Team:TUDelft/23 July 2009
From 2009.igem.org
(→Sriram) |
(→Sriram) |
||
| (20 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{Template:TUDelftiGEM2009}} | {{Template:TUDelftiGEM2009}} | ||
| + | {{Template:TUDelftiGEM2009_LabNotebook}} | ||
| + | |||
| + | ='''23 July 2009'''= | ||
===Tim Weenink=== | ===Tim Weenink=== | ||
| Line 17: | Line 20: | ||
===Sriram=== | ===Sriram=== | ||
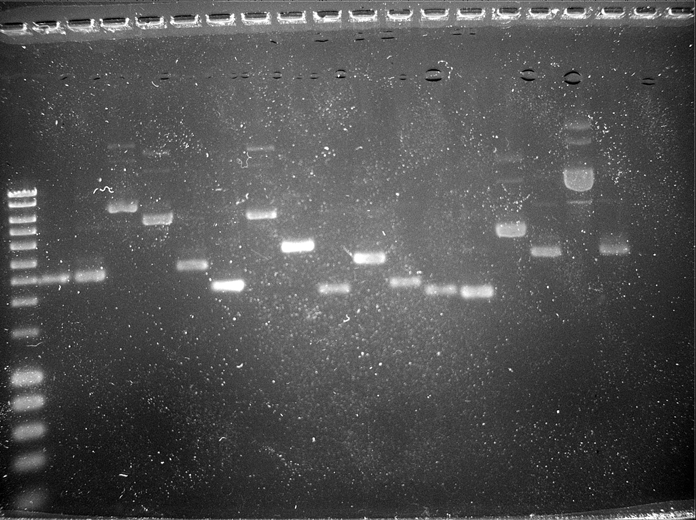
| - | + | The agarose gel electrophoresis was run for another 1 hour and 10 minutes at 110 V. Then the gel was immersed in TBE buffer stained with 20µl of Safe Green gel stain for 1 hour and the bands were viewed to check whether we have the right biobricks. By this we confirmed that all the amplified biobricks to be used in Delay were extracted well and they are pure. | |
| + | |||
| + | [[Image:Gel22072009-delay-conj.png|center|thumb|450px]] | ||
| + | |||
| + | |||
| + | {| border="1" align="center" | ||
| + | | Well || Part || Expected Plasmid Size || Status | ||
| + | |- align="center" | ||
| + | | 1 || [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] || || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 2 || BBa_C0040 TetR || 2739 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 3 || BBa_ E0040 GFP || 2799 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 4 || BBa_E1010 mRFPI || 5106 | ||
| + | |- align="center" | ||
| + | | 5 || BBa_I12006 pLambclin || 4507 | ||
| + | |- align="center" | ||
| + | | 6 || BBa_C0051 cI || 2829 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 7 || BBa_J23008 key3c || 2450 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 8 || BBa_I12006 pLambclin star || 4507 | ||
| + | |- align="center" | ||
| + | | 9 || BBa_B0015 Double Terminator || 3318 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 10 || BBa_R0010 pLacI || 2279 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 11 || BBa_K081013 RBS-cI-RBS || 2898 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 12 || BBa_J23031 lock3c || 2398 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 13 || BBa_R0040 pTet || 2133 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 14 || BBa_B0034 RBS || 2091 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 15 || BBa_I714031 OriT-R || 2357 || <font color=red>✖</font> | ||
| + | |- align="center" | ||
| + | | 16 || BBa_E0840 GFP generator || 2957 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 17 || BBa_J23100 strong promoter || 2948 || <font color=red>✖</font> | ||
| + | |- align="center" | ||
| + | | 18 || BBa_I13522 pTet GFP || 3016 || <font color=limegreen>✔</font> | ||
| + | |} | ||
| + | |||
| + | |||
| + | For preparing glycerol stocks and plasmid DNA extraction the 7 biobrick colonies [R0010, R0040, J23008, J23031, B0034, B0015, K081013] from the LB agar plates and 2 composite biobricks [S03335, S03473] received from iGEM HQ were cultured in 5 ml tubes with 1xAmp. The 7 biobricks were recultured since we anticipate that we may need more DNA for parts like RBS, Double terminator, etc. in more assemblies and also glycerol stocks were not made for these biobricks. | ||
| + | |||
| + | The 2 composite biobricks [S03335, S03473] were also streaked in LB Agar plates with 1xAmp for future use. | ||
| + | |||
| + | ===Orr=== | ||
| + | Prepared TBE and LB Agar mediums using the standard protocols. | ||
| + | |||
| + | Looked at the negative feedback Matlab code in an attempt to write a new code that will enable us to change two variables at a time and check a set of values to find the optimal value for each parameter. | ||
| + | |||
| - | + | ===Calin=== | |
| + | Gel showed that oriT and promoter miniprep didnt go right. Cultured oriT, promoter, GFP gen in 5 mL tubes. Made 2 GFP plates, an RFP plate, and one with both. Plated parts from MIT (BBa_J23015, BBa_J23055). Added composite part with GFP and oriT to sandbox. Acquired some 20% L-arabinose. Looked into the proofreading PCR kit in the lab for possible use in the knockout (Roche Expand High Fidelity PCR System). | ||
| - | |||
{{Template:TUDelftiGEM2009_end}} | {{Template:TUDelftiGEM2009_end}} | ||
Latest revision as of 10:28, 12 October 2009
Lab Notebook
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
23 July 2009
Tim Weenink
Details of the following to be provided tomorrow:
grew up *S in LC
looked at gel
Did !A assembly again with double the amount of backbone (and rest from fridge). Did it with both DH5alpha and homemade top10 chemically competent cells, to compare transformation efficiencies. Also the right concentration of K was used
PCR'd 8 colonies from my transformation plates with VF2 and I-SceI Reverse primers to see if there was incorporation of the I-SceI restriction site
isolated plasmid from *S
Sriram
The agarose gel electrophoresis was run for another 1 hour and 10 minutes at 110 V. Then the gel was immersed in TBE buffer stained with 20µl of Safe Green gel stain for 1 hour and the bands were viewed to check whether we have the right biobricks. By this we confirmed that all the amplified biobricks to be used in Delay were extracted well and they are pure.
| Well | Part | Expected Plasmid Size | Status |
| 1 | [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] | ✔ | |
| 2 | BBa_C0040 TetR | 2739 | ✔ |
| 3 | BBa_ E0040 GFP | 2799 | ✔ |
| 4 | BBa_E1010 mRFPI | 5106 | |
| 5 | BBa_I12006 pLambclin | 4507 | |
| 6 | BBa_C0051 cI | 2829 | ✔ |
| 7 | BBa_J23008 key3c | 2450 | ✔ |
| 8 | BBa_I12006 pLambclin star | 4507 | |
| 9 | BBa_B0015 Double Terminator | 3318 | ✔ |
| 10 | BBa_R0010 pLacI | 2279 | ✔ |
| 11 | BBa_K081013 RBS-cI-RBS | 2898 | ✔ |
| 12 | BBa_J23031 lock3c | 2398 | ✔ |
| 13 | BBa_R0040 pTet | 2133 | ✔ |
| 14 | BBa_B0034 RBS | 2091 | ✔ |
| 15 | BBa_I714031 OriT-R | 2357 | ✖ |
| 16 | BBa_E0840 GFP generator | 2957 | ✔ |
| 17 | BBa_J23100 strong promoter | 2948 | ✖ |
| 18 | BBa_I13522 pTet GFP | 3016 | ✔ |
For preparing glycerol stocks and plasmid DNA extraction the 7 biobrick colonies [R0010, R0040, J23008, J23031, B0034, B0015, K081013] from the LB agar plates and 2 composite biobricks [S03335, S03473] received from iGEM HQ were cultured in 5 ml tubes with 1xAmp. The 7 biobricks were recultured since we anticipate that we may need more DNA for parts like RBS, Double terminator, etc. in more assemblies and also glycerol stocks were not made for these biobricks.
The 2 composite biobricks [S03335, S03473] were also streaked in LB Agar plates with 1xAmp for future use.
Orr
Prepared TBE and LB Agar mediums using the standard protocols.
Looked at the negative feedback Matlab code in an attempt to write a new code that will enable us to change two variables at a time and check a set of values to find the optimal value for each parameter.
Calin
Gel showed that oriT and promoter miniprep didnt go right. Cultured oriT, promoter, GFP gen in 5 mL tubes. Made 2 GFP plates, an RFP plate, and one with both. Plated parts from MIT (BBa_J23015, BBa_J23055). Added composite part with GFP and oriT to sandbox. Acquired some 20% L-arabinose. Looked into the proofreading PCR kit in the lab for possible use in the knockout (Roche Expand High Fidelity PCR System).
 "
"