Team:Paris/Addressing overview3
From 2009.igem.org
(→A. ClyA) |
Christophe.R (Talk | contribs) |
||
| (36 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{Template:Paris2009}} | {{Template:Paris2009}} | ||
{{Template:Paris2009_menu3}} | {{Template:Paris2009_menu3}} | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| + | <span/ id="1"> | ||
| + | ==Addressing the message in the outer membrane : ClyA== | ||
| + | <html> | ||
| + | <style type="text/css"> | ||
| + | #left-side { | ||
| + | position: absolute; | ||
| + | height: 23px; | ||
| + | width: 30px; | ||
| + | top: 0px; | ||
| + | left: 160px; | ||
| + | margin-top:10px; | ||
| + | padding-top: 7px; | ||
| + | background: url(https://static.igem.org/mediawiki/2009/1/1b/Left_menu_pari.png); | ||
| + | z-index:4; | ||
| + | } | ||
| - | + | #middle-side { | |
| + | height: 25px; | ||
| + | width: 380px; | ||
| + | position: absolute; | ||
| + | top: 0px; | ||
| + | left: 170px; | ||
| + | margin-top:10px; | ||
| + | padding-top: 5px; | ||
| + | background: #dadada; | ||
| + | z-index:5; | ||
| + | } | ||
| - | + | #right-side { | |
| + | position: absolute; | ||
| + | height: 23px; | ||
| + | width: 30px; | ||
| + | margin-top:10px; | ||
| + | padding-top: 7px; | ||
| + | top: 0px; | ||
| + | left: 530px; | ||
| + | background: url(https://static.igem.org/mediawiki/2009/4/40/Right_menu_paris.png); | ||
| + | z-index:4; | ||
| + | } | ||
| + | a.menu_sub { | ||
| + | padding-left: 7px; | ||
| + | padding-right: 7px; | ||
| + | } | ||
| - | + | a.menu_sub_active { | |
| - | + | padding-left: 7px; | |
| + | padding-right: 7px; | ||
| + | color:#b0310e; | ||
| + | font-weight:bold; | ||
| + | } | ||
| + | </style> | ||
| + | <div id="left-side"></div> | ||
| + | <div id="middle-side"><center> | ||
| + | <a class="menu_sub"href="https://2009.igem.org/Team:Paris/Addressing_overview2#bottom"> Main </a>| | ||
| + | <a class="menu_sub_active" href="https://2009.igem.org/Team:Paris/Addressing_overview3#bottom"> ClyA</a>| | ||
| + | <a class="menu_sub"href="https://2009.igem.org/Team:Paris/Addressing_overview4#bottom"> OmpA</a>| | ||
| + | <a class="menu_sub"href="https://2009.igem.org/Team:Paris/Addressing_overview2_strategy#bottom"> Our strategy</a>| | ||
| + | <a class="menu_sub"href="https://2009.igem.org/Team:Paris/Addressing_overview_Construction#bottom"> Construction</a> | ||
| + | </center> | ||
| + | </div> | ||
| + | <div id="right-side"></div> | ||
| + | </html> | ||
| + | We work on the cell-cell communication using vesicle: | ||
| + | <br> | ||
| + | In this part we look into adressing a protein into the sender outer membrane that could be incoporated into outer membrane vesicles (OMVs). This protein would then be able to transmit a message after the fusion of OMVs with a receiver cell. | ||
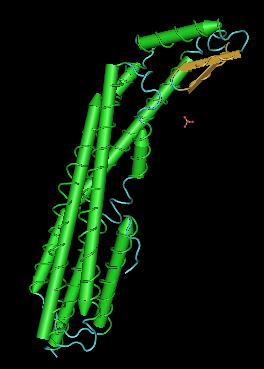
| + | In this direction ClyA (the cytolysine A of E.Coli) seems to be a good candidate. ClyA is one of the proteins that has been previously detected into OMVs and is known to be specificly exported to the outer membrane <sup>[[Team:Paris/Addressing_overview3#References|[3]]]</sup>. ClyA is thus expressed on bacteria and OMVs surface. Moreover, when ClyA is overproduced, it is accumulated into the periplasmic space<sup>[[Team:Paris/Addressing_overview3#References|[3]]]</sup>. | ||
| - | + | However there is an inconvenient to use this protein. ClyA is an alpha-Pore Forming Toxin (PFT). PFT are widely distributed proteins which form lesions in biological membranes. They exhibit their toxic effect in different manner. The first one is that ClyA allows the destruction of membrane permeability barrier. Furthermore, the toxic effect of ClyA could be explain by its capacity to deliver toxic component after the assembly of 8 or 13 of its subunits. PFTs can be subdivided into two classes; α-PFTs and β-PFTs, depending on the suspected mode of membrane integration, either by α-helical or β-sheet elements.<sup>[[Team:Paris/Addressing_overview3#References|[2]]]</sup> | |
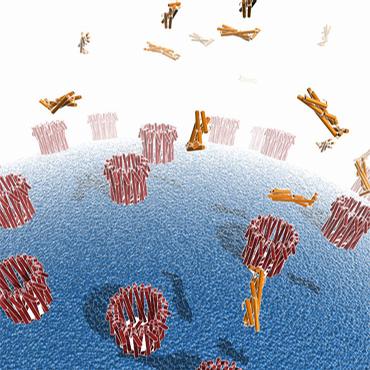
| + | [[Image:Clya_simple.jpg|ClyA subunit|150px|left]] [[Image:Clya_structure.jpg|ClyA assembled|150px|right]] [[Image:ClyA.jpg|ClyA are assembling in outer membrane of a host cell|150px|center]] | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | + | Some article argue that E.Coli K12 use this ClyA to lyse other cell (specially mamalian cell or eurcaryote cell<sup>[[Team:Paris/Addressing_overview3#References|[3]]]</sup>). But E. coli cells expressing clyA do not lyse each other. | |
| - | |||
| - | + | Kim et al. have successfully fused clyA to GFP in order to observe vesicles <sup>[[Team:Paris/Addressing_overview3#References|[1]]]</sup>, so we know that we can try to fuse clyA to a protein domain that would induce a signal transduction into the receiver cell. To see how we want to exploite clyA properties see [https://2009.igem.org/Team:Paris/Addressing_overview2_strategy#Overview our strategy]. | |
| - | |||
| - | + | {{Template:Paris2009_guided|Addressing_overview2#bottom|Addressing_overview4#bottom}} | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | <html> | ||
| + | </div> | ||
| + | <div id="paris_content_boxtop"> | ||
| + | </div> | ||
| + | <div id="paris_content"> | ||
| + | </html> | ||
| - | |||
| - | + | ====References==== | |
| - | - | + | <ol class="references"> |
| + | <li>[[Team:Paris/Addressing_overview3#1| ^]]Kim, J.-Y. & DeLisa, M.P. Engineered bacterial outer membrane vesicles with enhanced functionality J.Mol. Biol. (2008) 380, 51–66. [http://www.ncbi.nlm.nih.gov/pubmed/18511069 18511069]</li> | ||
| + | <li>[[Team:Paris/Addressing_overview3#1| ^]]Muller, M. & Ban, N. The structure of a cytolytic a-helical toxin pore reveals its assembly mechanism Nature (4 June 2009) 459, 726-730. [http://www.ncbi.nlm.nih.gov/pubmed/19421192 19421192]</li> | ||
| + | <li>[[Team:Paris/Addressing_overview3#1| ^]]Wai, S.N. & Lindmark, B. Vesicle-Mediated Export and Assembly of Pore-Forming Oligomers of the Enterobacterial ClyA Cytotoxin Cell (October 2003), 115,25-35. [http://www.ncbi.nlm.nih.gov/pubmed/14532000 14532000]</li> | ||
| + | </ol> | ||
Latest revision as of 02:49, 22 October 2009
Addressing the message in the outer membrane : ClyA
We work on the cell-cell communication using vesicle:
In this part we look into adressing a protein into the sender outer membrane that could be incoporated into outer membrane vesicles (OMVs). This protein would then be able to transmit a message after the fusion of OMVs with a receiver cell.
In this direction ClyA (the cytolysine A of E.Coli) seems to be a good candidate. ClyA is one of the proteins that has been previously detected into OMVs and is known to be specificly exported to the outer membrane [3]. ClyA is thus expressed on bacteria and OMVs surface. Moreover, when ClyA is overproduced, it is accumulated into the periplasmic space[3].
However there is an inconvenient to use this protein. ClyA is an alpha-Pore Forming Toxin (PFT). PFT are widely distributed proteins which form lesions in biological membranes. They exhibit their toxic effect in different manner. The first one is that ClyA allows the destruction of membrane permeability barrier. Furthermore, the toxic effect of ClyA could be explain by its capacity to deliver toxic component after the assembly of 8 or 13 of its subunits. PFTs can be subdivided into two classes; α-PFTs and β-PFTs, depending on the suspected mode of membrane integration, either by α-helical or β-sheet elements.[2]
Some article argue that E.Coli K12 use this ClyA to lyse other cell (specially mamalian cell or eurcaryote cell[3]). But E. coli cells expressing clyA do not lyse each other.
Kim et al. have successfully fused clyA to GFP in order to observe vesicles [1], so we know that we can try to fuse clyA to a protein domain that would induce a signal transduction into the receiver cell. To see how we want to exploite clyA properties see our strategy.
References
- ^Kim, J.-Y. & DeLisa, M.P. Engineered bacterial outer membrane vesicles with enhanced functionality J.Mol. Biol. (2008) 380, 51–66. [http://www.ncbi.nlm.nih.gov/pubmed/18511069 18511069]
- ^Muller, M. & Ban, N. The structure of a cytolytic a-helical toxin pore reveals its assembly mechanism Nature (4 June 2009) 459, 726-730. [http://www.ncbi.nlm.nih.gov/pubmed/19421192 19421192]
- ^Wai, S.N. & Lindmark, B. Vesicle-Mediated Export and Assembly of Pore-Forming Oligomers of the Enterobacterial ClyA Cytotoxin Cell (October 2003), 115,25-35. [http://www.ncbi.nlm.nih.gov/pubmed/14532000 14532000]
 "
"