivity</b> |
|- align="left" | |- align="left" | ||
| - | | | + | | d<sub>mRNA</sub> || mRNA degradation rate || 4.65 |
|- align="left" | |- align="left" | ||
| - | | | + | | d<sub>CI</sub> || CI degradation rate || 4.64 |
|- align="left" | |- align="left" | ||
| - | | | + | | a<sub>λp</sub> || transcription leakage of λp || 4.04 |
|- align="left" | |- align="left" | ||
| - | | | + | | α<sub>2</sub> || CI translation rate (RBS strength) || 1.06 |
|- align="left" | |- align="left" | ||
| - | | | + | | d<sub>TetR</sub> || TetR degradation rate || 0.53 |
|- align="left" | |- align="left" | ||
| - | | c<sub> | + | | c<sub>λp</sub> || λp promoter strength || 0.42 |
|- align="left" | |- align="left" | ||
| - | | | + | | α<sub>4</sub> || GFP translation rate (RBS strength) || 0.38 |
|- align="left" | |- align="left" | ||
| - | | | + | | c<sub>pTet</sub> || pTet promoter strength || 0.36 |
|- align="left" | |- align="left" | ||
| - | | | + | | c<sub>pLac</sub> || pLac promoter strength || 0.22 |
| + | |- align="left" | ||
| + | | α<sub>1</sub> || TetR translation rate (RBS strength) || 0.14 | ||
| + | |- align="left" | ||
| + | | a<sub>pTet</sub> || transcription leakage of pTet || 0.12 | ||
| + | |- align="left" | ||
| + | | d<sub>GFP</sub> || GFP degradation rate || 0.04 | ||
| + | |- align="left" | ||
| + | | a<sub>pLac</sub> || transcription leakage of pLac || 0.00 | ||
| + | |- align="left" | ||
| + | | d<sub>LacI</sub> || LacI degradation rate || 0.00 | ||
|- align="left" | |- align="left" | ||
|} | |} | ||
| + | <br> | ||
| + | As we can see in the table above, the system is most affected by changes in d<sub>mRNA</sub>, d<sub>CI</sub>, and a<sub>λp</sub>. This agrees well with the analysis done in the paper [http://bioinformatics.oxfordjournals.org/cgi/content/abstract/25/12/i169 A general computational method for robustness analysis with applications to synthetic gene networks] [[Team:TUDelft/Modeling_References|[9]]] on a similar cascade system. That analysis also found the sensitivity to be greatest for the degradation parameters. | ||
| + | |||
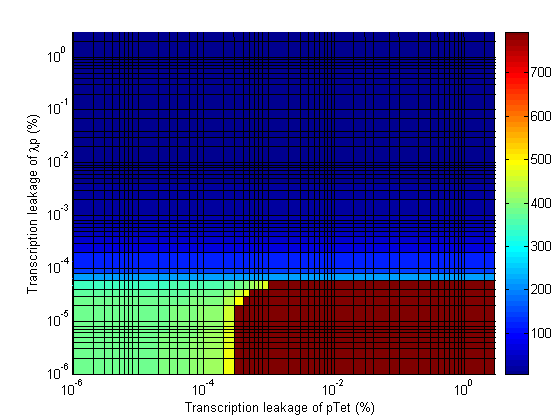
| + | The analysis also revealed that the leakage on the last stage of the cascade is also an important parameter. The plot below shows how the delay time quickly drops as the leakage passes 10<sup>-4</sup> %. | ||
| + | |||
| + | [[Image:2d leakage lamp.png|430px|frame| Delay Time vs Transcription Leakage of λp.]] | ||
| + | |||
| + | <br><br> | ||
==Parameter Sweeps== | ==Parameter Sweeps== | ||
| + | |||
| + | |||
| + | In the following plots the delay time of the cascade is shown as a function of two different parameters. A delay time of 800 is used to represent an infinite delay (maroon colour). | ||
{|border = "0" | {|border = "0" | ||
|- | |- | ||
| - | |rowspan=" | + | |rowspan="4"| |
| - | <gallery> | + | <gallery widths="230px" perrow="2" heights="188px" > |
| - | + | ||
| - | Image: | + | Image:3d_dCI_vs_dGFP.png|Degradation rate of GFP vs Degradation rate of CI. |
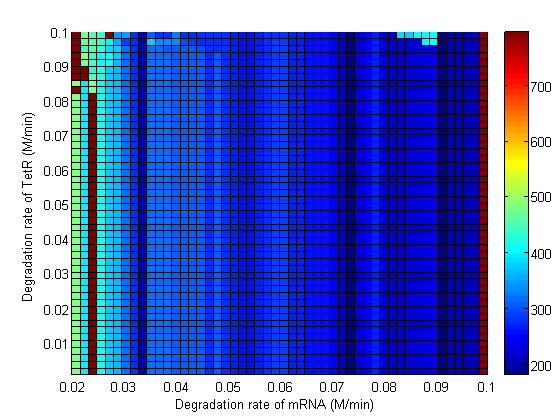
| - | Image: | + | Image:3d_dmRNA_vs_dTetR.png|Degradation rate of TetR vs Degradation rate of mRNA. |
| - | + | Image:3d alpha2 vs alpha4.png|Translation Rate α1 (of TetR mRNA) vs Translation Rate α4 (of GFP mRNA). | |
| - | Image: | + | Image:3d Clamp vs alpha4.png|Maximum Transcription Rate of λp vs Translation Rate α4 (of GFP mRNA). |
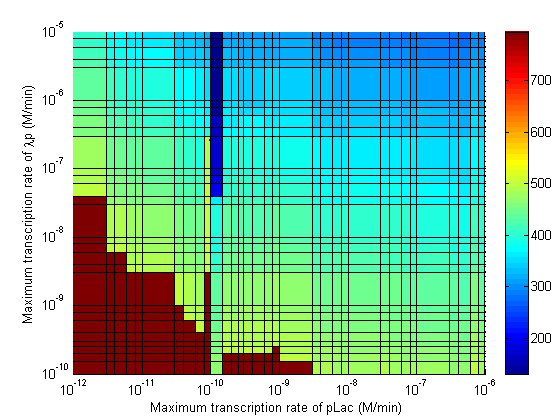
| - | Image: | + | Image:3d CpLac vs Clamp.png|Maximum Transcription Rate of pLac vs Maximum Transcription Rate of λp. |
| - | Image: | + | Image:3d CpLac vs CpTet.png|Maximum Transcription Rate of pLac vs Maximum Transcription Rate of pTet. |
| + | Image:3d CpTet vs alpha2.png|Maximum Transcription Rate of pTet vs Translation Rate α2 (of CI mRNA). | ||
| + | |||
| + | Image:3d CpTet vs Clamp.png|Maximum Transcription Rate of pTet vs Maximum Transcription Rate of λp. | ||
| + | |||
| + | Image:3d leakage a2 vs a4.png|Transcription Leakage of λp vs Transcription Leakage of pTet. | ||
</gallery> | </gallery> | ||
|} | |} | ||
| + | <br> | ||
==Stability== | ==Stability== | ||
| + | <br> | ||
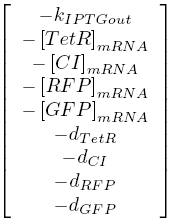
| + | The stability of the system was investigated by determining the Jacobian of the system of ODEs analytically. The eigenvalues of the Jacobian can be seen below. | ||
| + | |||
| + | <center> | ||
| + | [[Image:TUDjacobian.png]] | ||
| + | </center> | ||
| - | + | Since all of the values are real and negative we can conclude that our system will be stable. | |
| + | <br> | ||
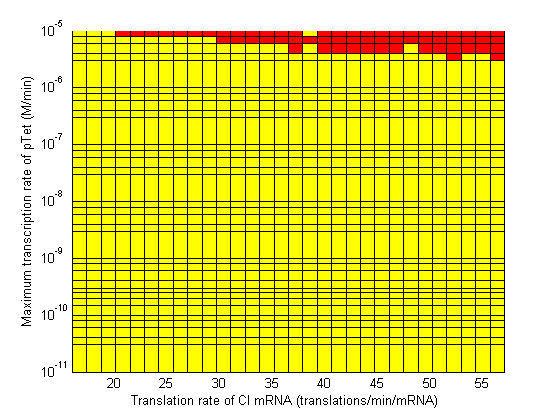
| + | As the parameter sweeps revealed, there are areas within the parameter space with a delay time of infinity. The plots below highlight the regions of the parameter space to avoid. Red areas correspond to a delay time of infinity, while blue areas correspond to a delay time less than 10 min (not enough time to conjugate the signal plasmid further). The cascade operates properly within the yellow areas. | ||
| + | <br> | ||
{|border = "0" | {|border = "0" | ||
|- | |- | ||
|rowspan="2"| | |rowspan="2"| | ||
| - | <gallery> | + | <gallery widths="230px" perrow="2" heights="188px"> |
| - | + | ||
| - | Image: | + | Image:3d_dCI_vs_dGFP_regions.png| Degradation rate of GFP vs Degradation rate of CI. |
| - | Image: | + | Image:3d_dmRNA_vs_dTetR_regions.png| Degradation rate of TetR vs Degradation rate of mRNA. |
| - | + | Image:3d Clamp vs alpha4 regions.png| Maximum Transcription Rate of λp vs Translation Rate α4 (of GFP mRNA). | |
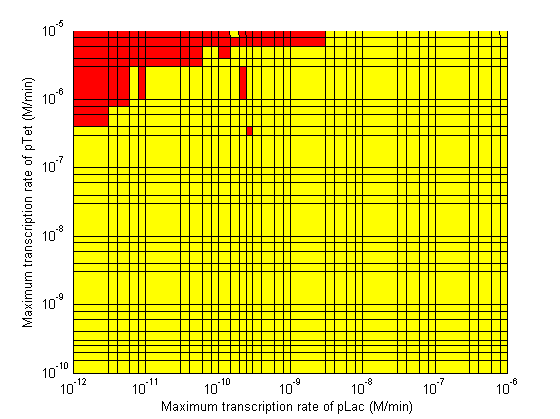
| - | Image: | + | Image:3d CpLac vs Clamp regions.png| Maximum Transcription Rate of pLac vs Maximum Transcription Rate of λp. |
| - | Image: | + | Image:3d CpLac vs CpTet regions.png| Maximum Transcription Rate of pLac vs Maximum Transcription Rate of pTet. |
| - | Image: | + | Image:3d CpTet vs alpha2 regions.png| Maximum Transcription Rate of pTet vs Translation Rate α2 (of CI mRNA). |
| + | Image:3d CpTet vs Clamp regions.png| Maximum Transcription Rate of pTet vs Maximum Transcription Rate of λp. | ||
| + | |||
| + | Image:3d leakage a2 vs a4 regions tri.png| Transcription Leakage of λp vs Transcription Leakage of pTet. | ||
</gallery> | </gallery> | ||
|} | |} | ||
| + | <br> | ||
==Design Recommendations== | ==Design Recommendations== | ||
| - | + | <br> | |
Based on the results of the simulations, a series of recommendations were given to the delay team to aid them in choosing parts which would maximize the delay time. | Based on the results of the simulations, a series of recommendations were given to the delay team to aid them in choosing parts which would maximize the delay time. | ||
| + | # Minimize the degradation rate of CI, do not use genes with [http://partsregistry.org/Protein_domains/Degradation degradation tags]. | ||
# Significant transcription leakages greatly shorten the delay time. Attempt to minimize leakages. Leakage of λp is a far bigger problem than pTet leakage. | # Significant transcription leakages greatly shorten the delay time. Attempt to minimize leakages. Leakage of λp is a far bigger problem than pTet leakage. | ||
# Use a weak promoter and a weak RBS on the last stage (λp) of the cascade. | # Use a weak promoter and a weak RBS on the last stage (λp) of the cascade. | ||
| Line 123: | Line 181: | ||
# A weak RBS on TetR gene is favorable. | # A weak RBS on TetR gene is favorable. | ||
# A weak RBS on the endonuclease is favorable although a strong RBS can be used for the GFP gene. | # A weak RBS on the endonuclease is favorable although a strong RBS can be used for the GFP gene. | ||
| - | # When choosing RBS and promoter strengths avoid the red areas on the stability plots. | + | # When choosing RBS and promoter strengths avoid the red and blue areas on the stability plots. |
| + | |||
| + | <br> | ||
| + | All of the source code used can be found on the [[Team:TUDelft/Downloads | downloads]] page. | ||
{{Template:TUDelftiGEM2009_end}} | {{Template:TUDelftiGEM2009_end}} | ||
Latest revision as of 22:47, 21 October 2009
Modeling the Transcriptional Cascade
The modeling of the transcriptional cascade had several objectives:
- to provide the delay team with design guidelines which would maximize the delay time
- to asses the affect of parameter variation on the delay time
- to determine areas of instability in the parameter space
A schematic of the system to be modeled can be seen below. A full description of the Transcriptional Cascade can be found here.
The system shown above has one input: IPTG and one primary output: GFP. It is characterized by the delay time, which is measured as the time between the induction with IPTG and a certain expression level in the final product (GFP).
ODEs
The kinetic equations were written out in a Matlab script. A total of ten equations were used: one for the diffusion of IPTG into the cell, one for the binding of IPTG to LacI, as well as four transcription equations, and four translation equations for the various levels of the cascade.
The notation in this system of equations can be seen in the table below:
| Symbol | Definition |
| kIPTGin, kIPTGout | rate constants |
| k50IPTG, k50LacI, k50TetR, k50CI | dissociation constants |
| dmRNA | mRNA degradation rate |
| dTetR, dCI, dRFP, dGFP | protein degradation rates |
| apLac, apTet, aλp | transcription leakage (%) |
| cpLac, cpTet, cλp | maximum transcription rates |
| α1, α2, α3, α4 | translation rates |
| nIPTG, nLacI, nTetR, nCI | Hill coefficients |
| [X]mRNA | concentration of X mRNA |
The solution with the default parameters of the system of ODEs can be seen below:
A function was written to determine the point at which the concentration of the final product reached a certain threshold. A threshold of 1E-8 M was used in our simulations, which corresponds to a handful of proteins within a cell, assuming a cell volume of around 1E-15 L. Using the default parameter values, a delay time of 385 min was predicted.
Sensitivity
A sensitivity analysis was done on the system. This looked at how variations in each parameter would influence the delay time. Parameters were swept over a range of values while the change in delay time was observed. The normalized sensitivity for various parameters is shown below, ordered with the most sensitive parameter at the top.
| Parameter | Description | Normalized Sensitivity |
| dmRNA | mRNA degradation rate | 4.65 |
| dCI | CI degradation rate | 4.64 |
| aλp | transcription leakage of λp | 4.04 |
| α2 | CI translation rate (RBS strength) | 1.06 |
| dTetR | TetR degradation rate | 0.53 |
| cλp | λp promoter strength | 0.42 |
| α4 | GFP translation rate (RBS strength) | 0.38 |
| cpTet | pTet promoter strength | 0.36 |
| cpLac | pLac promoter strength | 0.22 |
| α1 | TetR translation rate (RBS strength) | 0.14 |
| apTet | transcription leakage of pTet | 0.12 |
| dGFP | GFP degradation rate | 0.04 |
| apLac | transcription leakage of pLac | 0.00 |
| dLacI | LacI degradation rate | 0.00 |
As we can see in the table above, the system is most affected by changes in dmRNA, dCI, and aλp. This agrees well with the analysis done in the paper [http://bioinformatics.oxfordjournals.org/cgi/content/abstract/25/12/i169 A general computational method for robustness analysis with applications to synthetic gene networks] [9] on a similar cascade system. That analysis also found the sensitivity to be greatest for the degradation parameters.
The analysis also revealed that the leakage on the last stage of the cascade is also an important parameter. The plot below shows how the delay time quickly drops as the leakage passes 10-4 %.
Parameter Sweeps
In the following plots the delay time of the cascade is shown as a function of two different parameters. A delay time of 800 is used to represent an infinite delay (maroon colour).
|
|
Stability
The stability of the system was investigated by determining the Jacobian of the system of ODEs analytically. The eigenvalues of the Jacobian can be seen below.
Since all of the values are real and negative we can conclude that our system will be stable.
As the parameter sweeps revealed, there are areas within the parameter space with a delay time of infinity. The plots below highlight the regions of the parameter space to avoid. Red areas correspond to a delay time of infinity, while blue areas correspond to a delay time less than 10 min (not enough time to conjugate the signal plasmid further). The cascade operates properly within the yellow areas.
|
|
Design Recommendations
Based on the results of the simulations, a series of recommendations were given to the delay team to aid them in choosing parts which would maximize the delay time.
- Minimize the degradation rate of CI, do not use genes with [http://partsregistry.org/Protein_domains/Degradation degradation tags].
- Significant transcription leakages greatly shorten the delay time. Attempt to minimize leakages. Leakage of λp is a far bigger problem than pTet leakage.
- Use a weak promoter and a weak RBS on the last stage (λp) of the cascade.
- A weak pLac promoters is favorable.
- A strong pTet promoter is favorable.
- A strong RBS on CI gene is favorable.
- A weak RBS on TetR gene is favorable.
- A weak RBS on the endonuclease is favorable although a strong RBS can be used for the GFP gene.
- When choosing RBS and promoter strengths avoid the red and blue areas on the stability plots.
All of the source code used can be found on the downloads page.
 "
"