Team:TUDelft/10 August 2009
From 2009.igem.org
(Difference between revisions)
(New page: {{Template:TUDelftiGEM2009}} {{Template:TUDelftiGEM2009_LabNotebook}} ='''10 August 2009'''= {{Template:TUDelftiGEM2009_end}}) |
(→10 August 2009) |
||
| Line 4: | Line 4: | ||
='''10 August 2009'''= | ='''10 August 2009'''= | ||
| + | |||
| + | ===Calin=== | ||
| + | |||
| + | Knockout plasmids pKD3, pKD4, pKD46 arrived. Plates and tubes made for all with Amp. pKD46 kept at 30 C. | ||
| + | |||
| + | trbK plasmid from BaseClear was digested first with HindIII (no BSA) and after with BamHI (1 hr incubation, 20 min inactivation). pSB1A3 was digested with E+P (6uL DNA, 36.5uL H2O). pSB4A5 was digested with E+P (6.5uL DNA, 36uL H2O). | ||
| + | |||
| + | 2% Gel was run, Ladder was accidentally used instead of dye. | ||
| + | |||
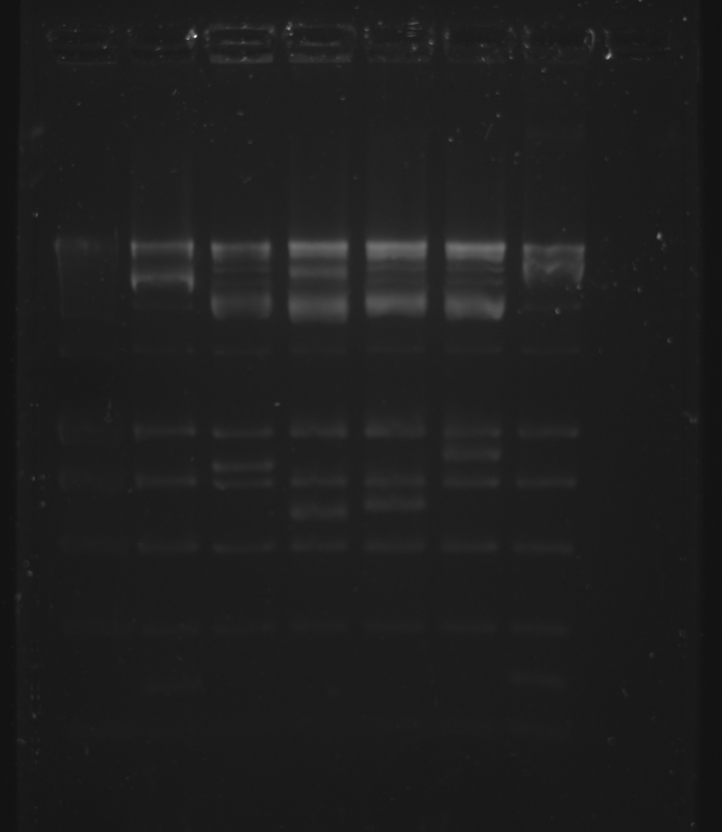
| + | [[Image:Gel10082009-conjdigest.png|600px]] | ||
| + | |||
| + | |||
| + | {| border="1" align="center" | ||
| + | | Well || Part || Expected Plasmid Size || Status | ||
| + | |- align="center" | ||
| + | | 1 || [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] || || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 2 || trbK BamHI + HindIII digest || 282 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 3 || pSB1A3 E+P digest || 2114 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 4 || pSB4A5 E+P digest || 3395 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 5 || pSB1C3 E+P digest || 2072 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 6 || GFP-gen E+S digest || 878 || <font color=limegreen>✔</font> | ||
| + | |- align="center" | ||
| + | | 7 || oriT-R X+P digest || 278 || <font color=limegreen>✔</font> | ||
| + | |} | ||
| + | |||
| + | The appropriate bands were cut out with a scalpel under the blue light imager and the QIAGEN Gel Extraction kit was used. | ||
| + | |||
| + | Small colonies on the pTet-RBS plate grew larger by end of the day. Daniel re-transformed pTet-RBS. | ||
| + | |||
| + | 5 mL tubes were made by Sriram for: | ||
| + | * Assembly CA | ||
| + | * pTet-RBS | ||
| + | |||
| + | Also a 50 mL R751 culture with 2 x TRI was made. | ||
| + | |||
{{Template:TUDelftiGEM2009_end}} | {{Template:TUDelftiGEM2009_end}} | ||
Revision as of 18:15, 10 August 2009
Lab Notebook
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
10 August 2009
Calin
Knockout plasmids pKD3, pKD4, pKD46 arrived. Plates and tubes made for all with Amp. pKD46 kept at 30 C.
trbK plasmid from BaseClear was digested first with HindIII (no BSA) and after with BamHI (1 hr incubation, 20 min inactivation). pSB1A3 was digested with E+P (6uL DNA, 36.5uL H2O). pSB4A5 was digested with E+P (6.5uL DNA, 36uL H2O).
2% Gel was run, Ladder was accidentally used instead of dye.
| Well | Part | Expected Plasmid Size | Status |
| 1 | [http://www.eurogentec.com/EGT/Images/RESALES/Electrophoresis/Regular%20DNA%20Ladder/7-SmartLadder.jpg DNA Ladder] | ✔ | |
| 2 | trbK BamHI + HindIII digest | 282 | ✔ |
| 3 | pSB1A3 E+P digest | 2114 | ✔ |
| 4 | pSB4A5 E+P digest | 3395 | ✔ |
| 5 | pSB1C3 E+P digest | 2072 | ✔ |
| 6 | GFP-gen E+S digest | 878 | ✔ |
| 7 | oriT-R X+P digest | 278 | ✔ |
The appropriate bands were cut out with a scalpel under the blue light imager and the QIAGEN Gel Extraction kit was used.
Small colonies on the pTet-RBS plate grew larger by end of the day. Daniel re-transformed pTet-RBS.
5 mL tubes were made by Sriram for:
- Assembly CA
- pTet-RBS
Also a 50 mL R751 culture with 2 x TRI was made.
 "
"