Team:UNICAMP-Brazil/Notebooks/October 10
From 2009.igem.org
Victor.negri (Talk | contribs) (→Cre-Recombinase - Confirmation: Stage II) |
|||
| (5 intermediate revisions not shown) | |||
| Line 48: | Line 48: | ||
====Cre-Recombinase - Confirmation: Stage II==== | ====Cre-Recombinase - Confirmation: Stage II==== | ||
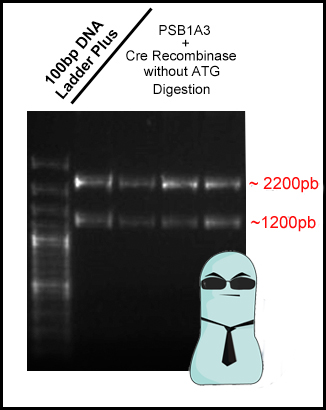
| - | *<p style=”text-align:justify;”>Now that stage I was successfully succeeded, today we performed stage II on confirming our Cre's biobrick. We performed a digestion of four of the samples confirmed in stage I, using XbaI and SpeI restriction enzymes.</p> | + | *<p style=”text-align:justify;”>Now that stage I was successfully succeeded, today we performed stage II on confirming our Cre's biobrick. We performed a digestion of four of the samples confirmed in stage I, using ''XbaI'' and ''SpeI'' restriction enzymes.</p> |
*<p style=”text-align:justify;”>Digestion lasted 3 hours. We then ran an 1% agarose gel, in order to confirm that the digestion actually worked.</p> | *<p style=”text-align:justify;”>Digestion lasted 3 hours. We then ran an 1% agarose gel, in order to confirm that the digestion actually worked.</p> | ||
*<p style=”text-align:justify;”>Finally, according to stage II...... yeah! it worked! We found two bands in all digested samples: one reaching 2200 bp (pSB1A3 vector) and other reaching 1200 bp (Cre-Recombinase).</p> | *<p style=”text-align:justify;”>Finally, according to stage II...... yeah! it worked! We found two bands in all digested samples: one reaching 2200 bp (pSB1A3 vector) and other reaching 1200 bp (Cre-Recombinase).</p> | ||
| + | |||
| + | |||
| + | [[image:Cre_final.jpg|center]] | ||
| + | |||
| + | |||
*<p style=”text-align:justify;”> We finally got a biobrick! Cre-Recombinase without ATG's biobrick now done! Next step will be assembling the entire device (see Differentiation Mechanism under Coliguard section for further information). | *<p style=”text-align:justify;”> We finally got a biobrick! Cre-Recombinase without ATG's biobrick now done! Next step will be assembling the entire device (see Differentiation Mechanism under Coliguard section for further information). | ||
| Line 57: | Line 62: | ||
==''' YeastGuard '''== | ==''' YeastGuard '''== | ||
====[https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/pGEMStrategy New Strategy: pGEM]==== | ====[https://2009.igem.org/Team:UNICAMP-Brazil/Protocols/pGEMStrategy New Strategy: pGEM]==== | ||
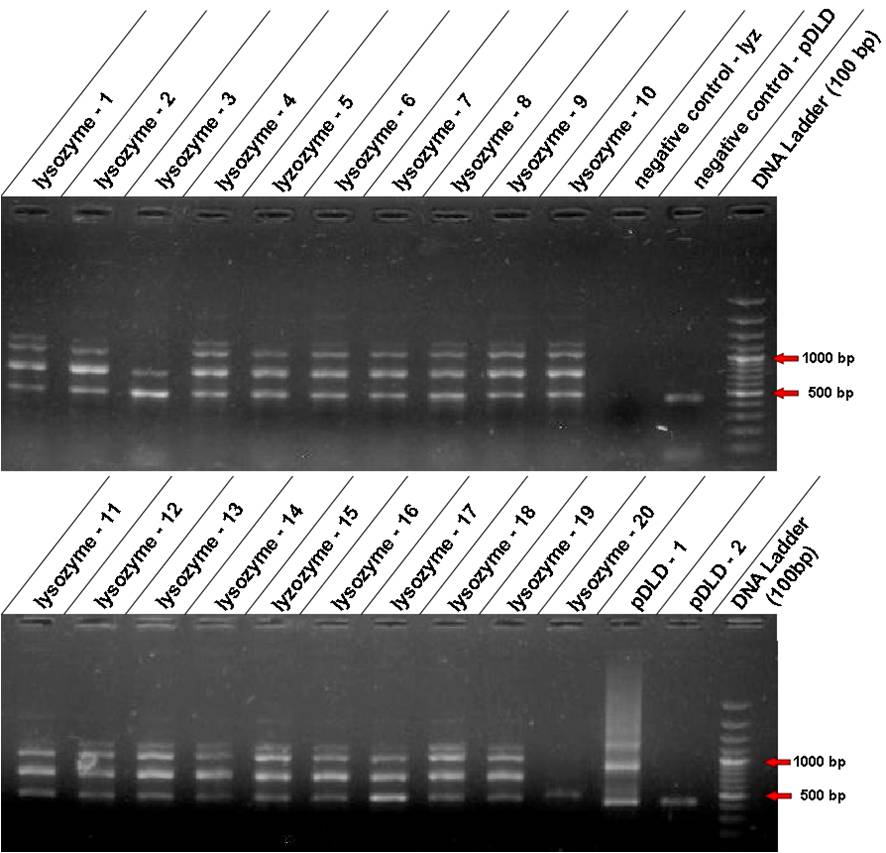
| - | * | + | * The plate of ''E. coli'' transformed with the pDLD+Biofusion had only two colonies. We did the colony PCR to find correct constructions of the pDLD and Lysozyme in Biofusion. We found two fragments corresponding to pDLD, but the negative control amplified the same fragment too! We decided to do miniprep of both colonies and re-confirm it by digestion. |
| - | * We found one expected band of lysozyme. We did miniprep of this colony and chose an inespecific one as well. | + | * We found one expected band of lysozyme. We did miniprep of this colony and chose an inespecific one as well. |
[[Image:lisozima1-20.jpg|600px|center]] | [[Image:lisozima1-20.jpg|600px|center]] | ||
| - | * | + | * JENorf+pGEM grew!! Later that day we found out that we forgot to plate X-Gal =/ We did more plates, this time with X-Gal!! Waste of time!! =( |
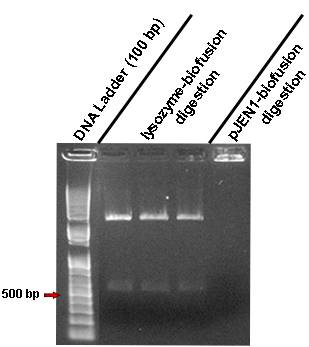
| - | * | + | * We digested the Lysozyme+Biofusion vector and pJEN1+Biofusion vector with ''Xba''I and ''Pst''I to recover the part fragment with one ''Not''I site. The Lysozyme digestion worked so we purified the correct fragment, ligated it in biofusion again in order to recover the second ''Not''I site. Unfortunately the pJEN1 digestion didn’t work. |
[[Image:lisozima-biofusion.jpg|300px|center]] | [[Image:lisozima-biofusion.jpg|300px|center]] | ||
| - | * | + | * We inoculated pJEN1+biofusion and pDLD+biofusion in liquid media to do miniprep tomorrow. |
====pADH1+YFP==== | ====pADH1+YFP==== | ||
| - | * | + | * An electrophoresis was made with the product of the digestion performed yesterday. Then, we purified the bands corresponding to the size of the pADH1 in the biofusion vector and the YFP part. |
| - | * | + | |
| + | * So we made the ligation of the YFP part in the biofusion vector that contains the ADH1 promoter. Then we transformed this construction in ''E. coli'' that has grown O/N in the kanamicin media plate. | ||
''Wesley and Gleidson'' | ''Wesley and Gleidson'' | ||
| Line 77: | Line 83: | ||
====YEP358==== | ====YEP358==== | ||
| - | * | + | * We had positive colinies with YEP358-β -galactosidase ! |
{{:Team:UNICAMP-Brazil/inc_rodape}} | {{:Team:UNICAMP-Brazil/inc_rodape}} | ||
Latest revision as of 02:39, 22 October 2009
| ||||||||||||||||||||||||||||||||||
 "
"