Team:UNICAMP-Brazil/Notebooks/September 13
From 2009.igem.org
(→New biobrick in biobrick format) |
(→finP+pSB1A3) |
||
| (11 intermediate revisions not shown) | |||
| Line 7: | Line 7: | ||
====finO+pSB1A3==== | ====finO+pSB1A3==== | ||
| - | * After the O/N period, today we transformed the ligation finO+pSB1A3 into electrocompetent E. coli bacteria, strain DH10B. We followed | + | *<p style=”text-align:justify;”>After the O/N period, today we transformed the ligation finO+pSB1A3 into electrocompetent ''E. coli'' bacteria, strain DH10B. We followed [https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Electroporation Protocol 3], without modifications.</p> |
| - | * We then plated the transfomed cells in LB-AMP | + | *<p style=”text-align:justify;”>We then plated the transfomed cells in LB-AMP medium, and let them grow at 37ºC for an O/N period.</p> |
| + | |||
| + | ''Marcelo'' | ||
====finP+pSB1A3==== | ====finP+pSB1A3==== | ||
| - | * We followed the same | + | *<p style=”text-align:justify;”>We followed the same procedure for the finP+pSB1A3 ligation.</p> |
''Marcelo'' | ''Marcelo'' | ||
| + | |||
| + | ====Purification: Cre-Recombinase without ATG==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>Today we realized the purification of the Cre-Recombinase without the ATG codon that was amplified on september 12th.</p> | ||
| + | *<p style=”text-align:justify;”>We purified the PCR's product by running an agarose gel of the entire product and then extracting the desirable band from the gel. We used Invitrogen's PureLink Quick Gel Extraction Kit to perform the purification, following the manufacturer's protocol without modifications ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Purification_of_DNA_fragments_from_agarose_gels Protocol 7]).</p> | ||
| + | *<p style=”text-align:justify;”>After the procedure, we ran an agarose gel with the resulting product in order to confirm purification. We purified the Cre-Recombinase without ATG codon!</p> | ||
| + | |||
| + | ''Víctor'' | ||
==''' YeastGuard '''== | ==''' YeastGuard '''== | ||
| - | ==== | + | ==== Parts+Biofusion - The first screening==== |
| - | * 6 colonies of each new biobrick were chosen of the plates made yesterday to grow in liquid LB. | + | *<p style=”text-align:justify;”>6 colonies of each new biobrick were chosen of the plates made yesterday to grow in liquid LB.</p> |
| - | * Then, we performed miniprep to get our new biobricks purified(Protocol 2). | + | *<p style=”text-align:justify;”>Then, we performed miniprep to get our new biobricks purified ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Mini-Prep Protocol 2]).</p> |
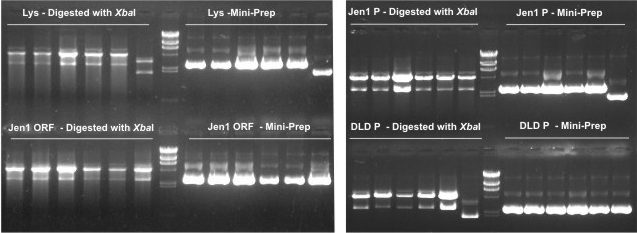
| - | * We then digested the plasmids obtained by the miniprep with ''Xba''I to confirm the correct ligation of the parts following the biobrick format ( | + | *<p style=”text-align:justify;”>We then digested the plasmids obtained by the miniprep with ''Xba''I to confirm the correct ligation of the parts following the biobrick format ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/T4_DNA_Ligase Protocol 11]). However, the electrophoresis gel (shown below) showed an unexpected pattern of bands that can't confirm correctly ligations of our biobricks. =(</p> |
[[Image:MInipreps e digestões Wesley.jpg |center|]] | [[Image:MInipreps e digestões Wesley.jpg |center|]] | ||
| - | ''Wesley | + | ''Wesley'' |
{{:Team:UNICAMP-Brazil/inc_rodape}} | {{:Team:UNICAMP-Brazil/inc_rodape}} | ||
Latest revision as of 02:30, 22 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"