Team:Paris/Transduction modeling The Fec Operon as used in our system : chemical equations and kinetics
From 2009.igem.org
iGEM > Paris > Reception > Modeling
Contents |
Fec operon in our system
To try and understand our system, we decided to divide the global in simple reactions traducing the chemical steps of the transduction cascade ; each one of these reactions is described with a cinetic law, thus allowing to run both deterministic and stochastic simulation (see here for further informations on these simulations). In our work, we assumed that the crossing of the periplasm is the limiting step ; consequently, we decided to distinguish two ways to describe this step:
- without a complexation between FecA_OM and FecR molecules.
- with a complexation between FecA_OM and FecR molecules.
Chemical Equations : no complexation in the periplasm
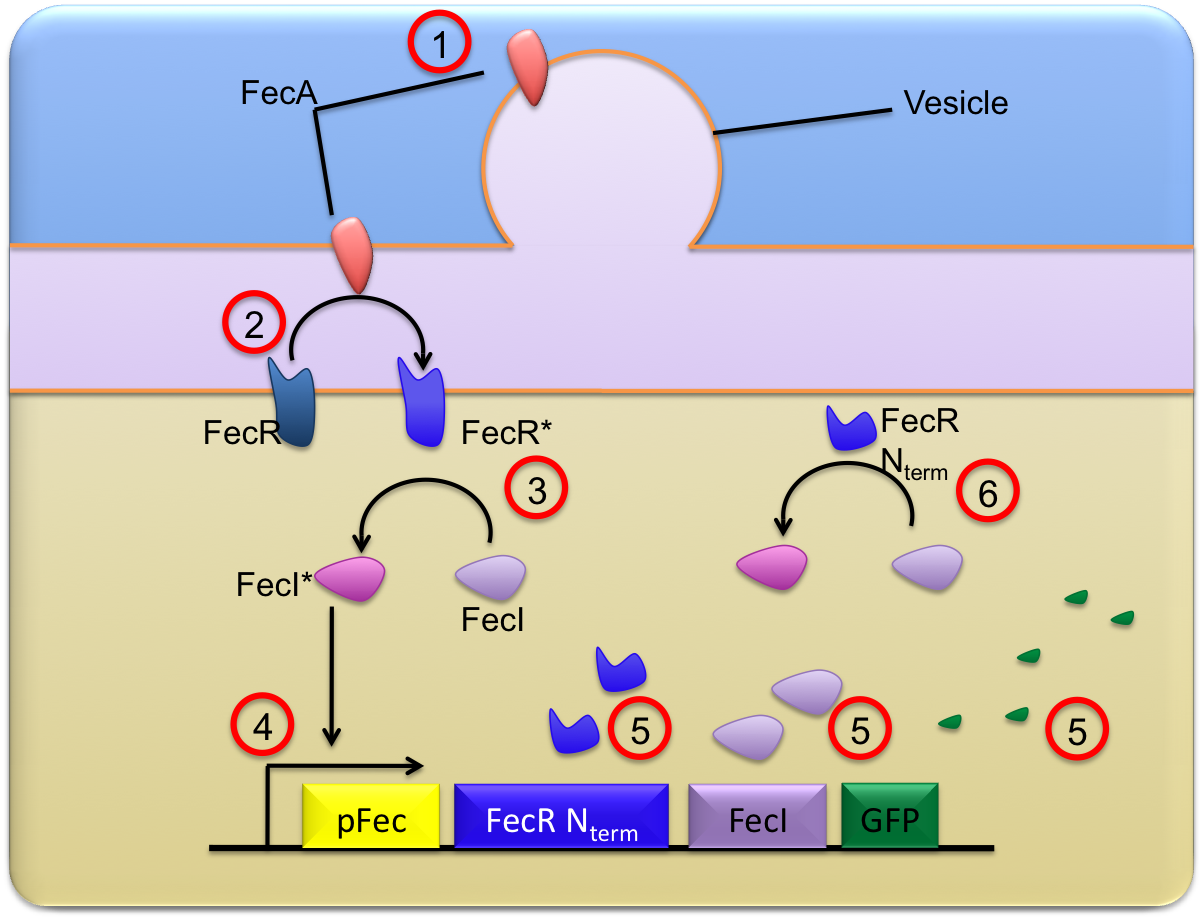
This trancriptional cascade can be described thanks to simple chemical equations traducing the chronological and chemical steps of our tranduction and activation system.
Description of main reactions
These equations are described on the scheme below :
- The FecA molecules coming from the vesicules are clowly diffusing on the lipid bilayer of the outer membrane while vesicules are fusioning ; they finally reach the bacteria outer membrane where they are in proper conditions to be able to activate the FeR molecules. This "crossing" from OMV to the outer membrane is explicited as a first reaction :
- Then, once in the outer membrane, these FecA molecules are able to activate the FecR proteins constitutively present in the receiver ;the FecA molecule directly activates FecR, and the phenomenon is described in a single reaction :
- Once FecR is activated, it can activate the FecI molecule ; to avoid an to much variability in the approaches, we decided to consider that this reaction occured as a single step (with no intermediate complex formation) :
- Then, FecI* can activate the pfec promoter in a reversible reaction :
- After the pfec activation, transcription and translation can start ; the creation of proteins is materialised by the single reaction :
where FecR_Nterm correspond to the residues ??-??? of FecR which make this molecule constitutively active (the FecR_Nterm is able to activate FecI without needing to be in presence of FecA)
- Finally, the constitutively active FecR_Nterm can activate the FecI proteins :
- It is also compulsory to consider some dilution and degradation reactions for the molecules FecA_OM, FecI, FecI*, FecR_Nterm, FecR* and GFP. The natural FecR is considered as a molecule having reached a steady state level ; as a consequence the only noticeable changes are the results of a reaction of activation.
Kinetics Equations
As we do not have much information on the kinetic laws, we have decided to chose a mass action law for each reaction. Each reaction has a kinetic constant k determining the reaction rate given the concentratin of reactants. This will lead us to write a differential system used for a deterministic resolution.
The first thing to do is to write the reaction rate of each chemical step for the two systems of reactions :
- A system without an intermediary step in the FecR activation
| Reaction Number | Kinetics Constants | Reaction | Reaction Rate |
| 1 | kf_1 | FecA_OMV ---> FecA_OM | kf_1*[FecA_OMV] |
| 2 | kf_2 | FecA_OM + FecR ---> FecA_OM + FecR* | kf_2*[FecA_OM]*[FecR] |
| 3 | kf_3 | FecR* + FecI ---> FecI* + FecR* | kf_3*[FecI*]*[FecR*] |
| 4 | kf_4 | FecI* + pfec <---> pfec* | kf_4*[FecI*]*[pfec]-kr_4[pfec*] |
| 5 | kf_5 | pfec* ---> FecI + FecR_Nterm + GFP | kf_5*[pfec*] |
| 6 | kf_6 | FecR_Nterm + FecI ---> FecI* + FecR_Nterm | kf_6*[FecR_Nterm]*[FecI] |
| 7 | kf_7 | FecA_OM ---> ∅ | kf_7*[FecA_OM] |
| 8 | kf_8 | FecI ---> ∅ | kf_8*[FecI] |
| 9 | kf_9 | FecI* ---> ∅ | kf_9*[FecI*] |
| 10 | kf_10 | FecR_Nterm ---> ∅ | kf_10*[FecR_Nterm] |
| 11 | kf_11 | FecR* ---> ∅ | kf_11*[FecR*] |
| 12 | kf_12 | GFP ---> ∅ | kf_12*[GFP] |
Chemical Equations : Complexation in the periplasm
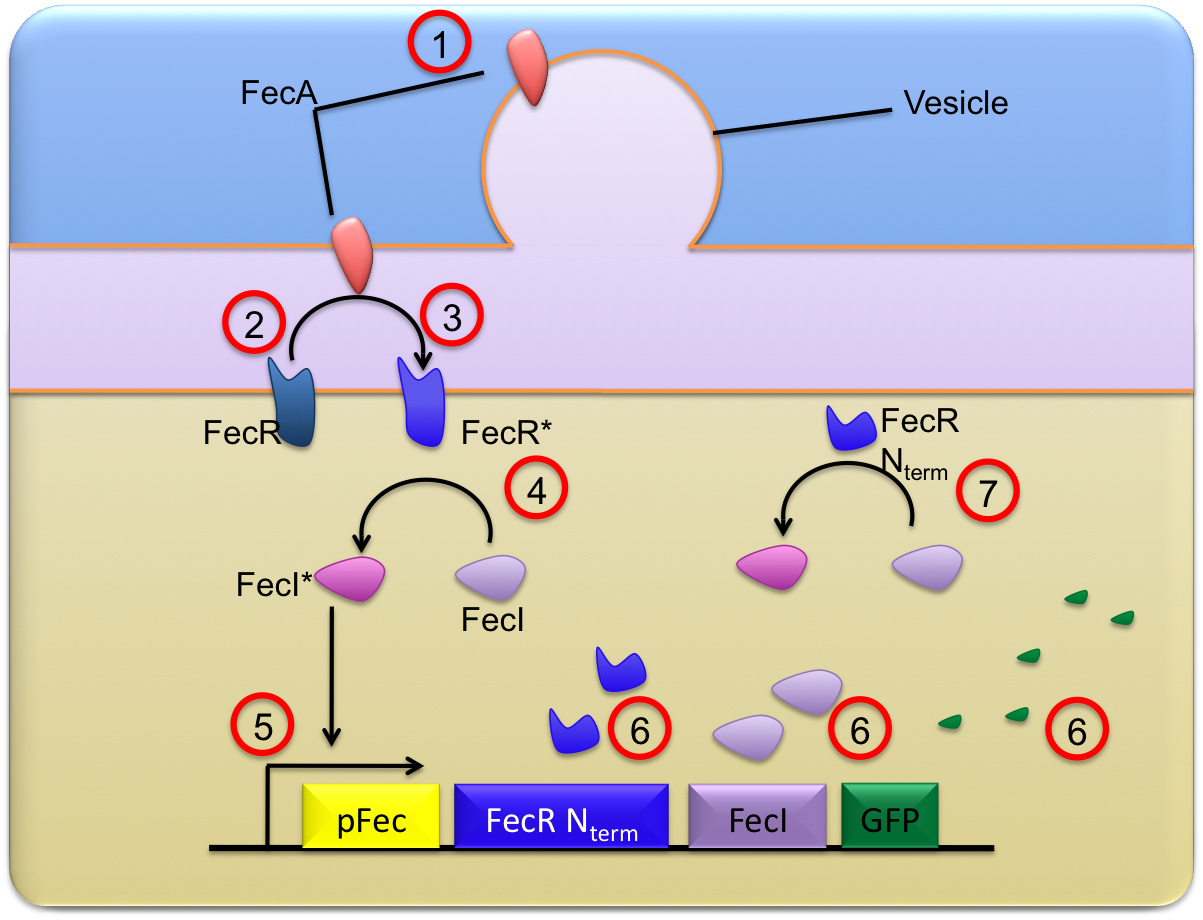
This trancriptional cascade can be described thanks to simple chemical equations traducing the chronological and chemical steps of our transduction and activation system.
Description of main reactions
These equations are described on the scheme below :
- The FecA molecules coming from the vesicules are clowly diffusing on the lipid bilayer of the outer membrane while vesicules are fusioning ; they finally reach the bacteria outer membrane where they are in proper conditions to be able to activate the FeR molecules. This "crossing" from OMV to the outer membrane is explicited as a first reaction :
- Then, once in the outer membrane, these FecA molecules are able to activate the FecR proteins constitutively present in the receiver ; FecA and FecR form a complex which is then destructed to release an activated FecR protein ; this mechanism is described through the 2 following reactions
- Once FecR is activated, it can activate the FecI molecule ; to avoid an to much variability in the approaches, we decided to consider that this reaction occured as a single step (with no intermediate complex formation) :
- Then, FecI* can activate the pfec promoter in a reversible reaction :
- After the pfec activation, transcription and translation can start ; the creation of proteins is materialised by the single reaction :
where FecR_Nterm correspond to the residues ??-??? of FecR which make this molecule constitutively active (the FecR_Nterm is able to activate FecI without needing to be in presence of FecA)
- Finally, the constitutively active FecR_Nterm can activate the FecI proteins :
- It is also compulsory to consider some dilution and degradation reactions for the molecules FecA_OM, FecI, FecI*, FecR_Nterm, FecR* and GFP. The natural FecR is considered as a molecule having reached a steady state level ; as a consequence the only noticeable changes are the results of a reaction of activation.
Kinetics Equations
As explained above, we do not have much information on the kinetic laws, we have decided to chose a mass action law for each reaction. Each reaction has a kinetic constant k determining the reaction rate given the concentratin of reactants. This will lead us to write a differential system used for a deterministic resolution.
- A system of equations with an intermediary reaction in the activation of FecR
| Reaction Number | Kinetics Constants | Reaction | Reaction Rate |
| 1 | kf_1 | FecA_OMV ---> FecA_OM | kf_1*[FecA_OMV] |
| 2 | kf_2 & kr_2 | FecA_OM + FecR <---> FecA_OM-FecR | kf_2*[FecA_OM]*[Fec_R] - kr_2*[FecA_OM-FecR] |
| 3 | kf_3 | FecA_OM-FecR ---> FecA_OM + FecR* | kf_3*[FecA_OM-FecR] |
| 4 | kf_4 | FecR* + FecI ---> FecI* + FecR* | kf_4*[FecR*]*[FecI] |
| 5 | kf_5 & kr_5 | FecI* + pfec <---> pfec* | kf_5*[FecI*]*[pfec] - kr_5*[pfec*] |
| 6 | kf_6 | pfec* ---> FecI + FecR_Nterm + GFP | kf_6*[pfec*] |
| 7 | kf_7 | FecR_Nterm + FecI ---> FecI* + FecR_Nterm | kf_7*[FecR_Nterm]*[FecI] |
| 8 | kf_8 | FecA_OM ---> ∅ | kf_8*[FecA_OM] |
| 9 | kf_9 | FecI ---> ∅ | kf_9*[FecI] |
| 10 | kf_10 | FecI* ---> ∅ | kf_10*[FecI*] |
| 11 | kf_11 | FecR_Nterm ---> ∅ | kf_11*[FecR_Nterm] |
| 12 | kf_12 | FecR* ---> ∅ | kf_12*[FecR*] |
| 13 | kf_13 | GFP ---> ∅ | kf_13*[GFP] |
 "
"