Team:Groningen/Project/WholeSystem
From 2009.igem.org
Whole SystemThe metal scavenger with a vertical gas drive is a modular system. The system is explored for arsenic in this project. The accumulation module was made out of the aquaglycerol porin, GlpF, that was used as importer and the sequestering protein fMT. On a complementary plasmid the gas vesicle protein (GVP) cluster was cloned downstream of the arsenic sensitive promotor ars. The two plasmids were transformed in one cell and analysed using the buoyancy assay. This showed succesfull transformation and a buoyancy phenotype.
|
Introduction
The metal scavenger with a vertical gas drive is a modular system which contains five modules; the transporter, an accumulation protein, a regulated promoter, a regulator and the gas vesicle cluster. The transporter imports the metal ion of choice. An accumulation protein facilitates accumulation of metal ions and prevents the cell from dying of metal toxicity. The imported metal ion also acts as a regulator for the metal sensitive promotor which activates the expression of the gvp-cluster. Thereby the recombinant bacterium, with this system, accumulates metal and upon accumulation starts to float which is convenient in bioremediation or mining applications. In principle this system may be used in order to accumulate any component for which these modules are available!
The system explored in this project was mainly based on arsenic, however, aspects of a copper/zinc system were also explored. For the copper/zinc system HmtA, () was chosen as transporter, Mymt and SmtA as accumulators and CueO, () and ZntR, () as metal sensitive promotors.
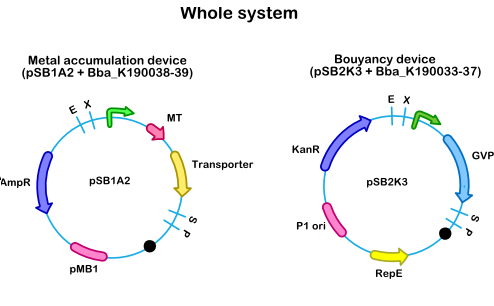
Arsenic is a very toxic metal and causes health problems all over the world. The transporter used in this system is GlpF () a glycerol transported known to import arsenic as well (Meng, YL, et al.2004), (Rosen, BR, et al.2009). Two accumulation proteins were chosen to accumulate arsenic, the fusion protein MBP-ArsR () and fMT (). Both accumulation proteins were combined with GlpF to create new accumulation devices (, ). As metal sensitive promotor, pArsR was chosen and together with the gvp cluster it makes up a buoyancy device (). The two devices, the accumulation device and the buoyancy device were transformed together in E. coli, to create the metal scavenger with a vertical gas drive.
Cloning Strategy
In order to use the buoyancy device() in combination with the accumulation device (, ) a two vector system was used (see Figure 1). A two vector system was chosen because it is not feasible to combine the 6kb gas vesicle cluster, with a 1kb transporter and 300bp metallothionein and their promoters in one vector. This could easily increase the size of a vector to 10kb, a vector size which can hardly be transformed to E. coli. This two vector system is composed of a vector with the accumulation device(, ) and a vector with the buoyancy device (). This combination was chosen because this makes it possible to tranform both vectors in one E.coli cell, since the vectors contain different origin of replications and antibiotic resistance markers. They contain respectively a pMB1 origin of replication and a P1 ori.
- Figure 1: Whole system, combining the bouyancy device on a pSB2K3 vector and the accumulation device on a pSB1AC3 vector
Results
Combining parts and was done by using a normal transformation protocol with both ampicillin and kanamycin as antibiotics. A buoyancy test was performed as described also using both ampicillin and kanamycin as antibiotics. IPTG was also added to the dayculture to induce the part. In exponential phase 10μM NaAsO2 was added to half of the samples. A restriction was done to check the transformation.
Restriction
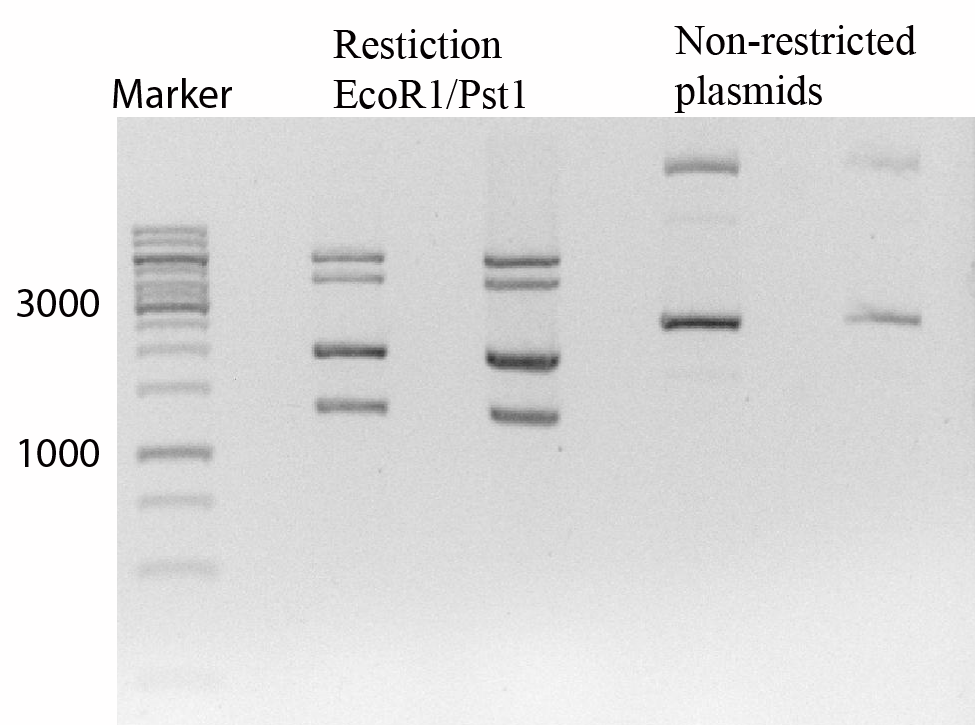
Figure 2 shows that the transformation of both vectors succeeded.
- Figure 2:Gel image of restricted and not restricted plasmids.
The gel shows a 1Kb marker to the far left, after that two times a restriction with EcoRI and PstI can be seen. This shows clearly fragments of 6000pb and 4500pb for the and fragments of 2000pb and 1300 bp for the . The next two slots show non-restricted plasmids, two distinc bands can be seen clearly indicating the presence of two plasmids.
Buoyancy test
The buoyancy test elegantly shows that combining both an accumulation device and the GVP-buoyancy device allows buoyancy.
- Figure 3: Result of the buoyancy test, the left shows E.coli cells without arsenic induction and to the right floating E.coli cells, induced with arsenic, are shown.
On the left E. coli cells (containing the whole system) without arsenic can be seen and on the right E. coli cells (containing the whole system) with arsenic is shown. It can be seen that the right sample floats. This indicates that the GlpF is transporting the arsenic inside the cells, the fMT accumulates it (otherwise the cells would be dead) and the GVP was induced by the arsenic so the cells start to float.
- Figure 4: Results of the buoyancy test, left E.coli with the GVP plasmid, the right tube shows E.coli cells with the fMT-glpF device.
As a control E.coli cells were also transformed with the GVP plasmid and others with the fMT-GlpF plasmid. Figure 4 shows that the E.coli cells with GVP float whereas the fMT-GlpF containing ones seem to sink.
Conclusion
From the restriction analysis it can be concluded that the transformation of E. coli DH10B with the two vectors was successfull. The strain was growing slowly, but was viable. The buoyancy test proved the functionality of the buoyancy phenotype but whether the presence of the accumulation device influences the GVP expression can not be determined with this experiment. The functionality of the accumulation device will be determined by the arsenic uptake assay and analyzed by ICP-MS.
 "
"