Team:Paris/Transduction overview2 transduction
From 2009.igem.org
Christophe.R (Talk | contribs) (→References) |
Christophe.R (Talk | contribs) (→Two-component system) |
||
| Line 200: | Line 200: | ||
{{Template:Paris2009_guided|Transduction_overview2#top.23top|Transduction_overview2_strategy#bottom}} | {{Template:Paris2009_guided|Transduction_overview2#top.23top|Transduction_overview2_strategy#bottom}} | ||
| + | |||
| + | |||
| + | <html> | ||
| + | </div> | ||
| + | <div id="paris_content_boxtop"> | ||
| + | </div> | ||
| + | <div id="paris_content"> | ||
| + | </html> | ||
| + | ====References==== | ||
| + | <ol class="references"> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Kentaro Tomii and Minoru Kanehisa, A Comparative Analysis of ABC Transporters in Complete Microbial Genomes,Genome Res. 1998 Oct;8(10):1048-59. [http://www.ncbi.nlm.nih.gov/pubmed/9799792 9799792] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Hollenstein, K.; R.J. Dawson; K.P. Locher. 2007. Structure and mechanism of ABC transporter proteins. Curr. Opin. Struct. Biol. 17, 412-418. [http://www.ncbi.nlm.nih.gov/pubmed/17723295 17723295] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Davidson, A.L.; E. Dassa; C. Orelle; and J. Chen. 2008. Structure, function, and evolution of bacterial ATP-binding cassette systems. Microbiol. Mol. Biol. Rev. 72(2), 317-364.[http://www.ncbi.nlm.nih.gov/pubmed/18535149 18535149] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Henderson, D.P. and S.M. Payne. 1994. Vibrio cholerae iron transport system: roles of heme and siderophore iron transport in virulence and identification of a gene associated with multiple iron transport systems. Infect. Immun. 62, 5120-5125. [http://www.ncbi.nlm.nih.gov/pubmed/7927795 7927795] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]] | ||
| + | Davidson, A.L.; E. Dassa; C. Orelle; and J. Chen. 2008. Structure, function, and evolution of bacterial ATP-binding cassette systems. Microbiol. Mol. Biol. Rev. 72(2), 317-364.[http://www.ncbi.nlm.nih.gov/pubmed/18535149 18535149]</li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Stock AM, Robinson VL, Goudreau PN. Two-component signal transduction.2000, Annu. Rev. Biochem. 69: 183–215.[http://www.ncbi.nlm.nih.gov/pubmed/10966457 10966457] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Mascher T, Helmann JD, Unden G. Stimulus perception in bacterial signal-transducing histidine kinases. 2006. Microbiol. Mol. Biol. Rev. 70 (4): 910–38.[http://www.ncbi.nlm.nih.gov/pubmed/17158704 17158704] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]] | ||
| + | Attwood PV, Piggott MJ, Zu XL, Besant PG. Focus on phosphohistidine, 2007, Amino Acids 32 (1): 145–56. [http://www.ncbi.nlm.nih.gov/pubmed/17103118 17103118] </li> | ||
| + | <li> [[Team:Paris/Transduction_overview2_transduction#1 | ^]]Kyriakidis DA, Tiligada E. Signal transduction and adaptive regulation through bacterial two-component systems: the Escherichia coli AtoSC paradigm, 2009, Amino Acids.37(3):443-58. [http://www.ncbi.nlm.nih.gov/pubmed/19198978 19198978] </li> | ||
| + | </ol> | ||
Latest revision as of 03:16, 22 October 2009
iGEM > Paris > Receiving the message > Transduction
Contents |
Transduction
In biology, signal transduction refers to any process by which a cell converts one kind of signal or stimulus into another. Most processes of signal transduction involve ordered sequences of biochemical reactions inside the cell, which are carried out by enzymes and sometimes activated by second messengers, resulting in a signal transduction pathway.
Such processes are usually rapid, lasting on the order of milliseconds in the case of ion flux, or minutes for the activation of proteic cascades, but some can take hours, and even days (as is the case with gene expression), to complete.
We must also refer to the amplification of the signal , in which a relative small stimulus can elicit a large response. It is often du to the increasing numbers of protein ativation
implied in the transduction pathways.
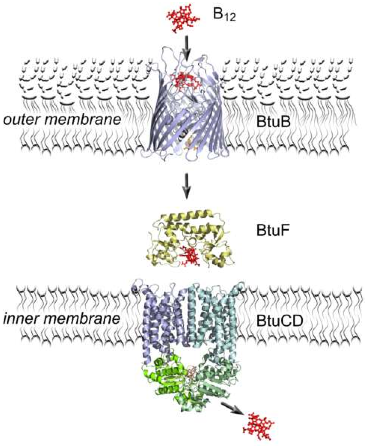
ABC transporters
The ABC transporter is a major class of cellular translocation machinery encoded in the largest set of paralogous genes [1].
ABC (ATP binding cassette) transporters are active transport systems of the cell, which is widespread in archaea, eubacteria, and eukaryotes. They require energy in the form of adenosine triphosphate (ATP) to translocate substrates across cell membranes. These proteins harness the energy of ATP binding and/or hydrolysis to drive conformational changes in the transmembrane domain (TMD) and consequently transports molecules [2].
ABC transporters are also known as the periplasmic binding protein-dependent transport system in Gram-negative bacteria and the binding-lipoprotein-dependent transport system in Gram positive bacteria.
The transporter shows a common global organization with three types of molecular components. Typically, it consists of two integral membrane proteins (permeases) each having six transmembrane segments, two peripheral membrane proteins that bind and hydrolyze ATP, and a periplasmic (or lipoprotein) substrate-binding protein. The ATP-binding protein component is the most conserved, the TMD is somewhat less conserved, and the substrate-binding protein component is most divergent in terms of the sequence similarity. The ABC transporters form the largest group of paralogous genes in bacterial and archaeal genomes , and the genes for the three components frequently form an operon.
Uses
Bacterial ABC transporters are essential in cell viability, virulence, and pathogenicity [3] Iron ABC uptake systems, for example, are important effectors of virulence [4]. Prokaryotic ABC transporters utilize the energy of ATP hydrolysis to transport lot of different substrates across cellular membranes. They are divided into 2 functionnal categories :
- importers : they mediate the uptake of amino-acids, peptids, sugar ... into the cell, so they allow the transport of protein directly into the cytolpasm to activate the transcription [5].
- exporters : they transport lipids and some polysaccharides from the cytoplasm to the periplasm.
Advantages/drawbacks
The import system of these transporters is really interesting and attractive four our project to transmit a "message" from vesicles to target cells. The main advantage is that the protein of interest is directly translocated in the cytoplasm and if it is a transcription factor it could activated immediatly the response. The communication is simple,fast and efficient. But this system is not perfect... One major drawback is that ABC transporter system is a nutriment uptake system. So, basically only (very) small molecules are able to pass thought the membranes.
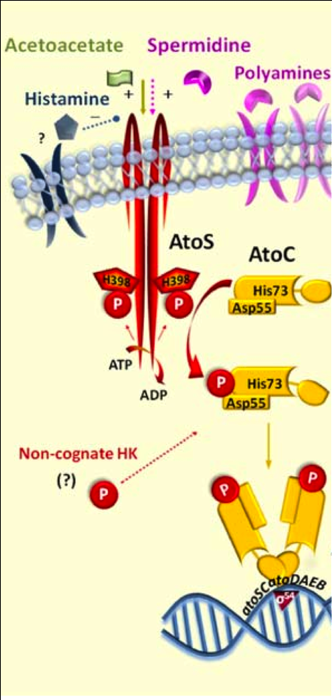
Two-component system
The Two-Component System (TCS) can be considered as a widely spread class of biosensor knowing that adaptive signal transduction within microbial cells involving a multi-faceted regulated phosphotransfer mechanism that comprises structural rearrangements of sensor histidine kinases upon ligand-binding and phosphorylation-induced conformational changes in response regulators of versatile TCS, arisen early in bacterial evolution [6], [7]
In most eubacteria, two-component proteins typically constitute 1% of encoded peptides. In pathogenic bacteria they control the expression of important pathogenetic factors, in addition to regulating basic housekeeping functions. The widespread distribution of two-component signal transduction systems in Bacteria and Achaea reflects their biological value as major sensing and response elements to a wide range of environmental insults that are tuned to respond from within milliseconds to hours [8]. Although TCSs are probably the most eficient means of adaptation to conventional stressful stimuli encountered by bacteria during their lifespan, the plasticity of some of these sophisticated systems may contribute to strain-specific cellular processes and to the acquisition of distinct features and phenotypes, particularly in pathogens [9].
To put the structure in a nutshell: A typical TCS consists of a transmembrane dimeric sensor histidine kinase (HK) and a cytoplasmic cognate response regulator (RR). In gram negative bacteria there is often a Periplasmic Binding Protein which optimize the detection of the molecule localized in the periplasm by a high affinity for the HK after binding the specific molecule. The following scheme shows a typical ABC mechanism :
Uses
Two-component systems serve as a basic stimulus-response coupling mechanism to allow organisms to sense and respond to changes in many different environmental conditions [6].
By transferring a packet of molecule synthesize by the donor but not present in the medium,
the arrival could activate the transcription of gene of interest. This will allow us to check for the "bacterial communication"
Advantages/drawbacks
Most gene regulation proteins that were known were single proteins, often homodimers or homotetramers, which bound to two ligands, a metabolic intermediate or a cis-acting gene regulation element. The functional state of the regulatory protein was thus modified by binding to the metabolic intermediate. The consequence of ligand binding, an altered state of the regulatory protein, was directed to the appropriate gene(s) by the protein's DNA binding activity.
The main advantage of TCS is that the mechanism is dedicated to the transcription of gene under a specific promoter. Four our project, the difficulty is to find an easily exportable and detectable signal.
In contrats, most of the biosensor are generally sensible to high diffusible molecule for which the vesicles transport is useless, so using vesicules to transmit a message doesn't seems the best way to explore.
References
- ^Kentaro Tomii and Minoru Kanehisa, A Comparative Analysis of ABC Transporters in Complete Microbial Genomes,Genome Res. 1998 Oct;8(10):1048-59. [http://www.ncbi.nlm.nih.gov/pubmed/9799792 9799792]
- ^Hollenstein, K.; R.J. Dawson; K.P. Locher. 2007. Structure and mechanism of ABC transporter proteins. Curr. Opin. Struct. Biol. 17, 412-418. [http://www.ncbi.nlm.nih.gov/pubmed/17723295 17723295]
- ^Davidson, A.L.; E. Dassa; C. Orelle; and J. Chen. 2008. Structure, function, and evolution of bacterial ATP-binding cassette systems. Microbiol. Mol. Biol. Rev. 72(2), 317-364.[http://www.ncbi.nlm.nih.gov/pubmed/18535149 18535149]
- ^Henderson, D.P. and S.M. Payne. 1994. Vibrio cholerae iron transport system: roles of heme and siderophore iron transport in virulence and identification of a gene associated with multiple iron transport systems. Infect. Immun. 62, 5120-5125. [http://www.ncbi.nlm.nih.gov/pubmed/7927795 7927795]
- ^ Davidson, A.L.; E. Dassa; C. Orelle; and J. Chen. 2008. Structure, function, and evolution of bacterial ATP-binding cassette systems. Microbiol. Mol. Biol. Rev. 72(2), 317-364.[http://www.ncbi.nlm.nih.gov/pubmed/18535149 18535149]
- ^Stock AM, Robinson VL, Goudreau PN. Two-component signal transduction.2000, Annu. Rev. Biochem. 69: 183–215.[http://www.ncbi.nlm.nih.gov/pubmed/10966457 10966457]
- ^Mascher T, Helmann JD, Unden G. Stimulus perception in bacterial signal-transducing histidine kinases. 2006. Microbiol. Mol. Biol. Rev. 70 (4): 910–38.[http://www.ncbi.nlm.nih.gov/pubmed/17158704 17158704]
- ^ Attwood PV, Piggott MJ, Zu XL, Besant PG. Focus on phosphohistidine, 2007, Amino Acids 32 (1): 145–56. [http://www.ncbi.nlm.nih.gov/pubmed/17103118 17103118]
- ^Kyriakidis DA, Tiligada E. Signal transduction and adaptive regulation through bacterial two-component systems: the Escherichia coli AtoSC paradigm, 2009, Amino Acids.37(3):443-58. [http://www.ncbi.nlm.nih.gov/pubmed/19198978 19198978]
 "
"