Team:TUDelft/4 September 2009
Lab Notebook
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
4 September 2009
Sriram
Today I minipreped the samples Final Riboregulator plasmid 1 and intermediate assemblies of locks and keys. The I continued with the assembly of the lock and key so that on monday I will get colonies of locks and keys.
Tim Weenink
I transformed all four batches of competent cells with !SIII ligation mix. Plates (Cam) were left on the bench in a plastic bag over the weekend. All the time constants in the electroporation were 4.4 or 4.5 (which is good). Also two plates without selection marker were streaked to check if the competent cells were viable at all.
Saeed
I made minipreps form 6 overnight cultures of !P in order to check the assembly of !P. These are called !P1 to !P6.
| Part | [DNA] ng/uL |
| !P1 | 1001.1 |
| !P2 | 92.3 |
| !P3 | 98.0 |
| !P4 | 102.2 |
| !P5 | 89.1 |
| !P6 | 86.1 |
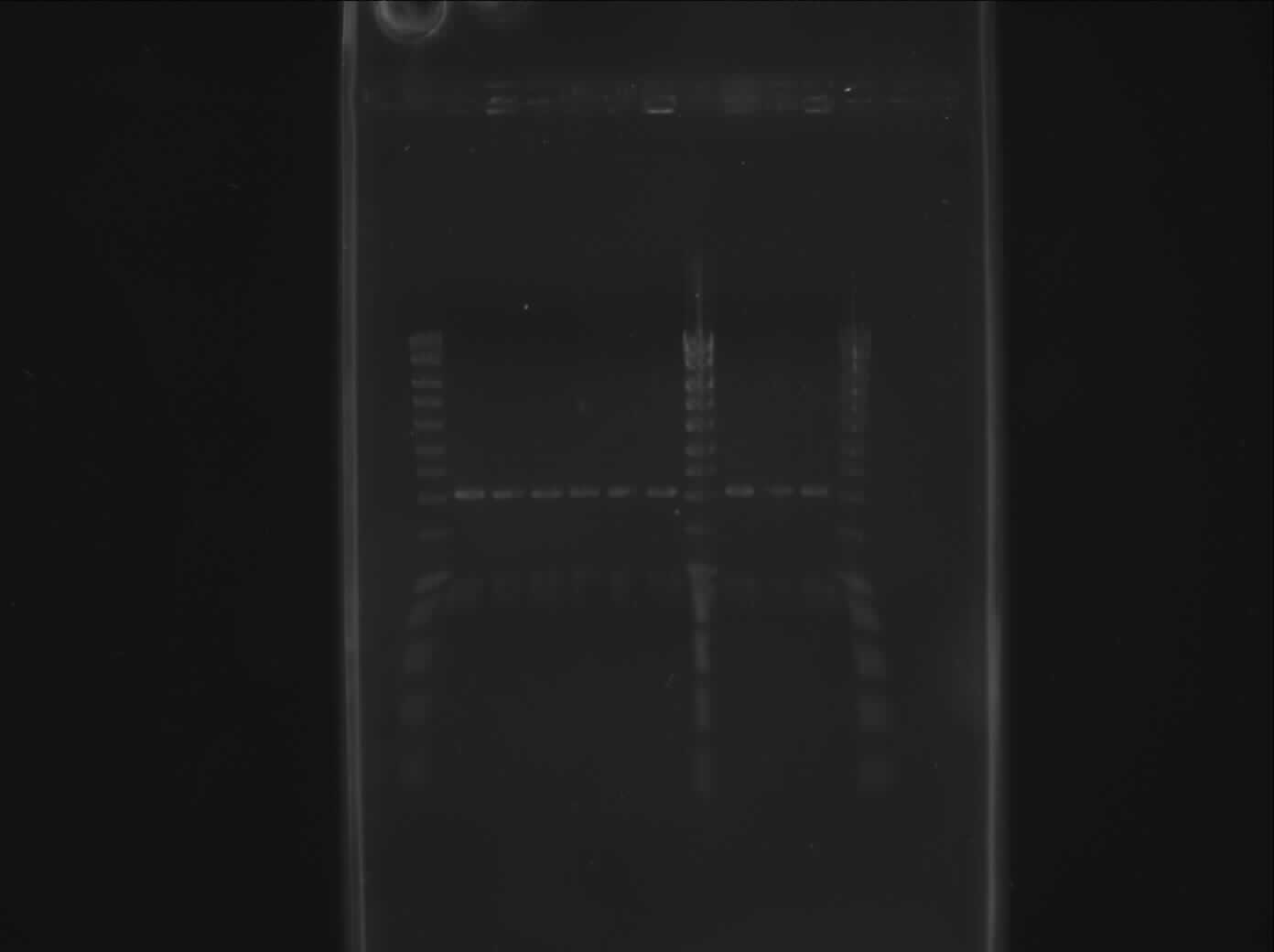
The isolated plasmide were restricted with E and P in order to check fragment size. !P1, !P3 and !P5 are also cut upstream and analysed on 1% agarose gel.
| lane | Part | Expected Plasmid Size | Status |
| 1 | [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] | ||
| 2 | !P1 | 833 | ok |
| 3 | !P2 | 833 | ok |
| 4 | !P3 | 833 | ok |
| 5 | !P4 | 833 | ok |
| 6 | !P5 | 833 | ok |
| 7 | !P6 | 833 | ok |
| 8 | [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] | ||
| 9 | !P1 upstream | 833 | ok |
| 10 | !P3 upstream | 833 | ok |
| 11 | !P5 upstream | 833 | ok |
| 12 | [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] |
The picture is not realy clear but the 833 bp fragment was visible. !P1 will be used for the next assembly.
 "
"