Team:UNICAMP-Brazil/Notebooks/September 13
From 2009.igem.org
(Difference between revisions)
(→New biobrick - The first screening) |
(→New biobrick - The first screening) |
||
| Line 22: | Line 22: | ||
* 6 colonies of each new biobrick were chosen of the plates made yesterday to grow in liquid LB. | * 6 colonies of each new biobrick were chosen of the plates made yesterday to grow in liquid LB. | ||
* Then, we performed miniprep to get our new biobricks purified ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Mini-Prep Protocol 2]). | * Then, we performed miniprep to get our new biobricks purified ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Mini-Prep Protocol 2]). | ||
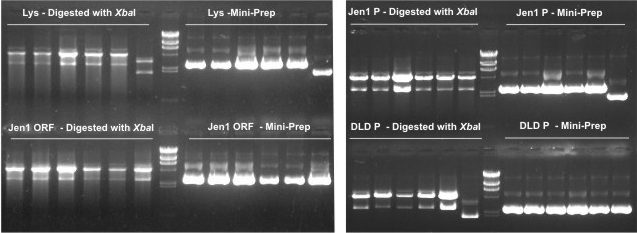
| - | * We then digested the plasmids obtained by the miniprep with ''Xba''I to confirm the correct ligation of the parts following the biobrick format ( | + | * We then digested the plasmids obtained by the miniprep with ''Xba''I to confirm the correct ligation of the parts following the biobrick format ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/T4_DNA_Ligase Protocol 11]). However, the electrophoresis gel (shown below) showed an unexpected pattern of bands that can't confirm correctly ligations of our biobricks. =( |
[[Image:MInipreps e digestões Wesley.jpg |center|]] | [[Image:MInipreps e digestões Wesley.jpg |center|]] | ||
Revision as of 05:02, 3 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"