Team:Groningen/Notebook/14 July 2009
From 2009.igem.org
m (→GVP Cluster) |
(→Transporters) |
||
| Line 45: | Line 45: | ||
===Transporters=== | ===Transporters=== | ||
| + | '''3nd PCR HmtA_Fw/HmtA_Rev''' | ||
| + | We have no products on our gel but we do have ON cultures, we picked 3 for isolation. | ||
| + | Today we have the other primers as well. Since we are almost out of template DNA we preform colony PCR on the Colonies of the HmtA plate 3. With a dynazyme master mix (2 samples picked) and homebrew (2 samples picked). To see which ON culture to isolate plasmide from. and continue Cloning by mutating with mutation primer on the PCR product. | ||
| + | |||
| + | {| border="1" | ||
| + | |+ '''MasterMix3''' | ||
| + | ! Component !! Mg/K | ||
| + | |- | ||
| + | ! MQ | ||
| + | | 28.5 uL | ||
| + | |- | ||
| + | ! F mut1 | ||
| + | | 2 uL | ||
| + | |- | ||
| + | ! R mut2 | ||
| + | | 2 uL | ||
| + | |- | ||
| + | ! dNTP | ||
| + | | 2 uL | ||
| + | |- | ||
| + | ! MgCl | ||
| + | | 3 uL | ||
| + | |- | ||
| + | ! taq buffer KCl | ||
| + | | 5 uL | ||
| + | |- | ||
| + | ! taq buffer (NH)SO | ||
| + | | 5 uL | ||
| + | |- | ||
| + | ! taq polymerase | ||
| + | | 2.5 uL | ||
| + | |- | ||
| + | |} | ||
| + | |||
| + | |||
| + | {| | ||
| + | ! PCR 3 program | ||
| + | ! Temperature | ||
| + | ! Time | ||
| + | |- | ||
| + | |Denaturing | ||
| + | |95° | ||
| + | |2.00 min | ||
| + | |- | ||
| + | | | ||
| + | |Start Cycles 25X | ||
| + | |- | ||
| + | |Denaturing | ||
| + | |95° | ||
| + | |30 sec | ||
| + | |- | ||
| + | |Annealing | ||
| + | |55° | ||
| + | |20 sec | ||
| + | |- | ||
| + | |Elongation | ||
| + | |72° | ||
| + | |2.10 min | ||
| + | |- | ||
| + | | | ||
| + | |End cycles | ||
| + | |- | ||
| + | |Final elongation | ||
| + | |72° | ||
| + | |10 min | ||
| + | |- | ||
| + | |Hold | ||
| + | |4° | ||
| + | |Forever | ||
| + | |} | ||
==Dry== | ==Dry== | ||
{{Team:Groningen/Notebook/Day/Footer}} | {{Team:Groningen/Notebook/Day/Footer}} | ||
Revision as of 11:02, 14 July 2009
Wet
GVP Cluster
Plasmid Purification
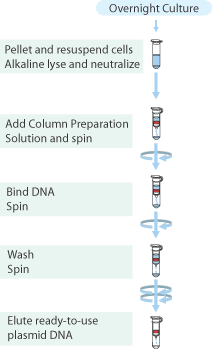
Plasmid isolation was performed on the cultures of GVP and Terminator containing cells with the "Sygma-Aldrich™ GenElute™ Plasmid Miniprep Kit".
- From each tube 5mL of culture was collected in a 2mL cup, and the cells were pelleted by centrifugation for 1 min. at max. speed and the supernatant discarded.
- Cells were resuspended in 200μL Resuspension Solution by up and down pipetting.
- To the mixture 200μL Lysis Solution was added, mixed by inverting the cup, and stored at room temperature for 5 minutes.
- 350μL of Neutralisation Solution was added and the tubes inverted.
- Cell debri was pelleted by centrifugation at full speed for 10 min.
- Column was prepared by adding 500μL Column Preparation Solution and centrifuging for 1 min. full speed
- The lysate was transfered to the column and centrifuged for 1 min. full speed
- Column was washed with both washing solutions
- Plasmids were eluted with 30μL MQ and stored in the fridge
Concentration of Plasmids
The concentration of isolated plasmid was determined with the use of a nano-drop.
GVP eluted in MQ
- 329.7 ng/μL
- 1.84 (260/280)
- 2.13 (260/230)
BBa_J23109 eluted in MQ
- 174.9 ng/μL
- 1.88 (260/280)
- 1.92 (260/230)
BBa_J23100 eluted in MQ
- 155.3 ng/μL
- 1.85 (260/280)
- 1.79 (260/230)
BBa_J23106 eluted in MQ
- 219.4 ng/μL
- 1.86 (260/280)
- 1.93 (260/230)
Transporters
3nd PCR HmtA_Fw/HmtA_Rev We have no products on our gel but we do have ON cultures, we picked 3 for isolation.
Today we have the other primers as well. Since we are almost out of template DNA we preform colony PCR on the Colonies of the HmtA plate 3. With a dynazyme master mix (2 samples picked) and homebrew (2 samples picked). To see which ON culture to isolate plasmide from. and continue Cloning by mutating with mutation primer on the PCR product.
| Component | Mg/K |
|---|---|
| MQ | 28.5 uL |
| F mut1 | 2 uL |
| R mut2 | 2 uL |
| dNTP | 2 uL |
| MgCl | 3 uL |
| taq buffer KCl | 5 uL |
| taq buffer (NH)SO | 5 uL |
| taq polymerase | 2.5 uL |
| PCR 3 program | Temperature | Time |
|---|---|---|
| Denaturing | 95° | 2.00 min |
| Start Cycles 25X | ||
| Denaturing | 95° | 30 sec |
| Annealing | 55° | 20 sec |
| Elongation | 72° | 2.10 min |
| End cycles | ||
| Final elongation | 72° | 10 min |
| Hold | 4° | Forever |
Dry
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 "
"