Team:Paris/Addressing overview4
From 2009.igem.org
(→Adressin the message in the outer membrane : OmpA) |
Christophe.R (Talk | contribs) |
||
| (9 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
| - | == | + | <span/ id="1"> |
| + | ==Addressing the message to the outer membrane : OmpA== | ||
<html> | <html> | ||
<style type="text/css"> | <style type="text/css"> | ||
| Line 67: | Line 68: | ||
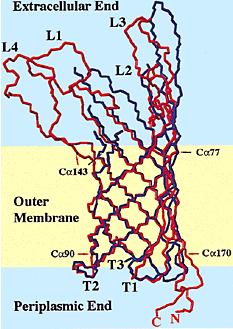
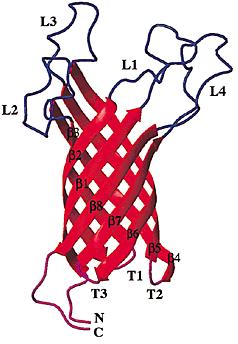
| - | OmpA as previously been used in protein fusion to expose heterologuous protein domains to the surface of | + | Outer membrane protein A (OmpA) as previously been used in protein fusion to expose heterologuous protein domains to the surface of ''Escherichia coli''. As an outer membrane protein, ompA could be incorporated into vesicles and is similarly to clyA also a good candidate for adressing protein domains to vesicles. |
| - | [[Image:OmpA1.JPG|250px|left]][[Image:OmpA2.JPG| | + | [[Image:OmpA1.JPG|250px|left]][[Image:OmpA2.JPG|250px|right]] |
| - | + | OmpA is a major protein in the Escherichia coli outer membrane. OmpA plays a vital structural role in ''E. coli'', and suggested that a perfect β-barrel structure of OmpA is important for outer membrane stability <sup>[[Team:Paris/Addressing_overview4#References|[1]]]</sup>. OmpA is the most well-studied outer membrane protein in ''E. coli''. This 325-residue protein was thought to contain two domains. The classic N-terminal domain, consisting of 171 amino acid residues, was shown to cross the membrane eight times in antiparallel β-strands with four relatively large and hydrophilic surface-exposed loops and short periplasmic turns<sup>[[Team:Paris/Addressing_overview4#References|[2]]]</sup>. The C-terminal domain is located in the periplasm, and binds to the peptidoglycan thus connecting it to the outer membrane<sup>[[Team:Paris/Addressing_overview4#References|[3]]]</sup>. The function of OmpA is thought to contribute to the structural integrity of the outer membrane | |
| - | + | along with murein lipoprotein<sup>[[Team:Paris/Addressing_overview4#References|[4]]]</sup> and peptidoglycanassociated lipoprotein . In addition to its structural role, OmpA serves as a receptor of colicin and several phages<sup>[[Team:Paris/Addressing_overview4#References|[5]]]</sup>, and it's required in F-conjugation<sup>[[Team:Paris/Addressing_overview4#References|[6]]]</sup>,<sup>[[Team:Paris/Addressing_overview4#References|[7]]]</sup> | |
| - | + | ||
| - | along with murein lipoprotein[ | + | |
| Line 86: | Line 85: | ||
| - | ==== | + | ====References==== |
| - | + | <ol class="references"> | |
| - | [ | + | <li>[[Team:Paris/Addressing_overview4#1| ^]]Ying Wang, (2002) The Function of OmpA in Escherichia coli, Biochem Biophys Res Commun.292(2):396-401. [http://www.ncbi.nlm.nih.gov/pubmed/11906175 11906175] </li> |
| - | + | <li>[[Team:Paris/Addressing_overview4#1| ^]]Arora, A., Abildgaard, F., Bushweller, J. H., and Tamm, L. K. (2001) Structure of outer membrane protein A transmembrane domain by NMR spectroscopy. Nat. Struct. Biol 8, 334–338. [http://www.ncbi.nlm.nih.gov/pubmed/11276254 11276254] </li> | |
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | <li>[[Team:Paris/Addressing_overview4#1| ^]] |
| - | + | Koebnik, R. (1995) Proposal for a peptidoglycan associating alpha-helical motif in the C-terminal regions of some bacterial cell-surface proteins. Mol. Microbiol. 16, 1269–1270. [http://www.ncbi.nlm.nih.gov/pubmed/8577259 8577259] </li> | |
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | <li>[[Team:Paris/Addressing_overview4#1| ^]] |
| - | + | Braun, V., and Bosch, V. (1972) Sequence of the mureinlipoprotein and the attachment site of the lipid. Eur. J. Biochem. 28, 51–69. [http://www.ncbi.nlm.nih.gov/pubmed/4261992 4261992] </li> | |
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | <li>[[Team:Paris/Addressing_overview4#1| ^]] |
| - | + | Lazzaroni, J.-C., and Portalier, R. (1992) The excC gene of Escherichia coli K-12 required for cell envelope integrity encodes the peptidoglycan-associated lipoprotein. Mol. Microbiol. 6, 735–742. [http://www.ncbi.nlm.nih.gov/pubmed/1574003 1574003] </li> | |
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | <li>[[Team:Paris/Addressing_overview4#1| ^]]Schweizer, M., and Henning, U. (1977) Action of major outer cell envelope membrane protein in conjugation of Escherichia coli K-12. J. Bacteriol. 129, 1651–1652. [http://www.ncbi.nlm.nih.gov/pubmed/321438 321438]</li> |
| - | + | <li>[[Team:Paris/Addressing_overview4#1| ^]]Koebnik, R. (1999) Structural and functional roles of the surfaceexposed loops of the β-barrel membrane protein OmpA from Escherichia coli. J. Bacteriol. 181, 3688–3694.[http://www.ncbi.nlm.nih.gov/pubmed/10368142 10368142] </li> | |
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | </ol> |
| - | + | ||
| - | [http://www.ncbi.nlm.nih.gov/pubmed/ | + | |
Latest revision as of 02:51, 22 October 2009
Addressing the message to the outer membrane : OmpA
Outer membrane protein A (OmpA) as previously been used in protein fusion to expose heterologuous protein domains to the surface of Escherichia coli. As an outer membrane protein, ompA could be incorporated into vesicles and is similarly to clyA also a good candidate for adressing protein domains to vesicles.
OmpA is a major protein in the Escherichia coli outer membrane. OmpA plays a vital structural role in E. coli, and suggested that a perfect β-barrel structure of OmpA is important for outer membrane stability [1]. OmpA is the most well-studied outer membrane protein in E. coli. This 325-residue protein was thought to contain two domains. The classic N-terminal domain, consisting of 171 amino acid residues, was shown to cross the membrane eight times in antiparallel β-strands with four relatively large and hydrophilic surface-exposed loops and short periplasmic turns[2]. The C-terminal domain is located in the periplasm, and binds to the peptidoglycan thus connecting it to the outer membrane[3]. The function of OmpA is thought to contribute to the structural integrity of the outer membrane along with murein lipoprotein[4] and peptidoglycanassociated lipoprotein . In addition to its structural role, OmpA serves as a receptor of colicin and several phages[5], and it's required in F-conjugation[6],[7]
References
- ^Ying Wang, (2002) The Function of OmpA in Escherichia coli, Biochem Biophys Res Commun.292(2):396-401. [http://www.ncbi.nlm.nih.gov/pubmed/11906175 11906175]
- ^Arora, A., Abildgaard, F., Bushweller, J. H., and Tamm, L. K. (2001) Structure of outer membrane protein A transmembrane domain by NMR spectroscopy. Nat. Struct. Biol 8, 334–338. [http://www.ncbi.nlm.nih.gov/pubmed/11276254 11276254]
- ^ Koebnik, R. (1995) Proposal for a peptidoglycan associating alpha-helical motif in the C-terminal regions of some bacterial cell-surface proteins. Mol. Microbiol. 16, 1269–1270. [http://www.ncbi.nlm.nih.gov/pubmed/8577259 8577259]
- ^ Braun, V., and Bosch, V. (1972) Sequence of the mureinlipoprotein and the attachment site of the lipid. Eur. J. Biochem. 28, 51–69. [http://www.ncbi.nlm.nih.gov/pubmed/4261992 4261992]
- ^ Lazzaroni, J.-C., and Portalier, R. (1992) The excC gene of Escherichia coli K-12 required for cell envelope integrity encodes the peptidoglycan-associated lipoprotein. Mol. Microbiol. 6, 735–742. [http://www.ncbi.nlm.nih.gov/pubmed/1574003 1574003]
- ^Schweizer, M., and Henning, U. (1977) Action of major outer cell envelope membrane protein in conjugation of Escherichia coli K-12. J. Bacteriol. 129, 1651–1652. [http://www.ncbi.nlm.nih.gov/pubmed/321438 321438]
- ^Koebnik, R. (1999) Structural and functional roles of the surfaceexposed loops of the β-barrel membrane protein OmpA from Escherichia coli. J. Bacteriol. 181, 3688–3694.[http://www.ncbi.nlm.nih.gov/pubmed/10368142 10368142]
 "
"