Team:Paris/Addressing overview4
From 2009.igem.org
Adressin the message in the outer membrane : OmpA
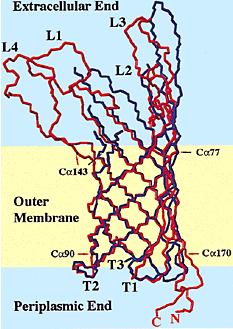
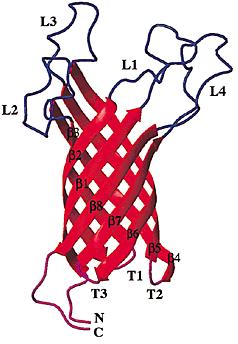
Outer membrane protein A (OmpA) is a major protein in the Escherichia coli outer membrane. OmpA plays a vital structural role in E. coli, and suggested that a perfect β-barrel structure of OmpA is important for outer membrane stability[1]. OmpA is the most well-studied outer membrane protein in Escherichia coli. This 325-residue protein was thought to contain two domains. The classic N-terminal domain, consisting of 171 amino acid residues, was shown to cross the membrane eight times in antiparallel β-strands with four relatively large and hydrophilic surface-exposed loops and short periplasmic turns[2]. The C-terminal domain is located in the periplasm, and binds to the peptidoglycan thus connecting it to the outer membrane[3]. The function of OmpA is thought to contribute to the structural integrity of the outer membrane along with murein lipoprotein[4] and peptidoglycanassociated lipoprotein . In addition to its structural role, OmpA serves as a receptor of colicin and several phages[5], and it's required in F-conjugation[6],[7]
Bibliography:
[1] Ying Wang, (2002) The Function of OmpA in Escherichia coli, Biochem Biophys Res Commun.292(2):396-401
[2] Arora, A., Abildgaard, F., Bushweller, J. H., and Tamm, L. K. (2001) Structure of outer membrane protein A transmembrane domain by NMR spectroscopy. Nat. Struct. Biol 8, 334–338.
[3] Koebnik, R. (1995) Proposal for a peptidoglycan associating alpha-helical motif in the C-terminal regions of some bacterial cell-surface proteins. Mol. Microbiol. 16, 1269–1270.
[4] Braun, V., and Bosch, V. (1972) Sequence of the mureinlipoprotein and the attachment site of the lipid. Eur. J. Biochem. 28, 51–69.
[5] Lazzaroni, J.-C., and Portalier, R. (1992) The excC gene of Escherichia coli K-12 required for cell envelope integrity encodes the peptidoglycan-associated lipoprotein. Mol. Microbiol. 6, 735–742.
[6 Schweizer, M., and Henning, U. (1977) Action of major outer cell envelope membrane protein in conjugation of Escherichia coli K-12. J. Bacteriol. 129, 1651–1652.
[7] Koebnik, R. (1999) Structural and functional roles of the surfaceexposed loops of the β-barrel membrane protein OmpA from Escherichia coli. J. Bacteriol. 181, 3688–3694.
 "
"