Team:UNICAMP-Brazil/Notebooks/October 11
From 2009.igem.org
(New page: ==<html><script>var page = wgPageName.substr(30);document.write(page.replace(/_/, " "));</script></html>== {{:Team:UNICAMP-Brazil/inc_topo}} {{:Team:UNICAMP-Brazil/inc calendar}} __NOTOC...) |
(→ColiGuard) |
||
| (26 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| - | |||
{{:Team:UNICAMP-Brazil/inc_topo}} | {{:Team:UNICAMP-Brazil/inc_topo}} | ||
{{:Team:UNICAMP-Brazil/inc calendar}} | {{:Team:UNICAMP-Brazil/inc calendar}} | ||
| + | __NOTOC__ | ||
| + | ==''' ColiGuard '''== | ||
| - | + | ====Cre-Recombinase without ATG + terminator==== | |
| + | *<p style=”text-align:justify;”>Now that we have the Cre-Recombinase biobrick, our new plan is to assemble its device. We first decided to add the terminator region BBa_B0015 to the Cre-Recombinase without ATG.</p> | ||
| + | *<p style=”text-align:justify;”>We performed the digestion from both terminator and the plasmids that have our biobrick (see October 5th, 9th and 10th). The Cre's digestion was performed with ''EcoRI'' and ''SpeI'' and the digestion of terminator with ''EcoRI'' and ''XbaI'', both incubated at 37°C for 3 hours.</p> | ||
| + | *<p style=”text-align:justify;”>We ran an 1% agarose gel and observed that the digestion of the terminator ocurred as expected, but not the plasmids containing Cre-Recombinase. What happened?</p> | ||
| - | + | ''Victor'' | |
| + | |||
| + | ====PCR colony of the BBa B0014 + BBa K112806 Ligation==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>We chose 9 colonies to do the PCR, 3 results were positive, the colonies 2, 5 and 7 and appeared in the agarose gel with the expected size! 2 colonies was used to do a innoculum that will be used in a miniprep tomorrow.</p> | ||
| + | |||
| + | [[Image:PCRB0014 K112806..JPG|center]] | ||
| + | |||
| + | ''Luige'' | ||
====finOP-pGEM digestion purification==== | ====finOP-pGEM digestion purification==== | ||
| - | * After confirming correctly finOP with pGEM | + | *<p style=”text-align:justify;”>After confirming correctly finOP with pGEM ligations, today we gather all confirmed samples (for finO and for finP) into a single sample for each one.</p> |
| - | * We ran 40 uL of each gathered sample in an agarose gel for later purification. Unfortunately, finP band appeared extremely weak in the gel and we weren't able to purify it. | + | *<p style=”text-align:justify;”>We ran 40 uL of each gathered sample in an agarose gel for later purification. Unfortunately, finP band appeared extremely weak in the gel and we weren't able to purify it. As for finO, it's band appeared in gel, but we decided to wait for finP in order to perform both purifications together.</p> |
| - | + | *<p style=”text-align:justify;”>Thus, we took all the gathered finP sample left and concentrated it in speed vacuum, until ir reaches about 20 uL. Then we ran another agarose gel with this concentrated sample and with another 40 uL from finO gathered sample.</p> | |
| + | *<p style=”text-align:justify;”>This time, finP appeared as a barely visible band, but we were able to purify it, as we did for finO to.</p> | ||
| + | *<p style=”text-align:justify;”>We performed purification using Invitrogen's Purelink Quick Gel Extractin Kit, following manufacturer's protocol ([https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Purification_of_DNA_fragments_from_agarose_gels Protocol 7]) without modifications).</p> | ||
''Marcelo'' | ''Marcelo'' | ||
| + | ====finOP purification results==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>After purification procedure, we quantified total DNA present in both samples.</p> | ||
| + | *<p style=”text-align:justify;”>finO resulted in 15 ng/uL. Although very low, this amount might be enough for proceeding in biobrick construction.</p> | ||
| + | *<p style=”text-align:justify;”>As for finP, no amount could be measured. =/ Probably, the barely visible band hadn't enough DNA for detection or there was a significant loss during purification process.</p> | ||
| + | |||
| + | ''Marcelo'' | ||
| + | |||
| + | ====finO==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>Even with no longer finP available, we continued finO's work by ligating finO purified sample into biofusion digested vector, according to [https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/T4_DNA_Ligase Protocol 11].</p> | ||
| + | *<p style=”text-align:justify;”>We then dialyzed the ligation product for 20 minutes and transformed them into electrocompetent ''E. coli'' bacteria, strain DH10B, according to [https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Electroporation Protocol 3].</p> | ||
| + | *<p style=”text-align:justify;”>After one hour being incubated at 37 ºC, transformed cells were plated into LB-AMP media.</p> | ||
| + | *<p style=”text-align:justify;”>Plates were incubated at 37 ºC for an O/N period.</p> | ||
| + | |||
| + | ''Marcelo'' | ||
| + | |||
| + | ==== PY Promoter - Ligation reaction and transformation ==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>After the digestion reactions we did yesterday we performed the ligation reactions of our fragment (PY1 + RFP reporter) with BBa_J23100 and BBa_B0015 following [https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/T4_DNA_Ligase Protocol 11].</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>We then transformed these 2 ligations into electrocompetent ''E. coli'' bacteria, strain DH10B. We followed [https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/Electroporation Protocol 3] (Electroporation).</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>After the transformation we plated the transformed cells in LB-AMP plates (for BBa_J23100) and LB-AMP-KAN plates (for BBa_B0015) and let them grow at 37ºC for an O/N period.</p> | ||
| + | |||
| + | ''Fabi and Léo'' | ||
| + | |||
| + | ==''' YeastGuard '''== | ||
| + | ====[https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/pGEMStrategy New Strategy: pGEM]==== | ||
| + | *<p style=”text-align:justify;”>We transformed the ligation reaction of lysozyme in biofusion.</p> | ||
| + | |||
| + | *<p style=”text-align:justify;”>We did miniprep of pJEN1+Biofusion and pDLD+Biofusion (without the final ''Not''I site), digested the plasmids with ''Xba''I and ''Pst''I, ligated the digested fragment with biofusion again, in order to recover the second ''Not''I site. Then we transformed competent ''E. coli'' and plated in LB+Amp media.</p> | ||
| + | [[Image:biofusion-digestion.jpg|400px|center]] | ||
| + | |||
| + | *<p style=”text-align:justify;”>We did ligation reactions of Adh1+Biofusion (digested with ''Spe''I and ''Pst''I) with lysozyme(digested with ''Xba''I and ''Pst''I). If it works it will be our first device!</p> | ||
| + | |||
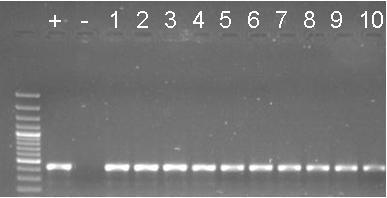
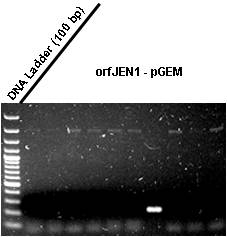
| + | *<p style=”text-align:justify;”>We screened JENorf+pGEM plates by colony PCR. Some colonies appeared to have the insert. We will do miniprep tomorrow of 3 colonies. The JENorf is still in pGEM!! Hurry up!!</p> | ||
| + | [[Image:screen.jpg|300px|center]] | ||
| + | |||
| + | ''Raíssa and Taís'' | ||
| + | |||
| + | ====pADH1+YFP==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>The colonies grew up very well in the plates. So, we selected 10 colonies to grow in liquid LB+AMP media O/N.</p> | ||
| + | |||
| + | ''Wesley and Gleidson'' | ||
| + | |||
| + | ====YEP358==== | ||
| + | |||
| + | *<p style=”text-align:justify;”>We inoculated positive colonies with YEP358 – β-galactosidase to plasmid extraction.</p> | ||
{{:Team:UNICAMP-Brazil/inc_rodape}} | {{:Team:UNICAMP-Brazil/inc_rodape}} | ||
Latest revision as of 01:55, 22 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"