Wisconsin-Madison/25 June 2009
From 2009.igem.org
Calendar
|
|
|
|
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
June 25, 2009
Other:
Reorganized -4 C fridge (plates) and -20 C freezer (samples)
Advisor Meeting
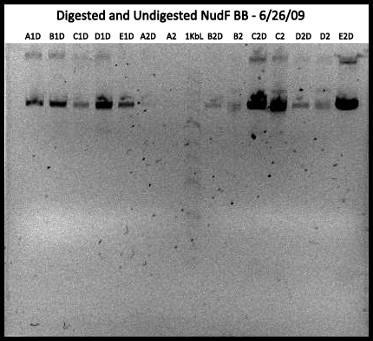
Ex: Miniprep of Suspected BB NudF: 5 different colonies (A-E): 2 of each colony (10samples)
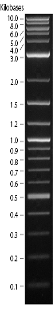
Ran 0.7% Gel:
Cut with Xba Expected size:~2700bp
Results: Does not mean much …
Need better controls:
1. BB cut nudF + backbone separate
2. Backbone
3. NudF
If inconclusive (several NudF’s have ligated together):
1. one single cut for entire BioBrick inside NudF
2. if multiple NudF’s = more than one bands
3. THIS IS P2 (BB*) – DUMB FOOLS
Ex 15: Inducing various Modified (Triple) E.Coli Strain Cultures 3
Transformed Confirmed Triple DNA into following strains and plated:
1. BL21 (DE3)
2. K-12 MG1655 AaraBAD
3. K-12 MG1655 (wildtype)
4. DH10B
Ex 17: GFP Regulation by ProU Promoter
Test 2:
Redo Salt Tests GFP,ProU BB
Conc. / Culture (uL) / 5M NaCl (uL)
0.0 / 200 / -
0.2 / 192 / 8
0.4 / 184 / 16
0.6 / 176 / 24
0.8 / 168 / 32
1.0 / 160 / 40
Well Assignment:
Conc. / Wells
0.0 / D5 / E8 / C6 / F9
0.2 / E5 / D8 / F6 / C5
0.4 / D6 / F4 / C8 / F8
0.6 / E9 / D5 / C7 / F7
0.8 / D9 / E4 / D7 / C4
1.0 / C9 / F5 / E6 / E8
All other wells were filled with MilliQ water
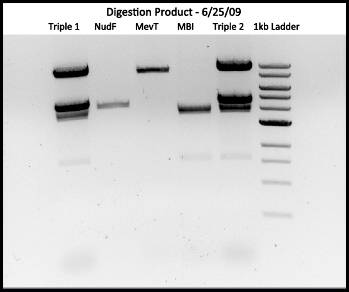
Ex 14:
Digestion of Trip 1, Trip 2, MevT, MBI, NudF 18 uL RXN:
1. 12uL H2O
2. 1.8uL Buffer 3
3. 0.18uL Protein BSA
4. 3uL DNA
Ran on 0.7% Agarose Gel
pNudF 4716 bp
pMevT 8593 bp
pMBI 6000 bp --> (cut twice: 4000, 2000)
Results: We have triple transformation of interest
Ex 10: Cyanobacteria
Set up cyanobacteria apparatus
Inoculated PCC6803
Obtained Synechocystis sp PCC 6803 from Dr. Jenny Reed (inoculated 1 mL BG11 with 200uL of frozen stock) - NP
 "
"