Team:Paris/Production modeling2
From 2009.igem.org
(Difference between revisions)
Christophe.R (Talk | contribs) |
Christophe.R (Talk | contribs) (→Modeling Vesicles creation.) |
||
| Line 13: | Line 13: | ||
| - | + | =='''Modeling Vesicles creation== | |
Our model is based on three different physical phenomenon: | Our model is based on three different physical phenomenon: | ||
Revision as of 21:59, 14 October 2009
iGEM > Paris > DryLab > Vesicle biophysics Model (vesicle model)
Modeling Vesicles creation
Our model is based on three different physical phenomenon:
- The lipid surface conformation, Tol-Pal proteins diffusion and the increase of the osmotic pressure in the periplasm
- The Lipid conformation of the outer membrane is a well known problem: at 35°c the lipid bilayer behaves like a liquid which conformation character is ruled by an energy called the bending energy. This energy represents the fact that the lipid bilayer will search a special conformation depending on the shape and the chemical properties of its constituents.
- The conformation of the lippopolysaccharides and the normal phospholipids can be seen has a lipid bilayer which conformed itself as a liquid crystal at the growth culture. The conformation of lipid bilayer has been well studied and a lot of theoretical results have been shown. Some of them are of great interest for our project. This conformation is something which can enable us to understand the way vesicle can be produced and how they will be received. In our project we first notice that from a basic calculus we can obtain very interesting results on the way outer membrane vesicles can be produced.
- We first use the bending energy has a rough shape for our model and its understanding:
- This formula give use the abilities to explain the way lipids will organize together. E is the energy of a whole lipid bilayer (or monolayer).Kb and Kg are Bending and Gaussian modulii which can be obtained by experiments. γ0 is the intrinsic curvature of the outer membrane which describes the local form of a lipid bilayer when it is at is lowest state of energy ,the more stable. γm and Hg are the mean curvature and the gaussian curvature. Our first task was to calculate the energy of two different shapes of membranes.
- We compute the E.coli Shape before the division and then the vesicles' shape.
- We consider E.coli's shape as a cylinder of rayon r =0.3 μm and of infinite length. The aim of this first representation was to estimate this energy in the division region of E.coli before division. With this approximation γm is equal to 2/r and γg=0. In this case:
- For the vesicles we consider their basic shape as a sphere of rayon r’ so the bending energy by lipid area units is:
- Thus as the area of a sphere is known and is independent of the location on the sphere we can write: In the same way we can write for an area of E.coli lipids that the bending energy is:
- The considerations aforementioned can provide a basic vision of the statistical repartition of vesicles in case of absence of integrity control system in the outer membrane.
- The first energy is the potential energy of the lipid area in E.coli outer membrane necessary to the construction of a vesicle and the second one the potential energy of the same lipid area but in the conformation of a vesicle shape. So the energy which must be given to the whole system to create a vesicle is:
- We can suppose that the most easily created vesicles will be the ones which requires a minimum energy . By derivation we find that the minimum his obtain for:
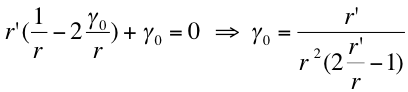
|
- Hence as we know that the range of created vesicles radii is 25 nm to 175nm we can suppose that the r’ is somehow about 100 nm and so:
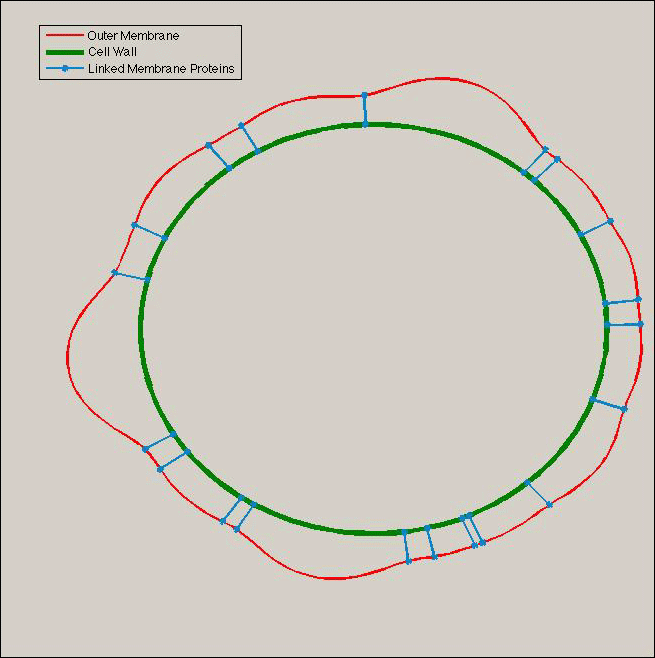
- We can consider the orders of magnitude as really realistic. In fact we know that the E.coli lipid bilayer is built of distinct types of lipids: Lippopolysacharides and simple phospholipids. LPS are located in the exterior lipid layer of the outer membrane. The others are located in the interior lipid bilayer. Moreover, those LPS present a sugar extension toward the medium. Those sugars can bind to each other. So we can assume that they are going to create clusters and to curve the membrane toward the exterior of E.coli.
- Tol and Pal are membrane proteins which are located respectively in the outer and the inner membrane. The diffusion of proteins in those lipid bilayers can be modelled by a probabilistic Brownian movement. This diffusion model gives us the law of probability for the location of Tol and Pal on the membranes. It has been observed that the Tol and Pal proteins interact with each other, which is linked to the membrane stability: indeed the Tol and Pal will bind inner and outer membrane and furthermore stabilize the outer membrane using the peptidoglycan rigidity.
- The osmotic pressure in the periplasm is the same that the medium pressure in normal time. But during the division period of the bacteria the peptidoglycan is degraded to be recycled in a new cell wall. During this phenomena of turn-over a part of the peptidoglycan is released in the periplasm which increased its osmotic pressure.
- Thus, we can consider that if there is not enough Tol-Pal linked proteins the outer membrane will distort to create a beginning of vesicle. But in this part of the membrane the tol pal proteins will not have the possibility to bind themselves and they will be free to diffuse in other parts of the membranes. The surface shape will guide the proteins to the border of the vesicle and stabilized the shape of the vesicle.
- To describe the time variation of the vesicles creation we first have to use a more complicated approach: we need to use an equation which is obtained by derivation of the bending energy:
Ou-Yang and Helfrich [Phys. Rev. Lett. 59 (1987) 2486]
- Our first model which didn't use the brownian movement :
- We first conciders that the membrane equation can be first simplified to a one dimentional model :in a first approach we use a one dimensional model based on a polar curvature simplification approximation :
- the curvature in polar is:
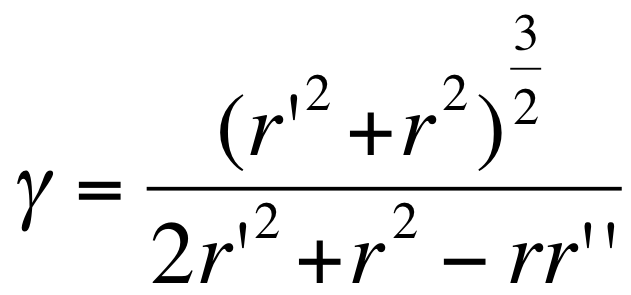
|
- With the hypothesis of a cylinder form the mean and gaussian curvatures can be written as:
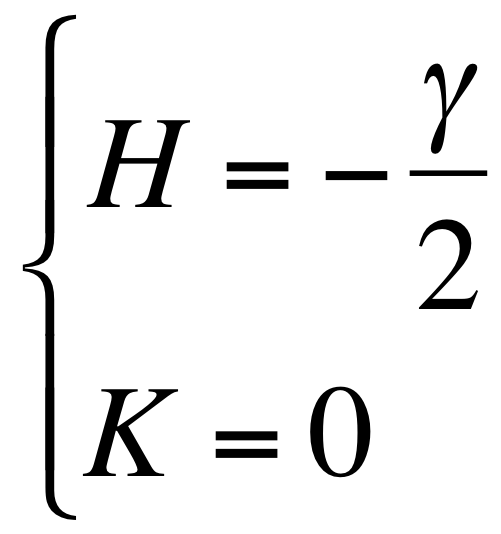
|
- indeed we have in those hypothesis:
|
- Thous we can concider the polar system of equation:
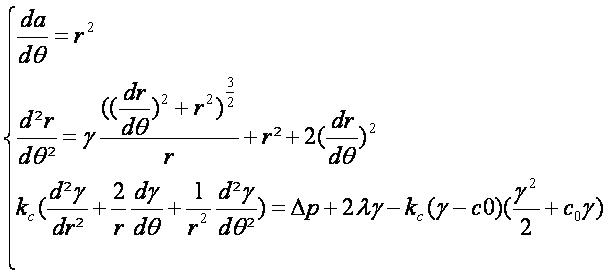
|
- to have a unique solution we must use some boundary conditions to compute our model (Cauchy Lipshitz thorem for the mathematician ;)) those conditions are simple without the peptidoglycan attachement lypoproteins (Pal). As the model is dependent of the angle we must impose the fact that r(0)=r(2π). In addition the Δp must be concidered as dependent of the quantity of periplasmic species.
- Indeed the osmotic pressure can be written in this way:
- And by concidering that we have weak concentrations we can concider a simpliest formula analogue to the perfect gases law:
- Finaly we can write:
- And Thous the whole parameters are given. In addition we can translate the role of the Tol/Pal System has boundary condition on the whole system: a cluster of tol-Pal can be translate as a point of radius equal to the peptidoglycan ones. Finally we can have this type of results:
|
- Brownian motion:
- The Brownian motion links the concentration of proteins with the shape of the membrane they are includes in:
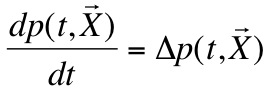
|
- P is the density probability of presence from proteins.
- The Laplacian of P explains the fact that the concentration of proteins like Pal increased in regions of negative curvature. Such a model can explain some last observations [Kumaran & Losick] “negative membrane curvatures as a cue for sub-cellular localization of a bacterial protein”. So we can assume that the Pal will be confined to region with negative curvature and so the probability of linking between Pal and Tol increased in those regions enhancing the stability and so enlarging those regions.
- So Brownian motion is a good explanation for the proteins migration toward the septa during division.
- But this Brownian motion can explain the vesiculation too. When there are too few proteins we can assume the creation of a vesicle at germinal states (called bleb). At the border of the blebs an amount of Pal protein prevent the whole membrane to swallow and keep the vesicle in form. But at the border between the blebs and the protein rich area curvature is highly negative and so the quantity of proteins in those regions increases enlarging the region toward the centre of the blebs.
- Then two different options are expectable: the blebs is too small and then the proteins are “zippering” back the blebs. But if the bleb is big enough the zippering became a separation between the vesicle and the bacteria. The good vision of this blebs forming can be understood in this way: the proteins rich region on the border pass from an annular form toward a circular one.
 "
"