User:DavidC/15 September 2009
From 2009.igem.org
(New page: {{Template:Supbiotechcss6.css}} {{Template:SupbiotechparisEn}} <center> <div style="color: black; margin-bottom: 30px; width: 500px;"> {|align="center" ||{{#calendar: title=User:DavidC |...) |
|||
| (One intermediate revision not shown) | |||
| Line 12: | Line 12: | ||
</div> | </div> | ||
</center> | </center> | ||
| + | |||
| + | |||
| + | === Tuesday the 15th === | ||
| + | |||
| + | ==== Restriction digest ==== | ||
| + | |||
| + | ==== Ligation between BBa_I712669 and BBa_B0014: ==== | ||
| + | |||
| + | I712669 (= 6,667µg/µL) restriction digest with PstI and XbaI (1380bp) : <br> | ||
| + | |||
| + | DNA (10µg final) = 1,5µL <br> | ||
| + | Buffer 3 (NEB) = 5µL <br> | ||
| + | H20 = 41,5µL <br> | ||
| + | Pst 1 = 1µL <br> | ||
| + | Xba 1 = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
| + | BBa_B0014 (= 2,381µg/µL) restriction digest by PstI and SpeI (3284bp): <br> | ||
| + | |||
| + | DNA (10µg final) = 4,2µL <br> | ||
| + | Buffer 2 (NEB) = 5µL <br> | ||
| + | H20 = 38,8µL <br> | ||
| + | Pst 1 = 1µL <br> | ||
| + | Spe 1 = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
| + | ==== Ligation between BBa_B0014 and BBa_P1003 ==== | ||
| + | |||
| + | BBa_P1003 (= 2,17µg/µL) restriction digest by PstI and SpeI (3675bp): <br> | ||
| + | |||
| + | DNA (10µg final) = 2,4µL <br> | ||
| + | Buffer 3 (NEB) = 5µL <br> | ||
| + | H20 = 40,6µL <br> | ||
| + | Pst 1 = 1µL <br> | ||
| + | Spe1 = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
| + | BBa_B0014 (=2,38µg/µL) by PstI and XbaI (95bp): <br> | ||
| + | |||
| + | DNA (10µg final) = 4,2µL <br> | ||
| + | Buffer 2 (NEB) = 5µL <br> | ||
| + | H20 = 38,8µL <br> | ||
| + | Pst1 = 1µL <br> | ||
| + | Xba1 = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
| + | ==== Ligation between BBa_J23100 with BBa_B0014 ==== | ||
| + | |||
| + | BBa_J23100 (=1,67µg/µL) restriction digest by EcoRI and XbaI (35bp): <br> | ||
| + | DNA (10µg final) = 6µL <br> | ||
| + | Buffer M (TAKARA) = 5µL <br> | ||
| + | H20 = 7µL <br> | ||
| + | EcoRI (TAKARA) = 1µL <br> | ||
| + | Xba 1 (TAKARA) = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
| + | BBa_B0014 (= 2,81µg/µL) restriction digest by PstI and SpeI (3284bp): <br> | ||
| + | |||
| + | DNA (10µg final) = 4,2µL <br> | ||
| + | Buffer H (TAKARA) = 5µL <br> | ||
| + | H20 = 38,8µL <br> | ||
| + | Pst 1 (TAKARA) = 1µL <br> | ||
| + | Spe 1 (TAKARA) = 1µL <br> | ||
| + | Incubation 1h at 37°C. <br> | ||
| + | |||
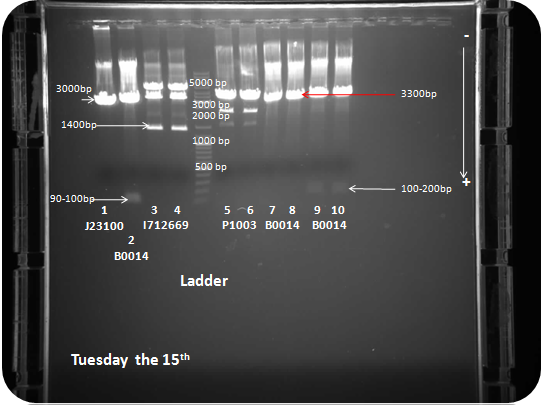
| + | ==== DNA electrophoresis ==== | ||
| + | |||
| + | 85 Volt, 15 minutes. <br> | ||
| + | 105 Volt, 40 minutes. <br> | ||
| + | Ladder fermentas 1 Kb. <br> | ||
| + | |||
| + | |||
| + | |||
| + | Samples: BBa_J23100, BBa_B0014, BBa_I712669, BBa_P1003, BBa_B0014, BBa_B0014. <br> | ||
| + | |||
| + | [[image:TU1509.png|center]] <br> | ||
| + | |||
| + | ==== DNA purification ==== | ||
| + | |||
| + | Kit Qiagen “gel extraction kit”, final volume = 50µL. <br> | ||
Latest revision as of 14:51, 21 October 2009
Contents |
Tuesday the 15th
Restriction digest
Ligation between BBa_I712669 and BBa_B0014:
I712669 (= 6,667µg/µL) restriction digest with PstI and XbaI (1380bp) :
DNA (10µg final) = 1,5µL
Buffer 3 (NEB) = 5µL
H20 = 41,5µL
Pst 1 = 1µL
Xba 1 = 1µL
Incubation 1h at 37°C.
BBa_B0014 (= 2,381µg/µL) restriction digest by PstI and SpeI (3284bp):
DNA (10µg final) = 4,2µL
Buffer 2 (NEB) = 5µL
H20 = 38,8µL
Pst 1 = 1µL
Spe 1 = 1µL
Incubation 1h at 37°C.
Ligation between BBa_B0014 and BBa_P1003
BBa_P1003 (= 2,17µg/µL) restriction digest by PstI and SpeI (3675bp):
DNA (10µg final) = 2,4µL
Buffer 3 (NEB) = 5µL
H20 = 40,6µL
Pst 1 = 1µL
Spe1 = 1µL
Incubation 1h at 37°C.
BBa_B0014 (=2,38µg/µL) by PstI and XbaI (95bp):
DNA (10µg final) = 4,2µL
Buffer 2 (NEB) = 5µL
H20 = 38,8µL
Pst1 = 1µL
Xba1 = 1µL
Incubation 1h at 37°C.
Ligation between BBa_J23100 with BBa_B0014
BBa_J23100 (=1,67µg/µL) restriction digest by EcoRI and XbaI (35bp):
DNA (10µg final) = 6µL
Buffer M (TAKARA) = 5µL
H20 = 7µL
EcoRI (TAKARA) = 1µL
Xba 1 (TAKARA) = 1µL
Incubation 1h at 37°C.
BBa_B0014 (= 2,81µg/µL) restriction digest by PstI and SpeI (3284bp):
DNA (10µg final) = 4,2µL
Buffer H (TAKARA) = 5µL
H20 = 38,8µL
Pst 1 (TAKARA) = 1µL
Spe 1 (TAKARA) = 1µL
Incubation 1h at 37°C.
DNA electrophoresis
85 Volt, 15 minutes.
105 Volt, 40 minutes.
Ladder fermentas 1 Kb.
Samples: BBa_J23100, BBa_B0014, BBa_I712669, BBa_P1003, BBa_B0014, BBa_B0014.
DNA purification
Kit Qiagen “gel extraction kit”, final volume = 50µL.
 "
"